Abstract
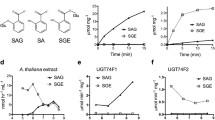
The phenylpropanoid pathway is used in biosynthesis of a wide range of soluble secondary metabolites including hydroxycinnamic acid esters, flavonoids and the precursors of lignin and lignans. In Arabidopsis thaliana a small cluster of three closely related genes, UGT72E1–E3, encode glycosyltransferases (GTs) that glucosylate phenylpropanoids in vitro. This study explores the effect of constitutively over-expressing two of these GTs (UGT72E1 and E3) in planta using the CaMV-35S promoter to determine whether phenylpropanoid homeostasis can be altered in a similar manner to that achieved by over-expression of UGT72E2 as previously reported. The data show that impact of over-expressing UGT72E3 in leaves is highly similar to that of UGT72E2 in that the production of massive levels of coniferyl and sinapyl alcohol 4-O-glucosides and a substantial loss in sinapoyl malate. In contrast, the over-expression of UGT72E1 in leaves led only to minimal changes in coniferyl alcohol 4-O-glucoside and no effect was observed on sinapoyl malate levels. In roots, over-expression of both UGTs led to some increase in the accumulation of the two glucosides. The cell specificity expression of the whole UGT72E gene cluster was investigated and interestingly only UGT72E3 was found to be wound and touch responsive.





Similar content being viewed by others
Abbreviations
- GT:
-
Glycosyltransferase
- OE:
-
Over-expression
- UV:
-
Ultra-violet
References
Anterola AM, Lewis NG (2002) Trends in lignin modification: a comprehensive analysis of the effects of genetic manipulations/mutations on lignification and vascular integrity. Phytochemistry 61:221–294
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Ann Rev Plant Biol 54:519–546
Booker J, Sieberer T, Wright W, Williamson L, Willett B, Stirnberg P, Turnbull C, Srinivasan M, Goddard P, Leyser O (2005) MAX1 encodes a cytochrome P450 family member that acts downstream of MAX3/4 to produce a carotenoid-derived branch-inhibiting hormone. Dev Cell 8:443–449
Bowles DJ, Lim EK, Poppenberger B, Vaistij FE (2006) Glycosyltransferases of lipophilic small molecules. Ann Rev Plant Biol 57:567–597
Capell T, Christou P (2004) Progress in plant metabolic engineering. Curr Opin Biotechnol 15:148–154
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Dixon RA (2005) Engineering of plant natural product pathways. Curr Opin Plant Biol 8:329–336
Dixon RA, Chen F, Guo DJ, Parvathi K (2001) The biosynthesis of monolignols: a “metabolic grid”, or independent pathways to guaiacyl and syringyl units? Phytochemistry 57:1069–1084
Gutierrez RA, Ewing RM, Cherry JM, Green PJ (2002) Identification of unstable transcripts in Arabidopsis by cDNA microarray analysis: Rapid decay is associated with a group of touch- and specific clock-controlled genes. Proc Natl Acad Sci USA 99:11513–11518
Hemm MR, Rider SD Ogas J, Murry DJ, Chapple C (2004) Light induces phenylpropanoid metabolism in Arabidopsis roots. Plant J 38:765–778
Humphreys JM, Chapple C (2002) Rewriting the lignin roadmap. Curr Opin Plant Biol 5:224–229
Kim SJ, Kim KW, Cho MH, Franceschi VR, Davin LB, Lewis NG (2007) Expression of cinnamyl alcohol dehydrogenases and their putative homologues during Arabidopsis thaliana growth and development: lessons for database annotations? Phytochemistry 68:1957–1974
Lanot A, Hodge D, Jackson RG, George GL, Elias L, Lim EK, Vaistij FE, Bowles DJ (2006) The glucosyltransferase UGT72E2 is responsible for monolignol 4-O-glucoside production in Arabidopsis thaliana. Plant J 48:286–295
Lee D, Polisensky DH, Braam J (2005) Genome-wide identification of touch- and darkness-regulated Arabidopsis genes: a focus on calmodulin-like and XTH genes. New Phytol 165:429–444
Li Y, Baldauf S, Lim EK, Bowles DJ (2001) Phylogenetic analysis of the UDP-glycosyltransferase multigene family of Arabidopsis thaliana. J Biol Chem 276:4338–4343
Lim EK, Li Y, Parr A, Jackson RG, Ashford DA, Bowles DJ (2001) Identification of glucosyltransferase genes involved in sinapate metabolism and lignin synthesis in Arabidopsis. J Biol Chem 276:4344–4349
Lim EK, Jackson RG, Bowles DJ (2005) Identification and characterisation of Arabidopsis glycosyltransferases capable of glucosylating coniferyl aldehyde and sinapyl aldehyde. FEBS Lett 579:2802–2806
Mol J, Cornish E, Mason J, Koes R (1999) Novel coloured flowers. Curr Opin Biotechnol 10:198–201
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nair RB, Bastress KL, Ruegger MO, Denault JW, Chapple C (2004) The Arabidopsis thaliana REDUCED EPIDERMAL FLUORESCENCE1 gene encodes an aldehyde dehydrogenase involved in ferulic acid and sinapic acid biosynthesis. Plant Cell 16:544–554
Narsai R, Howell KA, Millar AH, O’Toole N, Small I, Whelan J (2007) Genome-wide analysis of mRNA decay rates and their determinants in Arabidopsis thaliana. Plant Cell 19:3418–3436
Paquette S, Moller BL, Bak S (2003) On the origin of family 1 plant glycosyltransferases. Phytochemistry 62:399–413
Poppenberger B, Fujioka S, Soeno K, George GL, Vaistij FE, Hiranuma S, Seto H, Takatsuto S, Adam G, Yoshida S, Bowles D (2005) The UGT73C5 of Arabidopsis thaliana glucosylates brassinosteroids. Proc Natl Acad Sci USA 103:15253–15258
Priest DM, Ambrose SJ, Vaistij FE, Elias L, Higgins GS, Ross ARS, Abrams SR, Bowles DJ (2006) Use of the glucosyltransferase UGT76B1 to disturb abscisic acid homeostasis in Arabidopsis thaliana. Plant J 46:492–502
Quiel JA, Bender J (2003) Glucose conjugation of anthranilate by the Arabidopsis UGT74F2 glucosyltransferase is required for tryptophan mutant blue fluorescence. J Biol Chem 278:6275–6281
Sambrook J, Fritsch EF, Maniatis RS (1989) Molecular cloning: a Laboratory Manual, 2nd edn edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Sinlapadech T, Stout J, Ruegger MO, Deak M, Chapple C (2007) The hyper-fluorescent trichome phenotype of the brt1 mutant of Arabidopsis is the result of a defect in a sinapic acid: UDPG glucosyltransferase. Plant J 49:655–668
Ruegger M, Chapple C (2001) Mutations that reduces sinapoylmalate accumulation in Arabidopsis thaliana define loci with diverse roles in phenylpropanoid metabolism. Genetics 159:1741–1749
Tamagnone L, Merida A, Parr A, Mackay S, Culianez-Macia FA, Roberts K, Martin C (1998) The AmMYB308 and AmMYB330 transcription factors from Antirrhinum regulate phenylpropanoid and lignin biosynthesis in transgenic tobacco. Plant Cell 10:135–154
Weisshaar B, Jenkins GI (1998) Phenylpropanoid biosynthesis and its regulation. Curr Opin Plant Biol 1:251–257
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Zhu Q, Ordiz MI, Dabi T, Beachy RN, Lamb C (2002) Rice TATA binding protein interacts functionally with transcription factor IIB and the RF2a bZIP transcriptional activator in an enhanced plant in vitro transcription system. Plant Cell 14:795–803
Acknowledgments
The authors thank the members of Bowles’ group for fruitful discussions and their support. Meg Stark is thanked for helping to obtain the GUS stained root sections. This work was funded by United Kingdom Biotechnology and Biological Science Research Council and the Garfield Weston Foundations.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2008_763_MOESM1_ESM.doc
[Comparison of changes in leaf soluble phenolic metabolite accumulation in the UGT72E gene cluster over-expressing plants (DOC 28 kb)
Rights and permissions
About this article
Cite this article
Lanot, A., Hodge, D., Lim, EK. et al. Redirection of flux through the phenylpropanoid pathway by increased glucosylation of soluble intermediates. Planta 228, 609–616 (2008). https://doi.org/10.1007/s00425-008-0763-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-008-0763-8