Abstract
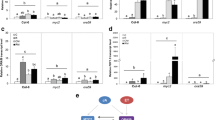
Glucosinolates are plant secondary compounds involved in direct chemical defence by cruciferous plants against herbivores. The glucosinolate profile can be affected by abiotic and biotic environmental stimuli. We studied changes in glucosinolate patterns in leaves of non-transgenic oilseed rape (Brassica napus ssp. oleifera) under elevated atmospheric CO2 or ozone (O3) concentrations and compared them with those from transgenic for herbivore-resistance (Bacillus thuringiensis Cry1Ac endotoxin), to assess herbivory dynamics. Both elevated CO2 and O3 levels decreased indolic glucosinolate concentrations in transgenic and non-transgenic lines, whereas O3 specifically increased the concentration of an aromatic glucosinolate, 2-phenylethylglucosinolate. The herbivore-inducible indolic glucosinolate response was reduced in elevated O3 whereas elevated CO2 altered the induction dynamics of indolic and aliphatic glucosinolates. Herbivore-resistant Bt plants experienced minimal leaf damage after target herbivore Plutella xylostella feeding, but exhibited comparatively similar increase in glucosinolate concentrations after herbivory as non-transgenic plants, indicating that the endogenous glucosinolate defence was not severely compromised by transgenic modifications. The observed differences in constitutive and inducible glucosinolate concentrations of oilseed rape under elevated atmospheric CO2 and O3 might have implications for plant–herbivore interactions in Brassica crop-ecosystems in future climate scenarios.




Similar content being viewed by others
Abbreviations
- Bt:
-
Bacillus thuringiensis
- GM:
-
Genetically modified
- GS:
-
Glucosinolate
References
Agrawal AA, Conner JK, Johnson MT, Wallsgrove R (2002) Ecological genetics of an induced plant defense against herbivores: additive genetic variance and costs of phenotypic plasticity. Evolution 56:2206–2213
Bartlet E, Kiddle G, Williams I, Wallsgrove R (1999) Wound-induced increases in the glucosinolate content of oilseed rape and their effect on subsequent herbivory by a crucifer specialist. Entomol Exp Appl 91:163–167
Bezemer TM, Jones TH (1998) Plant-insect herbivore interactions in elevated atmospheric CO2: quantitative analyses and guild effects. Oikos 82:212–222
Bidart-Bouzat MG, Mithen R, Berenbaum MR (2005) Elevated CO2 influences herbivory-induced defense responses of Arabidopsis thaliana. Oecologia 145:415–424
Bodnaryk RP (1992) Effects of wounding on glucosinolates in the cotyledons of oilseed rape and mustard. Phytochemistry 31:2671–2677
Bones AM, Rossiter JT (1996) The myrosinase-glucosinolate system, its organization and biochemistry. Physiol Plant 97:194–208
Brader G, Tas E, Palva ET (2001) Jasmonate-dependent induction of indole glucosinolates in Arabidopsis by culture filtrates of the nonspecific pathogen Erwinia carotovora. Plant Physiol 126:849–860
Brader G, Mikkelsen MD, Halkier BA, Palva ET (2006) Altering glucosinolate profiles modulates disease resistance in plants. Plant J 46:758–767
Bryant JP, Chapin FS III, Klein DR (1983) Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 40:357–368
Buskov S, Serra B, Rosa E, Sorensen H, Sorensen JC (2002) Effects of intact glucosinolates and products produced from glucosinolates in myrosinase-catalyzed hydrolysis on the potato cyst nematode (Globodera rostochiensis cv. Woll). J Agric Food Chem 50:690–695
Chen FJ, Wu G, Ge F, Parajulee MN, Shrestha RB (2005) Effects of elevated CO2 and transgenic Bt cotton on plant chemistry, performance, and feeding of an insect herbivore, the cotton bollworm. Entomol Exp Appl 115:341–350
Cipollini D, Enright S, Traw MB, Bergelson J (2004) Salicylic acid inhibits jasmonic acid-induced resistance of Arabidopsis thaliana to Spodoptera exigua. Mol Ecol 13:1643–1653
Cole R (1996) Abiotic induction of changes to glucosinolate profiles in Brassica species and increased resistance to the specialist aphid Brevicoryne brassicae. Entomol Exp Appl 80:228–230
Coviella CE, Morgan DJW, Trumble JT (2000) Interactions of elevated CO2 and nitrogen fertilization: effects on production of Bacillus thuringiensis toxins in transgenic plants. Environ Entomol 29:781–787
Coviella CE, Stipanovic RD, Trumble JT (2002) Plant allocation to defensive compounds: interactions between elevated CO2 and nitrogen in transgenic cotton plants. J Exp Bot 53:323–331
Dean JM, De Moraes CM (2006) Effects of genetic modification on herbivore-induced volatiles from maize. J Chem Ecol 32:713–724
Docherty M, Hurst DK, Holopainen JK, Whittaker JB, Lea PJ, Watt AD (1996) Carbon dioxide-induced changes in beech foliage cause female beech weevil larvae to feed in a compensatory manner. Global Change Biol 2:335–341
Doughty KJ, Kiddle GA, Pye BJ, Wallsgrove RM, Pickett JA (1995) Selective induction of glucosinolates in oilseed rape leaves by methyl jasmonate. Phytochemistry 38:347–350
Dudareva N, Negre F, Nagegowda DA, Orlova I (2006) Plant volatiles: recent advances and future perspectives. Crit Rev Plant Sci 25:417–440
Fahey JW, Zalcmann AT, Talalay P (2001) The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 56:5–51
Fiscus EL, Booker FL, Burkey KO (2005) Crop responses to ozone: Uptake, modes of action, carbon assimilation and partitioning. Plant Cell Environ 28:997–1011
Giamoustaris A, Mithen R (1995) The effect of modifying the glucosinolate content of leaves of oilseed rape (Brassica napus ssp. oleifera) on its interaction with specialist and generalist pests. Ann Appl Biol 126:347–363
Gielen B, Vandermeiren K, Horemans N, D’Haese D, Serneels R, Valcke R (2006) Chlorophyll a fluorescence imaging of ozone-stressed Brassica napus L. plants differing in glucosinolate concentrations. Plant Biol 8:698–705
Gouinguené SPD, Städler E (2006) Comparison of the egg-laying behaviour and electrophysiological responses of Delia radicum and Delia floralis to cabbage leaf compounds. Physiol Entomol 31:382–389
Grubb CD, Abel S (2006) Glucosinolate metabolism and its control. Trends Plant Sci 11:89–100
Halfhill MD, Richards HA, Mabon SA, Stewart CN Jr (2001) Expression of GFP and Bt transgenes in Brassica napus and hybridization with Brassica rapa. Theor Appl Genet 103:659–667
Halfhill MD, Sutherland JP, Moon HS, Poppy GM, Warwick SI, Weissinger AK, Rufty TW, Raymer PL, Stewart CN Jr (2005) Growth, productivity, and competitiveness of introgressed weedy Brassica rapa hybrids selected for the presence of Bt cry1Ac and gfp transgenes. Mol Ecol 14:3177–3189
Halkier BA, Gershenzon J (2006) Biology and biochemistry of glucosinolates. Annu Rev Plant Biol 57:303–333
Herms DA, Mattson WJ (1992) The dilemma of plants: To grow or defend. Q Rev Biol 67:283–335
IPCC (2007) Climate change 2007: the physical science basis. In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
James C (2006) Executive summary of global status of commercialized biotech/GM crops: 2006. ISAAA Brief No. 35, ISAAA, Ithaca, NY, http://www.isaaa.org
Jondrup PM, Barnes JD, Port GR (2002) The effect of ozone fumigation and different Brassica rapa lines on the feeding behavior of Pieris brassicae larvae. Entomol Exp Appl 104:143–151
Kangasjärvi J, Jaspers P, Kollist H (2005) Signalling and cell death in ozone-exposed plants. Plant Cell Environ 28:1021–1036
Karowe DN, Seimens DH, Mitchell-Olds T (1997) Species-specific response of glucosinolate content to elevated atmospheric CO2. J Chem Ecol 23:2569–2582
Kiddle GA, Doughty KJ, Wallsgrove RM (1994) Salicylic acid-induced accumulation of glucosinolates in oilseed rape (Brassica napus L.) leaves. J Exp Bot 45:1343–1346
Kim JH, Jander G (2007) Myzus persicae (green peach aphid) feeding on Arabidopsis induces the formation of a deterrent indole glucosinolate. Plant J 49:1008–1019
Koritsas VM, Lewis JA, Fenwick GR (1991) Glucosinolate responses of oilseed rape, mustard and kale to mechanical wounding and infestation by cabbage stem flea beetle (Psylliodes-Chrysocephala). Ann Appl Biol 118:209–221
Li Q, Eigenbrode SD, Stringam GR, Thiagarajah MR (2000) Feeding and growth of Plutella xylostella and Spodoptera eridania on Brassica juncea with varying glucosinolate concentrations and myrosinase activities. J Chem Ecol 26:2401–2420
Linsinger T, Kristiansen N, Beloufa N, Schimmel H, Pauwels J (2001) The certification of the total glucosinolate and sulphur contents of three rapeseed (colza) materials BCR-190R, -366R and -367R. BCR information series report EUR 19764 European Commission Luxembourg
Loivamäki M, Holopainen JK, Nerg AM (2004) Chemical changes induced by methyl jasmonate in oilseed rape grown in the laboratory and in the field. J Agric Food Chem 52:7607–7613
Long SP, Ainsworth EA, Leakey ADB, Nösberger J, Ort DR (2006) Food for thought: lower-than-expected crop yield stimulation with rising CO2 concentrations. Science 312:1918–1921
Louda S, Mole S (1991) Glucosinolates: chemistry and ecology. In: Rosenthal GA, Berenbaum MR (eds) Herbivores: their interactions with secondary plant metabolites, the chemical participants, vol I. Academic, New York, pp 123–163
Mason P, Braun L, Warwick SI, Zhu B, Stewart CN Jr (2003) Transgenic Bt-producing Brassica napus: Plutella xylostella selection pressure and fitness of weedy relatives. Environ Biosafety Res 2:263–276
Mewis I, Appel HM, Hom A, Raina R, Schultz JC (2005) Major signaling pathways modulate Arabidopsis glucosinolate accumulation and response to both phloem-feeding and chewing insects. Plant Physiol 138:1149–1162
Mikkelsen MD, Petersen BL, Glawischnig E, Jensen AB, Andreasson E, Halkier BA (2003) Modulation of CYP79 genes and glucosinolate profiles in Arabidopsis by defense signaling pathways. Plant Physiol 131:298–308
Penuelas J, Estiarte M (1998) Can elevated CO2 affect secondary metabolism and ecosystem function? Trends Ecol Evol 13:20–24
Poorter H, VanBerkel Y, Baxter R, DenHertog J, Dijkstra P, Gifford RM, Griffin KL, Roumet C, Roy J, Wong SC (1997) The effect of elevated CO2 on the chemical composition and construction costs of leaves of 27 C-3 species. Plant Cell Environ 20:472–482
Poppy GM, Wilkinson MJ (2005) Gene flow from GM plants. Blackwell, Kundli, India
Potting RPJ, Poppy GM, Schuler TH (1999) The role of volatiles from cruciferous plants and pre-flight experience in the foraging behaviour of the specialist parasitoid Cotesia plutellae. Entomol Exp Appl 93:87–95
Ratzka A, Vogel H, Kliebenstein DJ, Mitchell-Olds T, Kroymann J (2002) Disarming the mustard oil bomb. Proc Natl Acad Sci USA 99:11223–11228
Reddy GVP, Tossavainen P, Nerg AM, Holopainen JK (2004) Elevated atmospheric CO2 affects the chemical quality of Brassica plants and the growth rate of the specialist, Plutella xylostella, but not the generalist, Spodoptera littoralis. J Agric Food Chem 52:4185–4191
Sasaki-Sekimoto Y, Taki N, Obayashi T, Aono M, Matsumoto F, Sakurai N, Suzuki H, Hirai MY, Noji M, Saito K, Masuda T, Takamiya K, Shibata D, Ohta H (2005) Coordinated activation of metabolic pathways for antioxidants and defence compounds by jasmonates and their roles in stress tolerance in Arabidopsis. Plant J 44:653–668
Schonhof I, Kläring H-P, Krumbein A, Schreiner M (2007) Interaction between atmospheric CO2 and glucosinolates in broccoli. J Chem Ecol 33:105–114
Schuler TH, Poppy GM, Kerry BR, Denholm I (1999a) Potential side effects of insect-resistant transgenic plants on arthropod natural enemies. Trends Biotechnol 17:210–216
Schuler TH, Potting RPJ, Denholm I, Poppy GM (1999b) Parasitoid behaviour and Bt plants. Nature 400:825–826
Siemens DH, Mitchell-Olds T (1996) Glucosinolates and herbivory by specialists (Coleoptera: Chrysomelidae, Lepidoptera: Plutellidae): consequences of concentration and induced resistance. Environ Entomol 25:1344–1353
Siemens DH, Mitchell-Olds T (1998) Evolution of pest-induced defenses in Brassica plants: Tests of theory. Ecology 79:632–646
Stewart CN Jr (2004) Genetically modified planet: environmental impacts of genetically engineered plants. Oxford University Press, New York
Talekar NS, Shelton AM (1993) Biology, ecology, and management of the diamondback moth. Annu Rev Entomol 38:275–301
Tolrà RP, Alonso R, Poschenrieder C, Barceló D, Barceló J (2000) Determination of glucosinolates in rapeseed and Thlaspi caerulescens plants by liquid chromatography-atmospheric pressure chemical ionization mass spectrometry. J Chromatogr A 889:75–81
Traw MB, Kim J, Enright S, Cipollini DF, Bergelson J (2003) Negative cross-talk between salicylate- and jasmonate-mediated pathways in the Wassilewskija ecotype of Arabidopsis thaliana. Mol Ecol 12:1125–1135
Van Emden HF (1969) Plant resistance to Myzus persicae induced by a plant regulator and measured by aphid relative growth rate. Entomol Exp Appl 12:125–131
Vierheilig H, Bennett R, Kiddle G, Kaldorf M, Ludwig-Müller J (2000) Differences in glucosinolate patterns and arbuscular mycorrhizal status of glucosinolate-containing plant species. New Phytol 146:343–352
Acknowledgments
We thank Virpi Tiihonen (University of Kuopio, Finland) for glucosinolate sample preparations and HPLC work, and Timo Oksanen (University of Kuopio, Finland) for programming and maintaining the growth chambers. We are also grateful to Maaria Loivamäki (currently at Research Center Karlsruhe, Germany) and Delia M. Pinto (University of Kuopio, Finland) for their help in sampling during the individual experiments and Marja-Leena Hännilä (University of Kuopio, Finland) for her statistical advice. We also acknowledge Ewen MacDonald for revision of the language. This work was supported by the Academy of Finland (ESGEMO programme, decision no. 105209) (S.J.H., A.N., J.K.H. and A.-M.N.) and enabled by USDA BRAG grants to C.N.S.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Himanen, S.J., Nissinen, A., Auriola, S. et al. Constitutive and herbivore-inducible glucosinolate concentrations in oilseed rape (Brassica napus) leaves are not affected by Bt Cry1Ac insertion but change under elevated atmospheric CO2 and O3 . Planta 227, 427–437 (2008). https://doi.org/10.1007/s00425-007-0629-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-007-0629-5