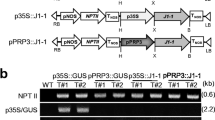
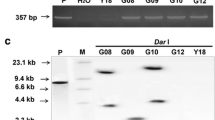
Abstract
Diseases of agricultural crops are caused by pathogens from several higher-order phylogenetic lineages including fungi, straminipila, eubacteria, and metazoa. These pathogens are commonly managed with pesticides due to the lack of broad-spectrum host resistance. Gastrodia anti-fungal protein (GAFP; gastrodianin) may provide a level of broad-spectrum resistance due to its documented anti-fungal activity in vitro and structural similarity to insecticidal lectins. We transformed tobacco (Nicotiana tabacum cv. Wisconsin 38) with GAFP-1 and challenged transformants with agriculturally important plant pathogens from several higher-order lineages including Rhizoctonia solani (fungus), Phytophthora nicotianae (straminipile), Ralstonia solanacearum (eubacterium), and Meloidogyne incognita (metazoan). Quantitative real-time PCR and western blotting analysis indicated that GAFP-1 was transcribed and translated in transgenic lines. When challenged by R. solani and P. nicotianae, GAFP-1 expressing lines had reduced symptom development and improved plant vigor compared to non-transformed and empty vector control lines. These lines also exhibited reduced root galling when challenged by M. incognita. Against R. solanacearum expression of GAFP-1 neither conferred resistance, nor exacerbated disease development. These results indicate that heterologous expression of GAFP-1 can confer enhanced resistance to a diverse set of plant pathogens and may be a good candidate gene for the development of transgenic, root-disease-resistant crops.




Similar content being viewed by others
Notes
Due to debate surrounding the categorization of taxa above the phylum level, we will refer to such taxa as higher-order phylogenetic lineages instead of using the kingdom designation.
References
Agrios GN (1997) Plant pathology. Academic, San Diego, p 635
Anonymous (2000) Protection of stratospheric ozone: incorporation of clean air act amendments for reductions in class I, group VI controlled substances. Fed Regist 65:70795–70804
Bajaj M, Soni G, Singh CK (2001) Interaction of pea (Pisum sativum L.) lectins with rhizobial strains. Microbiol Res 156:71–74
Beckman TG, Nyczepir AP (2004) Peach tree short life. In: Horton D, Johnson D (eds) Southeastern peach growers handbook. University of Georgia, College of Agricultural and Environmental Sciences. Cooperative Extension Service, Athens, pp 199–205
Broekaert WF, Van Parijs J, Leyns F, Joos H, Peumans WJ (1989) A chitin-binding lectin from stinging nettle rhizomes with antifungal properties. Science 245:1100–1102
Burow DM, Chlan CA, Sen P, LIsca A, Murai N (1990) High-frequency generation of transgenic tobacco plants after modified cocultivation with Agrobacterium tumefaciens. Plant Mol Biol Rep 8:124–139
Burrows PR, Barker AD, Newell CA, Hamilton WDO (1998) Plant-derived enzyme inhibitors and lectins for resistance against plant-parasitic nematodes in transgenic crops. Pestic Sci 52:176–183
Chen H, Nelson RS, Sherwood JL (1994) Enhanced recovery of transformants of Agrobacterium tumefaciens after freeze-thaw transformation and drug selection. Biotechniques 16:664–668
Chomczynski P (1993) A reagent for the single-step simultaneous isolation of RNA, DNA and proteins from cell and tissue samples. Biotechniques 15:532–537
Chrispeels M, Raikhel N (1991) Lectins, lectin genes, and their role in plant defense. Plant Cell 3:1–9
Csinos A, Stephenson M (1999) Evaluation of fungicides and tobacco cultivar resistance to Rhizoctonia solani incited target spot, damping off and sore shin. Crop Prot 18:373–377
Diaz C, Logman T, Stam H, Kijne J (1995) Sugar-binding activity of pea lectin expressed in white clover hairy roots. Plant Physiol 109:1167–1177
Does MP, Houterman PM, Dekker HL, Cornelissen BJC (1999) Processing, targeting, and antifungal activity of stinging nettle agglutinin in transgenic tobacco. Plant Physiol 120:421–431
Down R, Ford L, Woodhouse S, Davison G, Majerus M, Gatehouse J, Gatehouse A (2003) Tritrophic interactions between transgenic potato expressing snowdrop lectin (GNA), an aphid pest (peach-potato aphid; Myzus persicae (Sulz.)) and a beneficial predator (2-spot ladybird; Adalia bipunctata L.). Transgenic Res 12:229–241
Eijsden R, Diaz C, Pater B, Kijne J (1995) Sugar-binding activity of pea (Pisum sativum) lectin is essential for heterologous infection of transgenic white clover hairy roots by Rhizobium leguminosarum biovar viciae. Plant Mol Biol Rep 29:431–439
Etzler ME (1986) Distribution and function of plant lectins. In: Liener IE, Sharon N, Goldstein IJ (eds) The lectins. Academic, San Diego, pp 371–435
Fitches E, Woodhouse SD, Edwards JP, Gatehouse JA (2001) In vitro and in vivo binding of snowdrop (Galanthus nivalis agglutinin; GNA) and jackbean (Canavalia ensiformis; Con A) lectins with tomato moth (Lacanobia oleracea) larvae; mechanisms of insecticidal action. J Insect Physiol 47:777–787
Gallagher S, Winston SE, Fuller SA, Hurrell JR (1997) Immunoblotting and immunodetection. In: Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (eds) Current protocols in molecular biology. Wiley, Hoboken, pp 10.8.1–10.8.21
Gatehouse A, Davison G, Stewart J, Gatehouse L, Kumar A, Geoghegan I, Birch A, Gatehouse J (1999) Concanavalin A inhibits development of tomato moth (Lacanobia olecea) and peach-potato aphid (Myzus persicae) when expressed in transgenic potato plants. Mol Breed 5:153–165
Hu Z, Yang Z, Wang J (1988) Isolation and partial characterization of an antifungal protein from Gastrodia elata corm. Acta Bot Yunnan 10:373–380
Hussey RS, Barker KR (1973) A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Dis Rep 57:1025–1028
Hussey RS, Janssen GJW (2002) Root-knot nematodes: Meloidogyne species. In: Starr JL, Cook R, Bridge J (eds) Plant resistance to parasitic nematodes. CABI Publishing, New York, pp 43–70
Lin H-J, McClure MA (1996) Surface coat of Meloidogyne incognita. J Nematol 28:216–224
Lui W, Yang N, Ding J, Huang R-H, Hu Z, Wang D-C (2005) Structural mechanism governing the quaternary organization of monocot mannose-binding lectin revealed by the novel monomeric structure of an orchid lectin. J Biol Chem 280:14865–14876
Mazzola M (1998) Elucidation of the microbial complex having a causal role in the development of apple replant disease in Washington. Phytopathology 88:930–938
McClure MA, Stynes BA (1988) Lectin binding sites on the amphidial exudates of Meloidogyne. J Nematol 20:321–326
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:2002–2007
Rhijn P, Goldberg R, Hirsch A (1998) Lotus corniculatus nodulation specificity is changed by the presence of a soybean lectin gene. Plant Cell 10:1233–1249
Rhijn P, Fujishige N, Lim P, Hirsch A (2001) Sugar-binding activity of pea lectin enhances heterologous infection of transgenic alfalfa plants by Rhizobium leguminosarum biovar viciae. Plant Physiol 126:133–144
Richmond JY, McKinney RW (1993) Biosafety in Microbiological and Biomedical Laboratories. US Department of Health and Human Services, CDC/NIH, US Government Printing Office, Washington
Rioger DJ, Jeffers SN (1991) Evaluation of Trichoderma spp. for biological control of phytophthora crown and root rot of apple seedlings. Phytopathology 81:910–917
Ripoll C, Favery B, Lecomte P, Van-Damme E, Peumans W, Abad P, Jouanin L (2003) Evaluation of the ability of lectin from snowdrop (Galanthus nivalis) to protect plants against root-knot nematodes. Plant Sci 164:517–523
Robertson A, Wechter W, Denny T, Fortnum B, Kluepfel D (2004) Relationship between avirulence gene (avrA) diversity in Ralstonia solanacearum and bacterial wilt incidence. Mol Plant Microbe Interact 17:1376–1384
Rumberger A, Yao S, Merwin IA, Nelson EB, Thies JE (2004) Rootstock genotype and orchard replant position rather than soil fumigation or compost amendment determine tree growth and rhizosphere bacterial community composition in an apple replant soil. Plant Soil 264:247–260
Setamou M, Bernal J, Legaspi J, Mirkov T, Legaspi B (2002) Evaluation of lectin-expressing transgenic sugarcane against stalkborers (Lepidoptera: Pyralidae): effects on life history parameters. J Econ Entomol 95:469–477
Sun X, Wu A, Tang K (2002) Transgenic rice lines enhanced resistance to the small brown planthopper. Crop Prot 21:511–514
Van Parijs J, Broekaert W, Goldstein I, Peumans W (1991) Hevein: an antifungal protein from rubber-tree (Hevea brasiliensis) latex. Planta 183:258–264
Wang XC, Bauw G, Van Damme EJM, Peumans WJ, Chen ZL, Van Montagu M, Angenon G, Dillen W (2001) Gastrodianin-like mannose-binding proteins: a novel class of plant proteins with antifungal properties. Plant J 25:651–661
Wang P, Wang Y, Sa Q, Li W, Sun Y (2003) The site-directed mutagenesis of gastrodia anti-fungal protein mannose-binding sites and its expression in Escherichia coli. Protein Pept Lett 10:599–606
Wang Y, Chen L, Wang D, Huang Q, Yao Z, Liu F, Wei X, Li R, Zhang Z, Sun Y (2004) Over-expression of Gastrodia anti-fungal protein enhances Verticillium wilt resistance in coloured cotton. Plant Breed 123:454–459
Wu A, Sun X, Pang Y, Tang K (2002) Homozygous transgenic rice lines expressing GNA with enhanced resistance to the rice sap-sucking pest Laodelphax striatellus. Plant Breed 121:93–95
Xu Q, Liu Y, Wang X, Gu H, Chen Z (1998) Purification and characterization of a novel anti-fungal protein from Gastrodia elata. Plant Physiol Biochem 36:899–905
Zentmyer G (1980) Phytophthora cinnamomi and the diseases it causes. The American Phytopathological Society, St Paul
Zuckerman BM (1983) Hypotheses and possibilities of intervention in nematode chemo-responses. J Nematol 15:173–182
Acknowledgments
This study was funded in part by the South Carolina Peach Council, USDA-CSREES SR-IPM grant no. 2005-34103-15588, USDA-CSREES special grant no. 2004-34126-14388, USDA NRI grant no. 2002-35319-12527, ISA Research Trust, and the John Z. Duling Research Grant Program. We thank Jessica Eisenmann for conducting the initial Phytophthora disease screening experiments and the RT-PCR gene expression experiments. We also thank Phaedra Kennedy for carrying out the transformation experiments.
Author information
Authors and Affiliations
Corresponding author
Additional information
Technical Contribution No. 5172 of the Clemson University Experiment Station. This material is based upon work supported by the CSREES/USDA, under project number SC-1000642.
Rights and permissions
About this article
Cite this article
Cox, K.D., Layne, D.R., Scorza, R. et al. Gastrodia anti-fungal protein from the orchid Gastrodia elata confers disease resistance to root pathogens in transgenic tobacco. Planta 224, 1373–1383 (2006). https://doi.org/10.1007/s00425-006-0322-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-006-0322-0