Abstract
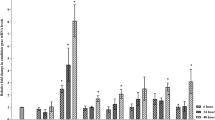
Erwinia amylovora is a member of the harpin proteins that induces pathogen resistance and hypersensitive cell death in plants. To obtain tobacco plants displaying a hypersensitive response, the hrpN gene from Erwinia amylovora was cloned into vector pMJC-GB under the control of the rice cytochrome promoter and transfected into tobacco. Southern hybridization with a hrpN probe revealed that the gene was present in one copy in the transgenic plants. In addition, hrpN transcripts could be detected in transgenic plants but not in wild-type tobacco. The wild type gave 75 products in RAPD analysis with 12 primers while the transgenic plants gave 73, suggesting that hrpN gene had been integrated into the transgenic plant genomic DNA. The distribution of cell cycle phases in the wild type and transgenic plants was G0-G1: 71.25%, G2-M: 20.41%, S: 8.33%, while in transgenic plant was G0-G1: 54.95%, G2-M: 43.82%, S: 10.23%. The sizes of stomata and guard cells on transgenic leaves were similar to those of the wild type, but the epidermal cells were clearly smaller. The transgenic plants showed accelerated growth and development as well as enhanced resistance to Botrytis cinerea.







Similar content being viewed by others
Abbreviations
- HR:
-
Hypersensitive response
- PI:
-
Propidium iodide
- SEM:
-
Scanning electron microscopy
- RAPD:
-
Randomly amplified polymorphic DNA
References
Alfano JR, Collmer A (1997) The type β (hrp) secretion pathway of plant pathogenic bacteria: trafficking harpins, Avr proteins, and death. J Bacteriol 179:5655–5662
Bogdanove AJ, Wei ZM, Zhao LP, Beer SV (1996) Erwinia amylovora secretes harpin via a type β pathway and contains a homolog of yopN of Yersinia spp. J Bacteriol 178:1720–1730
Dangl JL, Dietrich RA, Richberg MH (1996) Death don’t have no mercy: cell death programs in plant-microbe interactions. Plant Cell 8:1793–1807
Desikan R, Hancock JT, Ichimura K, Shinozaki K, Neill SJ (2001) Harpin induces activation of the Arabidopsis mitogen activated protein kinases AtMPK4 and AtMPK6. Plant Physiol 126:1579–1587
Frederick RD, Ahmad M, Majerczak DR, Arroyo-Rodriguez AS, Manulis S, Coplin DL (2001) Genetic organization of the Pantoea stewartii subsp.stewartii hrp gene cluter and sequence anylysis of the hrpA, hrpC, hrpN, and wtsE Operons. Mol Plant Microbe In 14:1213–1222
Ger MJ, Chen CH, Hwang SY, Huang HE, Podile AR, Dayakar BV, Feng TY (2002) Constitutive expression of hrp gene in transgenic tobacco plant enhances resistance against virulent bacterial pathogens by induction of hypersensitive response. Mol Plant Microbe In 15:764–773
Govrin EM, Levine A (2000) The hypersensitive response facilitates plant infection by the necrotrophic pathogen Botrytis cinerea. Curr Biol 10:751–757
He SY, Huang HC, Collmer A (1993) Pseudomonas syringae pv. syringae harpinpss: a protein that is secreted via the hrp pathway and elicits the hypersensitive response in plants. Cell 73:1255–1266
Hoeberichts FA, Have AT, Woltering EJ (2003) A tomato metacaspase gene is upregulated during programmed cell death in Botrytis cinerea-infected leaves. Planta 217:517–522
Horsch RB, Fry JE, Hofmann NL, Eicholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Huang HE, Ger MJ, Yip MK, Chen CY, Pandey AK, Feng TY (2004) A hypersensitive response was induced by virulent bacteria in transgenic tobacco plants overexpressing a plant ferredoxin-like protein (PFLP). Physiol Mol Plant P 64:103–110
Keller H, Pamboukdjian N, Ponchet M, Poupet A, Delon R, Verrier JL, Roby D, Ricci P (1999) Pathogen-Induced elicitin production in transgenic tobacco generates a hypersensitive response and nonspecific disease resistance. Plant Cell 11:223–235
Keshava C, Keshava N, Zhou G, Whong WZ, Ong TM (1999) Genomic instability in silica- and cadmium chloride-transformed BALB/c-3T3 and tumor cell lines by random amplified polymorphic DNA analysis. Mutat Res 425:117–123
Krause M, Durner J (2004) Harpin inactivates mitochondria in Arabidopsis suspension cells. Mol Plant Microbe In 17:131–139
Krizhanovsky V, Golenser E, Ben-Arie N (2004) Genotype identification of Math1/LacZ knockout mice based on real-time PCR with SYBR Green I dye. J Neurosci Meth 136:187–192
Lehtimäki S, Rantakari A, Routtu J, Tuikkala A, Li J, Virtaharju O, Palva ET, Romantschuk M, Saarilahti HT (2003) Charaterization of the hrp pathogenicity cluster of Erwinia carotovora subsp. carotovora: high basal level expression in a mutant is associated with reduced virulence. Mol Gen Genomics 270:263–272
Lorbiecke R, Sauter M (1998) Induction of cell growth and cell division in the intercalary meristem of submerged deepwater rice (Oryza sativa L.). Planta 204:140–145
Mauseth JD (1988) Plant anatomy. The Benjamin/Cummings publishing company, California, p: 44
Mor H, Manulis S, Zuck M, Nizan R, Coplin DL, Barash L (2001) Genetic organization of hrp gene cluster and dspAE/BF operon in Erwinia herbicola pv. gypsophilae. Mol Plant Microbe In 14:431–436
Oakenfull EA, Riou-Khamlichi C, Murray JA (2002) Plant D-type cyclins and the control of G1 progression. Philos Trans R Soc Lond B Biol Sci 357:749–760
Peng JL, Bao ZL, Ren HY, Wang JS, Dong HS (2004) Expression of Harpinxoo in transgenic tobacco induces pathogen defense in the absence of hypersensitive cell death. J Bacteriol 94:1048–1055
Petersen K, Leah R, Knudsen S, Mill VC (2002) Matrix attachment regions (MARs) enhance transformation frequencies and reduce variance of transgene expression in barley. Plant Mol Biol 49:45–58
Prins TW, Tudzynski P, von Tiedemann A, Tudzynski B, Have A, Hansen ME, Tenberge K, van Kan JAL (eds) (2000) Infection Strategies of Botrytis cinerea and related necrotrophic pathogens. Kluwer, Netherlands
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory press, Cold Spring Harbor
Taguchi F, Tanaka R, Kinoshita S, Ichinose Y, Imura Y, Andi S, Toyoda K, Shiraishi T, Yamada T (2001) Harpinpsta from Pseudomonas syringae pv. tabaci is defective and deficient in its expression and HR-inducing activity. J Gen Plant Pathol 67:116–123
Tripathy S, Kleppinger-Sparace K, Dixon RA, Chapman KD (2003) N-Acylethanolamine signaling in tobacco is mediated by a membrane-associated, high-affinity binding protein. Plant Physiol 131:1781–1791
Wei ZM, Laby RJ, Zumoff CH, Bauer DW, He SY, Collmer A, Beer SV (1992) Harpin, elicitor of the hypersensitive response produced by the plant pathogen Erwinia amylovora. Science 257:85–88
Williams JGK, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 18:6531–6535
Author information
Authors and Affiliations
Corresponding author
Additional information
Young-Sun Jang and Soo-In Sohn are contributed equally to this work
Rights and permissions
About this article
Cite this article
Jang, YS., Sohn, SI. & Wang, MH. The hrpN gene of Erwinia amylovora stimulates tobacco growth and enhances resistance to Botrytis cinerea . Planta 223, 449–456 (2006). https://doi.org/10.1007/s00425-005-0100-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-005-0100-4