Abstract
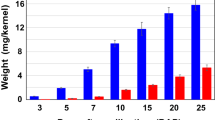
Endosperm was isolated from Arabica Caturra coffee beans 11, 15, 21, 26, 31 and 37 weeks after flowering, and the chemical composition and relative solubility of its component polysaccharides determined at each growth stage. Chemical analysis of the total mannan content of the cell wall material was done after solubilisation of galactomannans by alkaline extraction of the cell wall material followed by enzymatic digestion of the alkali-insoluble residue with a mixture of endo-mannanse and endo-glucanase. Eleven weeks after flowering, galactomannans accounted for approximately 10% of the polysaccharides but were highly substituted, with galactose/mannose ratios between 1:2 and 1:7. As the bean matured, galactomannan became the predominant polysaccharide, until 31 weeks after flowering it accounted for approximately 50% of the polysaccharides. However, it was less substituted, possessing galactose/mannose ratios between 1:7 and 1:40. Early in bean growth, up to 50% of the cell wall polysaccharides were extractable but as the galactomannan content of the bean increased there was a reduction in the extractability of all polysaccharides. The decrease in the galactose/mannose ratio of the galactomannans commenced between 21 and 26 weeks after flowering and was in synchrony with a rise in the concentration of free galactose in the beans. The results indicated that the degree of substitution of the galactomannans in coffee beans is developmentally regulated and may result, in part, from the modification of a primary synthetic product by the action of an α-galactosidase.







Similar content being viewed by others
Abbreviations
- AIR:
-
alcohol-insoluble residue
- CWM:
-
cell wall material
- Gal/Man:
-
galactose/mannose ratio
- PAW:
-
phenol:acetic acid:water
- WAF:
-
weeks after flowering
References
Albersheim P, Nevins DJ, English PD, Karr A (1967) A method for the analysis of sugars in plant cell wall polysaccharides by gas-liquid chromatography. Carbohydr Res 5:340–345
Blumenkrantz N, Asboe-Hansen G (1973) New method for quantitative determination of uronic acid. Anal Biochem 17:484–489
Bradbury AGW (2001) Chemistry Ⅰ. Non-volatile compounds 1A: carbohydrates. In: Clarke RJ, Vitzthum OG (eds) Coffee, recent developments. Blackwell Science, London, pp 3–6
Bradbury AGW, Halliday DJ (1990) Chemical structures of green coffee bean polysaccharides. J Agric Food Chem 38:389–392
Ciucanu I, Kerek F (1984) A simple and rapid method for the permethylation of carbohydrates. Carbohydr Res 131:209–217
Clifford MN (1985) Chemical and physical aspects of green coffee and coffee products. In: Clifford MN, Willson KC (eds) Coffee, botany, biochemistry and production of beans and beverage. Croom Helm, London, pp 305–374
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for the estimation of sugars and related substances. Anal Chem 28:350–356
Edwards M, Scott C, Gidley MJ, Reid JSG (1992) Control of mannose/galactose ratio during galactomannan formation in developing legume seeds. Planta 187:67–74
Fischer M, Reimann S, Trovato V, Redgwell RJ (2000) Structural aspects of polysaccharides from Arabica coffee. In: Proceedings of the 18th international conference on coffee science. ASIC, Paris, pp 91–94
Fischer M, Reimann S, Trovato V, Redgwell RJ (2001) Polysaccharides of green Arabica and Robusta coffee beans. Carbohydr Res 330:93–101
Hansen SA (1975) Thin-layer chromatographic method for identification of oligosaccharides in starch hydrolysates. J Chromatogr 105:388–390
Illy A, Viani, R (1995) Espresso coffee. The chemistry of quality. Academic Press, London, pp 5–7
Joersbo M, Marcussen J, Brunstedt J (2001) In vivo modification of the cell wall polysaccharide galactomannan of guar transformed with an α-galactosidase gene cloned from Senna. Mol Breed 7:211–219
Kim JB, Carpita NC (1992) Changes in esterification of the uronic acid groups of cell wall polysaccharides during elongation of maize coleoptiles. Plant Physiol 98:646–653
McCleary BV, Matheson NK (1983) Action patterns and substrate binding requirements of β-d-mannanase with mannosaccharides and mannan-type polysaccharides. Carbohydr Res 119:191–219
McCleary BV, Amado R, Wabel R, Neukom H (1981) Effect of galactose content on the solution and interaction properties of guar and carob galactomannan. Carbohydr Res. 92:269–285
Redgwell R, Curti D, Fischer M, Nicolas P, Fay L (2002) Coffee arabinogalactans: acidic polysaccharides covalently linked to proteins. Carbohydr Res 337:239–253
Rogers WJ, Michaux S, Bastin M, Bucheli P (1999) Changes to the content of sugars, sugar alcohols, myo-inositol, carboxylic acids and inorganic anions in developing grains from different varieties of Robusta (Coffea canephora) and Arabica (C. arabica) coffees. Plant Sci 149:115–123
Sweet DP, Shapiro RH, Albersheim P (1975) Quantitative analysis by various G.L.C. response-factor theories for partially methylated and partially ethylated alditol acetates. Carbohydr Res 40:217–225
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Redgwell, R.J., Curti, D., Rogers, J. et al. Changes to the galactose/mannose ratio in galactomannans during coffee bean (Coffea arabica L.) development: implications for in vivo modification of galactomannan synthesis. Planta 217, 316–326 (2003). https://doi.org/10.1007/s00425-003-1003-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-003-1003-x