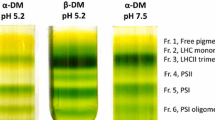
Abstract
The light stimulation of transglutaminase (TGase EC 2.3.2.13) activity was verified by incubating isolated chloroplasts of Helianthus tuberosus L. continuously or for alternate periods of light or dark (light/dark and dark/light). The first 10 min of incubation always represented the critical period. Light-harvesting complexes of photosystem II (LHCII) were more intensely labelled by 14C-polyamines under light and light/dark than under dark and dark/light conditions. Chloroplasts were fractionated into thylakoid- and stroma-enriched fractions in which multiple TGase forms and substrates were found. Antibodies against TGase recognised 58- and 24-kDa bands in thylakoids and a 150-kDa band in the stroma. The latter, and its 150-kDa fraction, catalysed the conjugation of 14C-polyamines to Rubisco. In both fractions (thylakoid-pre and stroma-pre) the analysis of polyamine glutamyl derivatives showed a significant light-affected conjugation of polyamines to endogenous proteins. Alternatively, entire chloroplasts were incubated and afterwards their sub-fractions were isolated (thylakoid-post and stroma-post). The PSII and LHCII complexes were more intensely immunodetected in thylakoid-post than in thylakoid-pre, especially under dark conditions. Conversely, the conjugation of polyamines to thylakoid proteins was clearly light-stimulated in thylakoid-post, and much less in thylakoid-pre. Stroma-pre proteins were poorly polyamine-conjugated and not light-affected; on the contrary, stroma-post proteins were much more polyamine-modified and strongly light-stimulated. Thus, the light-activated conjugation depends mainly on the presence of the thylakoid fraction during the assay. The protective effect on chloroplasts under photo-damage, stress or senescence conditions attributed in the literature to free polyamines is discussed with regard to the occurrence of polyamine conjugates catalysed by TGases.








Similar content being viewed by others
Abbreviations
- bis-, mono-PU:
-
bis-, mono-(γ-glutamyl) putrescine
- bis-, mono-SD:
-
bis-, mono-(γ-glutamyl) spermidine
- bis-, mono-SM:
-
bis-, mono-(γ-glutamyl) spermine
- PA:
-
polyamine
- PU:
-
putrescine
- RPG-TGase:
-
rat prostatic gland transglutaminase
- Rubisco:
-
ribulose 1,5-bisphosphate carboxylase/oxygenase
- SD:
-
spermidine
- SM:
-
spermine
- stroma-post:
-
stromal fraction isolated after the TGase assay of the entire chloroplast
- stroma-pre:
-
stromal fraction isolated before the TGase assay
- TGase:
-
transglutaminase
- thylakoid-post:
-
thylakoids isolated after TGase assay of the entire chloroplast
- thylakoid-pre:
-
thylakoids isolated before TGase assay
References
Andreadakis A, Kotzabasis K (1995) Changes in the biosynthesis and catabolism of polyamines in isolated plastids during the chloroplast photodevelopment. J Photochem Photobiol B 33:163–170
Bagni N, Torrigiani P (1992) Polyamines: a new class of growth substances. In: Karssen CM, van Loon LC, Vreugdenhil D (eds) Progress in plant growth regulation. Kluwer, Dordrecht, pp 264–275
Ballestar E, Abad C, Franco L (1996) Core histones are glutaminyl substrates for tissue transglutaminase. J Biol Chem 271:817–824
Beigbeder A, Vavadakis M, Navakoudis M, Kotzabasis K (1995) Influence of polyamine inhibitors on light-independent and light-dependent chlorophyll biosynthesis and on the photosynthetic rate. J. Photochem Photobiol B 28:235–242
Bernet E, Claparols I, Dondini L, Santos MA, Serafini-Fracassini D, Torné JM (1999) Changes in polyamine content, arginine and ornithine decarboxylases and transglutaminase activities during light/dark phases of initial differentiation in maize calluses and their chloroplasts. Plant Physiol Biochem 37:1–11
Besford RT, Richardson CM, Campos JL, Tiburcio AF (1993) Effect of polyamines on stabilization of molecular complexes in thylakoid membranes of osmotically stressed oat leaves. Planta 189:201–206
Borrell A, Culianez-Macià FA, Altabella T, Besford RT, Flores D, Tiburcio A (1995) Arginine decarboxylase is localized in chloroplasts. Plant Physiol 109:771–776
Cohen AS, Popovic RB, Zalik S (1979) Effects of polyamines on chlorophyll and protein content, photochemical activity, and chloroplast ultrastructure of barley leaf discs during senescence. Plant Physiol 64:717–780
Corley E, Wolosiuk RA, Hertig CM (1983) Regulation of the activation of chloroplast fructose-1,6-bisphosphatase: inhibition by spermidine and spermine. Biochem Biophys Res Commun 115:707–714
Dai YR, Galston AW (1981) Simultaneous phytochrome-controlled promotion and inhibition of arginine decarboxylase activity in buds and epicotyls of etiolated peas. Plant Physiol 67:266–269
Dainese P, Santini C, Ghiretti-Magaldi A, Marquardt J, Tidu V, Mauro S, Bergantino E, Bassi R (1992) The organization of pigment proteins within photosystem II. In: Murata N (ed) Research in photosynthesis, Kluwer, Dordrecht, pp 13–20
Del Duca S, Serafini Fracassini D (1993) Bound polyamines in plants. Curr Top Plant Physiol 1:83–102
Del Duca S, Favali A, Serafini-Fracassini D, Pedrazzini R (1993) Transglutaminase-like activity during greening and growth of Helianthus tuberosus explants. Protoplasma 174:1–9
Del Duca S, Tidu V, Bassi R, Serafini-Fracassini D, Esposito C (1994) Identification of transglutaminase activity and its substrates in isolated chloroplast of Helianthus tuberosus. Planta 193:283–289
Del Duca S, Beninati S, Serafini-Fracassini D (1995) Polyamines in chloroplasts: identification of their glutamyl- and acetyl- derivatives. Biochem J 305:233–237
Del Duca S, Dondini L, Della Mea M, Muñoz de Rueda P, Serafini-Fracassini D (2000) Factors affecting transglutaminase activity catalysing polyamine conjugation to endogenous substrates in the entire chloroplast. Plant Physiol Biochem 38:429–439
Desimone M, Henke A, Wagner E (1996) Oxidative stress induces partial degradation of the large subunit of ribulose-1,5-bis-phosphate carboxylase/oxygenase in isolated chloroplasts of barley. Plant Physiol 111:789–796
Di Paolo ML, Peruffo dal Belin A, Bassi R (1990) Immunological studies on chlorophyll a/b proteins and their location in chloroplast membrane domains. Planta 181:275–286
Dondini L (1998) Poliammine legate e transglutaminasi nelle piante. PhD thesis, Biologia e Fisiologia cellulare, University of Bologna, pp 1–252
Dondini L, Bonazzi S, Serafini-Fracassini D (2000) Recovery of growth capacity and of chloroplast transglutaminase activity induced by polyamines in a polyamine-deficient variant strain of Dunaliella salina. J Plant Physiol 157:473–480
Dondini L, Bonazzi S, Del Duca S, Bregoli AM, Serafini-Fracassini D (2001) Acclimation of chloroplast transglutaminase to high NaCl concentration in a polyamine-deficient variant strain of Dunaliella salina and in its wild type. J Plant Physiol 158:185–197
Dörnemann D, Navakoudis E, Kotzabasis K (1996) Changes in the polyamine content of plastidial membranes in light- and dark-grown wild type and pigment mutants of the unicellular green alga Scenedesmus obliquus and their possible role in chloroplast photodevelopment. J Photochem Photobiol B 36:293–299
Falcone P, Serafini-Fracassini D, Del Duca S (1993) Comparative studies of transglutaminase-like activity and substrates in different organs of Helianthus tuberosus. J Plant Physiol 142:265–273
Folk JE (1980) Transglutaminases. Annu Rev Biochem 49:517–531
Folk JE, Park MH, Chung SI, Schrode J, Lester EP, Cooper HL (1980) Polyamines as physiological substrates for transglutaminase. J Biol Chem 255: 3695–3700
Galston AW, Kaur-Sawhney R (1990) Polyamines in plant physiology Plant Physiol 94:406–410
Hames BD (1990) One-dimensional polyacrylamide gel electrophoresis. In: Hames BD, Rickwood D (eds) Gel electrophoresis of proteins. A practical approach. Oxford University Press, Oxford, pp 1–139
Heldt HW, Werdan K, Milovancev M, Geller G (1973) Alkalinization of the chloroplast stroma caused by light-dependent proton flux into the thylakoid space. Biochim Biophys Acta 314:224–241
Horton P, Ruban AV, Rees D, Pascal AA, Noctor G, Young AJ (1991) Control of the light-harvesting function of chloroplast membranes by aggregation of the LHCII chlorophyll–protein complex. FEBS Lett 292:1–4
Johnson CH, Knight MR, Kondo T, Masson P, Haley A, Trewavas A (1995) Circadian oscillations of cytosolic and chloroplastic free calcium in plants. Science 269:1863–1865
Kotzabasis K, Fotinou C, Roubelakis-Angelakis KA, Ghanotakis D (1993) Polyamines in the photosynthetic apparatus. Photosynth Res 38:83–88
Kotzabasis K, Strasser B, Navakoudis E, Senger H, Dörnemann D (1999) The regulatory role of polyamines in structure and functioning of the photosynthetic apparatus during photoadaptation. J Photochem Photobiol B 50:45–52
Kramer GF, Krizek DT, Mirecki RM (1992) Influence of photosynthetically active radiation and spectral quality on UV-B-induced polyamine accumulation in soybean. Phytochemistry 31:1119–1125
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–683
Lilley G, Skill J, Griffin M, Bonner P (1998) Detection of Ca2+-dependent transglutaminase activity in root and leaf tissue of monocotyledonous and dicotyledonous plants. Plant Physiol 117:1115–1123
Lorand L, Conrad SM (1984) Transglutaminases. Mol Cell Biochem 58:9–35
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Machold O, Meister A (1979) Resolution of the light-harvesting chlorophyll a/b-protein of Vicia faba chloroplasts into two different chlorophyll–protein complexes. Biochim Biophys Acta 546:472–480
Margosiak SA, Dharma A, Bruce-Carver MR, Gonzales AP, Louie D, Kuehn GD (1990) Identification of the large subunit of ribulose 1,5-bisphosphate carboxylase/oxygenase as a substrate for transglutaminase in Medicago sativa L. (Alfalfa). Plant Physiol 92:88–96
McCurry SD, Gee R, Tolbert NE (1982) Ribulose-1,5-bisphosphate carboxylase/oxygenase from spinach, tomato or tobacco leaves. Methods Enzymol 90:515–518
Peter GF, Thornberg PJ (1991) Biochemical composition and organization of higher plant photosystem II light-harvesting pigment-proteins. J Biol Chem 266:16745–16754
Popovic RB, Kyle DJ, Cohen AS, Zalik S (1979) Stabilization of thylakoid membranes by spermine during stress-induced senescence of barley leaf discs. Plant Physiol 64:721–726
Roulin S, Feller U (1998) Light-dependent degradation of stromal proteins in intact chloroplasts isolated from Pisum sativum L. leaves: requirement for divalent cations. Planta 205:297–304
Santini C, Tidu V, Tognon G, Ghiretti Magaldi A, Bassi R (1994) Three-dimensional structure of the higher-plant photosystem II reaction centre and evidence for its dimeric organization in vivo. Eur J Biochem 221:307–315
Schägger H, von Jagow G (1987) Tricine–sodium sulphate–polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem 166:368–379
Serafini-Fracassini D, Del Duca S (2002) Biochemistry and function of plant transglutaminases. Minerva Biotechnol 14:135–141
Serafini-Fracassini D, Del Duca S, Beninati S (1995) Plant transglutaminases. Phytochemistry 40:355–365
Shih L, Kaur-Shawhney R, Fuhrer J, Samantha S, Galston AW (1982) Effects of exogenous 1,3-diaminopropane and spermidine on senescence of oat leaves, ethylene production and chlorophyll loss related to polyamine content. Plant Physiol 70:1592–1596
Signorini M, Beninati S, Bergamini C (1991) Identification of transglutaminase activity in the leaves of silver beet (Beta vulgaris L.). J Plant Physiol 137:547–552
Torrigiani P, Serafini-Fracassini D, Biondi S, Bagni N (1986) Evidence for subcellular localization of polyamines and their biosynthetic enzymes in plant cells. J Plant Physiol 124:23–29
Trezzi F, Galli MG, Bellini E (1965) L'osmo-resistenza di Dunaliella salina ricerche ultrastrutturali. G Bot Ital 72:255–263
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfers of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Villalobos E, Torné JM, Rigau J, Olles I, Claparols I, Santos M (2001) Immunogold localization of a transglutaminase related to grana development in different maize cell types. Protoplasma 216:155–163
Acknowledgements
This work was supported by a grant from Consiglio Nazionale delle Ricerche Contributo di Ricerca 98. 00514.CTO4 to D. Serafini-Fracassini and by the University of Bologna (Funds for Selected Topics: 'Segnali molecolari nel differenziamento cellulare'). The authors are grateful to Henkel S.p.A. (Fino Mornasco, Como), Prof. Carla Esposito (Salerno University), and Carlo Bergamini (Ferrara University) for their kind gifts of Deriphat C 160 KPC, TGase antibody, and erythrocyte purified TGase, respectively. We acknowledge N. Mele for technical assistance with photography and imaging, and the 'ESF protein Cross-linking-The ESF Transglutaminases Programme', for a concessional Travel Grant to Dr. I. Claparols to visit the Botany Laboratory of Bologna University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dondini, L., Del Duca, S., Dall'Agata, L. et al. Suborganellar localisation and effect of light on Helianthus tuberosus chloroplast transglutaminases and their substrates. Planta 217, 84–95 (2003). https://doi.org/10.1007/s00425-003-0998-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-003-0998-3