Abstract
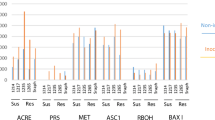
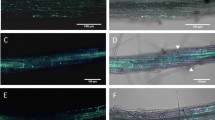
Recently, it has been reported that tubers of transgenic potato (Solanum tuberosum L.) plants with decreased activity of the plastidic ATP/ADP transporter (AATP1) contain less starch, despite having an increased glucose level [P. Geigenberger et al. (2001) Plant Physiol 125:1667–1678]. The metabolic alterations correlated with enhanced resistance to the bacterium Erwinia carotovora. Here it is shown that transgenic potato tubers, possessing less starch yet increased glucose levels due to the expression of a cytoplasm-localized yeast invertase, exhibit drastic susceptibility to E. carotovora. In addition, it is demonstrated that AATP1 anti-sense tubers show an increased capacity to ward off the pathogenic fungus Alternaria solani. In contrast to AATP1 anti-sense tubers, the corresponding leaf tissue does not show changes in carbohydrate accumulation. However, upon elicitor treatment, AATP1 anti-sense leaves possess an increased capacity to release H2O2 and activate various defence-related genes, reactions that are associated with substantially delayed appearance of disease symptoms caused by Phytophthora infestans. Grafting experiments between AATP1 anti-sense plants and wild-type plants indicate the presence of a signal that is generated in AATP1 rootstocks and primes wild-type scions for potentiated activation of cellular defence responses in leaves. Together, the results suggest that (i) the enhanced pathogen tolerance of AATP1 anti-sense tubers is not due to "high sugar resistance", (ii) the increased disease resistance of AATP1 anti-sense tubers is effective against different types of pathogen and (iii) a systemic signal induced by antisensing the plastidic ATP/ADP transporter in potato tubers confers increased resistance to pathogens.







Similar content being viewed by others
Abbreviations
- AATP1:
-
plastidic ATP/ADP transporter
- BGL:
-
β-1,3-glucanase
- DHSA:
-
dihydroxy stearic acid
- PAL:
-
phenylalanine ammonia-lyase
- Pmg:
-
cell wall elicitor from Phytophthora sojae
- PR:
-
pathogenesis related
References
Berrocal-Lobo M, Molina A, Solano R (2002) Constitutive expression of ETHYLENE-RESPONSE-FACTOR1 in Arabidopsis confers resistance to several necrotrophic fungi. Plant J 29:23–32
Boch J, Verbsky ML, Roberston, TL, Larkin JC, Kunkel BN (1998) Analysis of resistance gene-mediated defense responses in Arabidopsis thaliana plants carrying a mutation in CPR5. Plant Microbe Interact 12:1196–1206
Bolwell GP, Blee KA, Butt VS, Davies DR, Gardner SL, Gerrish C, MinobayevaF, Rowntree FG, Wojtaszek P (1999) Recent advances in understanding the origin of the apoplastic oxidative burst in plant cells. Free Radic Res [Suppl] 31:137–145
Börnke R, Hajirezaei M-R, Heineke D, Melzer M, Herbers K, Sonnewald U (2002) High-level production of the non-cariogenic sucrose isomer palatinose in transgenic tobacco plants strongly impairs development. Planta 214:356–364
Bradley DJ, Kjellbom P, Lamb CJ (1992) Elicitor- and wound-induced oxidative cross-linking of a proline-rich plant cell wall protein: a novel, rapid defense response. Cell 70:21–30
Bruce RJ, West CA (1989) Elicitation of lignin biosynthesis and isoperoxidase activity by pectic fragments in suspension cultures of castor bean. Plant Physiol 91:889–897
Chen C, Chen Z (2002) Potentiation of developmentally regulated plant defense response by AtWRKY18, a pathogen-induced Arabidopsis transcription factor. Plant Physiol 129:716
Conrath U, Pieterse CMJ, Mauch-Mani B (2002) Priming in plant–pathogen interactions. Trends Plant Sci 7:210–217
Düring K, Porsch P, Fladung M, Lörz H (1993) Transgenic potato plants resistant to the phytopathogenic bacterium Erwinia carotovora. Plant J 3:587–598
Ehness R, Ecker M, Godt DE, Roitsch T (1997) Glucose and stress independently regulate source and sink metabolism and defense mechanisms via signal transduction pathways involving protein phosphorylation. Plant Cell 9:1825–1841
Farré EM, Tiessen A, Roessner U, Geigenberger P, Trethewey RN, Willmitzer L (2001) Analysis of the compartmentation of glycolytic intermediates, nucleotides, sugars, organic acids, amino acids, and sugar alcohols in potato tubers using a nonaqueous fractionation method. Plant Physiol 127:685–700
Fauth M, Merten A, Hahn MG, Jeblick W, Kauss H (1996) Competence for elicitation of H2O2 in hypocotyls of cucumber is induced by breaching the cuticle and is enhanced by salicylic acid. Plant Physiol 110:347–354
Frommer WB, Sonnewald U (1995) Molecular analysis of carbon partitioning in solanaceous species. J Exp Bot 46:587–607
Geigenberger P, Stamme C, Tjaden J, Schulze A, Quick WP, Betsche T, Kersting HJ, Neuhaus HE (2001) Tuber physiology and properties of starch from tubers of transgenic potato plants with altered plastidic adenylate transporter activity. Plant Physiol 125:1667–1678
Hajirezaei M-R, Takahata Y, Trethewey RN, Willmitzer L, Sonnewald U (2000) Impact of elevated cytosolic and apoplastic invertase activity on carbon metabolism during potato tuber development. J Exp Bot 51:439–445
Heineke D, Sonnewald U, Büssis D, Gunter G, Leidreiter K, Wilke I, Raschke K, Willmitzer L, Heldt HW (1992) Apoplastic expression of yeast-derived invertase in potato. Effects on photosynthesis, leaf solute composition, water relations, and tuber composition. Plant Physiol 100:301–308
Herbers K, Meuwly P, Frommer WB, Métraux J-P, Sonnewald U (1996a) Systemic acquired resistance mediated by the ectopic expression of invertase: possible hexose sensing in the secretory pathway. Plant Cell 8:793–803
Herbers K, Meuwly P, Métraux J-P, Sonnewald U (1996b) Salicylic acid-independent induction of pathogenesis-related protein transcripts by sugars is dependent on leaf developmental stage. FEBS Lett 397:239–244
Horsfall JG, Dimond AE (1957) Interactions of tissue sugar, growth substances, and disease susceptibility. Z Pflanzenkr Pflanzenschutz 27:415–421
Jackson SD, James P, Prat S, Thomas B (1998) Phytochrome B affects the levels of a graft-transmissible signal involved in tuberization. Plant Physiol 117:29–32.
Jang JC, Sheen J (1994) Sugar sensing in higher plants. Plant Cell 6:1665–1679
Johnson R, Ryan CA (1990) Wound-inducible potato inhibitor II genes: enhancement of expression by sucrose. Plant Mol Biol 14:527–536
Kauss H (1992) Callose and callose synthase. In: Guro SJ, McPherson MJ, Bowles DJ (eds) Molecular plant pathology. Oxford University Press, Oxford, pp 1–8
Kauss H, Fauth M, Merten A, Jeblick W (1999) Cucumber hypocotyls respond to cutin monomers via both an inducible and a constitutive H2O2-generating system. Plant Physiol 120:1175–1182
Keil M, Sanchez-Serrano JJ, Willmitzer L (1986) Primary structure of a proteinase inhibitor II gene from potato (Solanum tuberosum). Nucleic Acids Res 14:5641–5650
Kombrink E, Hahlbrock K, Hinze K, Schröder M (1991) Molecular responses of potato to infection with Phytophtora infestans. In: Smith CJ (ed) Biochemistry and molecular biology of plant–pathogen interaction. Clarendon Press, Oxford
Lalonde S, Boles E, Hellmann H, Barker L, Patrick JW, Frommer WB, Ward JM (1999) The dual function of sugar carriers: transport and sugar sensing. Plant Cell 11:707–726
Lamb C, Dixon R (1997) The oxidative burst in plant disease resistance. Annu Rev Plant Physiol Plant Mol Biol 48:251–275
Levine, A, Tenhaken R, Dixon R, Lamb C (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79:583–593
Linke, C, Conrath U, Jeblick W, Betsche T, Mahn A, Düring K, Neuhaus HE. (2002) Inhibition of the plastidic ATP/ADP-transporter protein primes potato tubers for augmented elicitation of defense responses and enhances their resistance against Erwinia carotovora. Plant Physiol 129:1607–1615
Neuhaus HE, Emes MJ (2000) Development of non-green plastids. Annu Rev Plant Physiol Plant Mol Biol 51:111–140
Oparka KJ, Prior DAM (1988) Movement of lucifer yellow CH in potato tuber storage tissue: a comparison of symplastic and apoplastic transport. Planta 176:533–540
Otazu V, Secor GA (1981) Soft rot susceptibility of potatoes with high reducing sugar content. Phytopathology 71:290–295
Renz A, Stitt M (1993) Substrate specificity and product inhibition of different forms of fructokinase and hexokinase in developing potato tubers. Planta 190:166–175
Robinson SP, Wiskich JT (1977) Uptake of ATP analogs by isolated pea chloroplasts and their effect on CO2 fixation and electron transport. Biochim Biophys Acta 461:131–140
Stitt M, McLilley R, Gerhardt R, Heldt HW (1989) Metabolite levels in specific cells and subcellular compartments of plant leaves. Methods Enzymol 174:518–552
Thines E, Weber RW, Talbot NJ (2000) MAP kinase and protein kinase A-dependent mobilization of triacylglycerol and glycogen during appressorium turgor generation by Magnaporthe grisea. Plant Cell 12:1703–1718
Tjaden J, Möhlmann T, Kampfenkel K, Henrichs G, Neuhaus HE (1998) Altered plastidic ATP/ADP-transporter activity influences potato (Solanum tuberosum) morphology, amount and composition of tuber starch, and tuber morphology. Plant J 16:531–540
Tsukaya H, Ohshima T, Naito S, Chino M, Komeda Y (1991) Sugar-dependent expression of the CHS-A gene for chalcone synthase from petunia in transgenic Arabidopsis. Plant Physiol 97:1414–1421
Van Loon LC, Van Strien EA (1999) The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1 type proteins. Physiol Mol Plant Pathol 55:85–97
Yu I-C, Parker J, Bent AF (1998) Gene-for-gene disease resistance without the hypersensitive response in Arabidopsis dnd1 mutant. Proc Natl Acad Sci USA 95:7819–7824
Acknowledgements
We thank Drs. Jürgen Siegrist and Eckhard Thines for providing fungal and oomycete cultures, respectively. Prof. Klaus Hahlbrock, and Drs. Erich Kombrink and Guenther Strittmatter are thanked for providing cDNA clones. The provision of yeast invertase-expressing plants, originally generated by Prof. U. Sonnewald (Gatersleben), by Dr. U. Rössner (Max Planck Institute for Molecular Plant Physiology, Golm, Germany) is greatly appreciated. We thank Prof. U. Sonnewald for allowing us to work with the invertase potato lines. In the laboratories of U.C. and H.E.N., this project is financially supported by the Deutsche Forschungsgemeinschaft (Ne 418/4-1), and the Schwerpunkt Biotechnologie of the Federal State Rheinland Pfalz.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Conrath, U., Linke, C., Jeblick, W. et al. Enhanced resistance to Phytophthora infestans and Alternaria solani in leaves and tubers, respectively, of potato plants with decreased activity of the plastidic ATP/ADP transporter. Planta 217, 75–83 (2003). https://doi.org/10.1007/s00425-003-0974-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-003-0974-y