Abstract
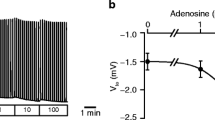
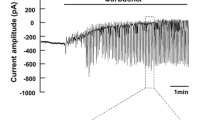
Acetylcholine-controlled exocrine secretion by pancreatic acini has been explained by two hypotheses. One suggests that NaCl secretion occurs by secondary active secretion as has been originally described for the rectal gland of Squalus acanthias. The other is based on a “push-pull” model whereby Cl- is extruded luminally and sequentially taken up basolaterally. In the former model Cl- uptake is coupled to Na+ and basolateral K+ conductances play a crucial role, in the latter model, Na+ uptake supposedly occurs via basolateral non-selective cation channels. The present whole-cell patch-clamp studies were designed to further explore the conductive properties of rat pancreatic acini. Pilot studies in approximately 300 cells revealed that viable cells usually had a membrane voltage (Vm) more hyperpolarized than - 30 mV. In all further studies Vm had to meet this criterion. Under control conditions Vm was - 49 ± 1 mV (n = 149). The fractional K+ conductance (fK) was 0.13 ± 0.1 (n = 49). Carbachol (CCH, 0.5 μmol/1) depolarized to-19 ± 1.1 mV (n = 63) and increased the membrane conductance (Gm) by a factor of 2-3. In the seeming absence of Na+ [replacement by N-methyl-D-glucamine (NMDG+)] Vm hyperpolarized slowly to -59 ±2 mV (n = 90) and CCH still induced depolarizations to- 24 ± 2 mV (n = 34). The hyperpolarization induced by NMDG+ was accompanied by a fall in cytosolic pH by 0.4 units, and a very slow and slight increase in cytosolic Ca2+. (fK) increased to 0.34. The effect of NMDG+ on Vm was mimicked by the acidifying agents propionate and acetate (10mmol/l) added to the bath. The present study suggests that fK makes a substantial contribution to Gm under control conditions. The NMDG+ experiments indicate that the non-selective cation conductance contributes little to Vm in the presence of CCH. Hence the present data in rat pancreatic acinar cells do not support the push-pull model.
Similar content being viewed by others
References
Ecke D, Bleich M, Greger R (1996) Crypt base cells show cAMP-dependent Cl- secretion but no cation inward current. Pflüegers Arch 431: 427–434
Greger R (1985) Ion transport mechanisms in thick ascending limb of Henle’s loop of mammalian nephron. Physiol Rev 65:760–797
Greger R, Schlatter E, Wang F, Forrest JNJ (1984) Mechanism of NaCl secretion in rectal gland tubules of spiny dogfish (Squalus acanthias). III. Effects of stimulation of secretion by cyclic AMP. Pflügers Arch 402:376–384
Greger R, Schlatter E, Gögelein H (1986) Sodium chloride secretion in rectal gland of dogfish Squalus acanthias. News Physiol Sci 1:134–136
Grynkiewicz G, Poenie M, Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260:3440–3450
Guinamard R, Chraibi A, Teulon J (1995) A small conductance Cl channel in the mouse thick ascending limb that is activated by ATP and protein kinase A. J Physiol (Lond) 485:97–112
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ (1981) Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflügers Arch 391:85–100
Horn R, Marty A (1988) Muscarinic activation of ionic currents measured by a new whole-cell recording method. J Gen Physiol 92:145–159
Iwatsuki N, Maruyama Y, Matsumoto O, Nishiyama A (1985) Activation of Ca2+-dependent Cl- and K+ conductances in rat and mouse parotid acinar cells. Jpn J Physiol 35:933–944
Kasai H, Augustine GJ (1990) Cytosolic Ca2+ gradients triggering unidirectional fluid secretion from exocrine pancreas. Nature 348:735–738
Kasai H, Petersen OH (1994) Spatial dynamics of second messengers: IP3 and cAMP as long-range and associative messengers. Trends Neurosci 17:95–101
Kasai H, Li YX, Miyashita Y (1993) Subcellular distribution of Ca2+ release channels underlying Ca2+ waves and oscillations in exocrine pancreas. Cell 74:669–677
Kerst G, Fischer KG, Normann C, Kramer A, Leipziger J, Greger R (1995) Ca2+ influx induced by store release and cytosolic Ca2+ chelation in HT29 colon carcinoma cells. Pflügers Arch 430:653–665
Klär B, Leipziger J, Nitschke R, Greger R (1993) Ca2+ as a second messenger in CFPAC-1 cells. Cell Physiol Biochem 3:17–27
KöttgenM, Leipziger J, Fischer KG, Greger R (1994) pH regulation in HT29 colon carcinoma cells. Pflügers Arch 428: 179–185
Nitschke R, Fröbe U, Greger R (1991) Antidiuretic hormone acts via VI receptors on intracellular calcium in the isolated perfused rabbit cortical thick ascending limb. Pflügers Arch Eur J Physiol 417:622–632
Nitschke R, Leipziger J, Greger R (1993) Agonist induced intracellular Ca2+ transients in HT29, cells. Pflügers Arch 423: 519–526
Petersen OH (1992) Stimulus-secretion coupling: cytoplasmic calcium signals and the control of ion channels in exocrine acinar cells. J Physiol (Lond) 448:1–51
Petersen OH, Maruyama Y (1984) Calcium-activated potassium channels and their role in secretion. Nature 307: 693–696
Bleich M, Riedemann N, Warth R, Kerstan D, Leipziger J, Hör M, Van Driessche W, Greger R (1996) Ca2+ regulated K+ and non-selective cation channels in the basolateral membrane of rat colonic crypt base cells Pflügers Arch (submitted)
Robertson MA, Foskett JK (1994) Na2+ transport pathways in secretory acinar cells: membrane cross talk mediated by [Cl]j. Am J Physiol 267:C146-C156
Schlatter E, Greger R, Weidtke C (1983) Effect of “high ceiling” diuretics on active salt transport in the cortical thick ascending limb of Henle’s loop of rabbit kidney. Correlation of chemical structure and inhibitory potency. Pflügers Arch 396:210–217
Schmid A, Schulz I (1995) Characterization of single potassium channels in mouse pancreatic acinar cells. J Physiol (Lond) 484:661–676
Suzuki K, Petersen OH (1988) Patch-clamp study of single- channel and whole-cell K+ currents in guinea pig pancreatic acinar cells. Am J Physiol 255:G275-G285
Suzuki K, Onoe K, Takahira H (1992) Activation of Ca2+- dependent K+ channel and Cl- conductance in canine pancreatic acinar cells through a cyclic AMP pathway. Jpn J Physiol 42:267–281
Thorn P, Petersen OH (1992) Activation of nonselective cation channels by physiological cholecystokinin concentrations in mouse pancreatic acinar cells. J Gen Physiol 100:11–25
Thorn P, Petersen OH (1994) A voltage-sensitive transient potassium current in mouse pancreatic acinar cells. Pflügers Arch 428:288–295
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Slawik, M., Zdebik, A., Hug, M.J. et al. Whole-cell conductive properties of rat pancreatic acini. Pflügers Arch — Eur J Physiol 432, 112–120 (1996). https://doi.org/10.1007/s004240050112
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s004240050112