Abstract
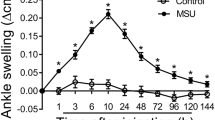
Acid-sensing ion channels (ASICs) in dorsal root ganglion (DRG) neurons play an important role in inflammatory pain. The objective of this study is to observe the regulatory role of ASICs in monosodium urate (MSU) crystal-induced gout pain and explore the basis for ASICs in DRG neurons as a target for gout pain treatment. The gout arthritis model was induced by injecting MSU crystals into the ankle joint of mice. The circumference of the ankle joint was used to evaluate the degree of swelling; the von Frey filaments were used to determine the withdrawal threshold of the paw. ASIC currents and action potentials (APs) were recorded by patch clamp technique in DRG neurons. The results displayed that injecting MSU crystals caused ankle edema and mechanical hyperalgesia of the paw, which was relieved after amiloride treatment. The ASIC currents in DRG neurons were increased to a peak on the second day after injecting MSU crystals, which were decreased after amiloride treatment. MSU treatment increased the current density of ASICs in different diameter DRG cells. MSU treatment does not change the characteristics of AP. The results suggest that ASICs in DRG neurons participate in MSU crystal-induced gout pain.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adedara IA, Costa FV, Biasuz E, Canzian J, Farombi EO, Rosemberg DB (2022) Influence of acid-sensing ion channel blocker on behavioral responses in a zebrafish model of acute visceral pain. Behav Brain Res 416:113565. https://doi.org/10.1016/j.bbr.2021.113565
Deval E, Gasull X, Noël J, Salinas M, Baron A, Diochot S, Lingueglia E (2010) Acid-sensing ion channels (ASICs): pharmacology and implication in pain. Pharmacol Ther 128:549–558. https://doi.org/10.1016/j.pharmthera.2010.08.006
Deval E, Noël J, Lay N, Alloui A, Diochot S, Friend V, Jodar M, Lazdunski M, Lingueglia E (2008) ASIC3, a sensor of acidic and primary inflammatory pain. Embo J 27:3047–3055. https://doi.org/10.1038/emboj.2008.213
Diochot S, Baron A, Salinas M, Douguet D, Scarzello S, Dabert-Gay AS, Debayle D, Friend V, Alloui A, Lazdunski M, Lingueglia E (2012) Black mamba venom peptides target acid-sensing ion channels to abolish pain. Nature 490:552–555. https://doi.org/10.1038/nature11494
Ferreira J, Santos AR, Calixto JB (1999) Antinociception produced by systemic, spinal and supraspinal administration of amiloride in mice. Life Sci 65:1059–1066. https://doi.org/10.1016/s0024-3205(99)00336-7
Gao M, Long H, Ma W, Liao L, Yang X, Zhou Y, Shan D, Huang R, Jian F, Wang Y, Lai W (2016) The role of periodontal ASIC3 in orofacial pain induced by experimental tooth movement in rats. Eur J Orthod 38:577–583. https://doi.org/10.1093/ejo/cjv082
Gautam M, Benson CJ, Sluka KA (2010) Increased response of muscle sensory neurons to decreases in pH after muscle inflammation. Neuroscience 170:893–900. https://doi.org/10.1016/j.neuroscience.2010.08.003
Gründer S, Pusch M (2015) Biophysical properties of acid-sensing ion channels (ASICs). Neuropharmacology 94:9–18. https://doi.org/10.1016/j.neuropharm.2014.12.016
Gu Q, Lee LY (2010) Acid-sensing ion channels and pain. Pharmaceuticals (Basel, Switzerland) 3:1411–1425. https://doi.org/10.3390/ph3051411
Hesselager M, Timmermann DB, Ahring PK (2004) pH Dependency and desensitization kinetics of heterologously expressed combinations of acid-sensing ion channel subunits. J Biol Chem 279:11006–11015. https://doi.org/10.1074/jbc.M313507200
Hong J, Qiu J, Wang X, Zhang G (2020) Characteristics of voltage-gated potassium currents in monosodium urate induced gouty arthritis in mice. Inflamm Res 69:589–598. https://doi.org/10.1007/s00011-020-01343-z
Hsieh WS, Kung CC, Huang SL, Lin SC, Sun WH (2017) TDAG8, TRPV1, and ASIC3 involved in establishing hyperalgesic priming in experimental rheumatoid arthritis. Sci Rep 7:8870. https://doi.org/10.1038/s41598-017-09200-6
Ikeuchi M, Kolker SJ, Burnes LA, Walder RY, Sluka KA (2008) Role of ASIC3 in the primary and secondary hyperalgesia produced by joint inflammation in mice. Pain 137:662–669. https://doi.org/10.1016/j.pain.2008.01.020
Izumi M, Ikeuchi M, Ji Q, Tani T (2012) Local ASIC3 modulates pain and disease progression in a rat model of osteoarthritis. J Biomed Sci 19:77. https://doi.org/10.1186/1423-0127-19-77
Jeong S, Lee SH, Kim YO, Yoon MH (2013) Antinociceptive effects of amiloride and benzamil in neuropathic pain model rats. J Korean Med Sci 28:1238–1243. https://doi.org/10.3346/jkms.2013.28.8.1238
Leng TD, Si HF, Li J, Yang T, Zhu M, Wang B, Simon RP, Xiong ZG (2016) Amiloride analogs as ASIC1a inhibitors. CNS Neurosci Ther 22:468–476. https://doi.org/10.1111/cns.12524
Liu HJ, Pan XX, Liu BQ, Gui X, Hu L, Jiang CY, Han Y, Fan YX, Tang YL, Liu WT (2017) Grape seed-derived procyanidins alleviate gout pain via NLRP3 inflammasome suppression. J Neuroinflammation 14:74. https://doi.org/10.1186/s12974-017-0849-y
Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440:237–241. https://doi.org/10.1038/nature04516
Munro G, Christensen JK, Erichsen HK, Dyhring T, Demnitz J, Dam E, Ahring PK (2016) NS383 selectively inhibits acid-sensing ion channels containing 1a and 3 subunits to reverse inflammatory and neuropathic hyperalgesia in rats. CNS Neurosci Ther 22:135–145. https://doi.org/10.1111/cns.12487
Nagae M, Hiraga T, Wakabayashi H, Wang L, Iwata K, Yoneda T (2006) Osteoclasts play a part in pain due to the inflammation adjacent to bone. Bone 39:1107–1115. https://doi.org/10.1016/j.bone.2006.04.033
Orchard CH, Cingolani HE (1994) Acidosis and arrhythmias in cardiac muscle. Cardiovasc Res 28:1312–1319. https://doi.org/10.1093/cvr/28.9.1312
Páez O, Segura-Chama P, Almanza A, Pellicer F, Mercado F (2021) Properties and differential expression of H+ receptors in dorsal root ganglia: is a labeled-line coding for acid nociception possible? Front Physiol 12:733267. https://doi.org/10.3389/fphys.2021.733267
Qiu J, Xu X, Zhang S, Li G, Zhang G (2021) Modulations of Nav1.8 and Nav1.9 channels in monosodium urate-induced gouty arthritis in mice. Inflammation 44:1405–1415. https://doi.org/10.1007/s10753-021-01425-y
Reeh PW, Kress M (2001) Molecular physiology of proton transduction in nociceptors. Curr Opin Pharmacol 1:45–51. https://doi.org/10.1016/s1471-4892(01)00014-5
Reeh PW, Steen KH (1996) Tissue acidosis in nociception and pain. Prog Brain Res 113:143–151. https://doi.org/10.1016/s0079-6123(08)61085-7
Riemann A, Wußling H, Loppnow H, Fu H, Reime S, Thews O (2016) Acidosis differently modulates the inflammatory program in monocytes and macrophages. Biochim Biophys Acta 1862:72–81. https://doi.org/10.1016/j.bbadis.2015.10.017
Rocha-González HI, Herrejon-Abreu EB, López-Santillán FJ, García-López BE, Murbartián J, Granados-Soto V (2009) Acid increases inflammatory pain in rats: effect of local peripheral ASICs inhibitors. Eur J Pharmacol 603:56–61. https://doi.org/10.1016/j.ejphar.2008.12.017
Roh J, Hwang SM, Lee SH, Lee K, Kim YH, Park CK (2020) Functional expression of Piezo1 in dorsal root ganglion (DRG) Neurons. Int J Mol Sci 21:3834. https://doi.org/10.3390/ijms21113834
Romanelli MR, Thayer JA, Neumeister MW (2020) Ischemic pain. Clin Plast Surg 47:261–265. https://doi.org/10.1016/j.cps.2019.11.002
Sheng A, Zhang Y, Li G, Zhang G (2018) Inhibitory effects of honokiol on the voltage-gated potassium channels in freshly isolated mouse dorsal root ganglion neurons. Neurochem Res 43:450–457. https://doi.org/10.1007/s11064-017-2440-4
Sluka KA, Radhakrishnan R, Benson CJ, Eshcol JO, Price MP, Babinski K, Audette KM, Yeomans DC, Wilson SP (2007) ASIC3 in muscle mediates mechanical, but not heat, hyperalgesia associated with muscle inflammation. Pain 129:102–112. https://doi.org/10.1016/j.pain.2006.09.038
Tatlock S, Rüdell K, Panter C, Arbuckle R, Harrold LR, Taylor WJ, Symonds T (2017) What outcomes are important for gout patients? In-depth qualitative research into the gout patient experience to determine optimal endpoints for evaluating therapeutic interventions. The patient 10:65–79. https://doi.org/10.1007/s40271-016-0184-x
Voilley N, De Weille J, Mamet J, Lazdunski M (2001) Nonsteroid anti-inflammatory drugs inhibit both the activity and the inflammation-induced expression of acid-sensing ion channels in nociceptors. J Neurosci 21:8026–8033. https://doi.org/10.1523/JNEUROSCI.21-20-08026.2001
Walder RY, Rasmussen LA, Rainier JD, Light AR, Wemmie JA, Sluka KA (2010) ASIC1 and ASIC3 play different roles in the development of Hyperalgesia after inflammatory muscle injury. J Pain 11:210–218. https://doi.org/10.1016/j.jpain.2009.07.004
Woo YC, Park SS, Subieta AR, Brennan TJ (2004) Changes in tissue pH and temperature after incision indicate acidosis may contribute to postoperative pain. Anesthesiology 101:468–475. https://doi.org/10.1097/00000542-200408000-00029
Xu M, Wu R, Zhang L, Zhu HY, Xu GY, Qian W, Zhang PA (2020) Decreased MiR-485-5p contributes to inflammatory pain through post-transcriptional upregulation of ASIC1 in rat dorsal root ganglion. J Pain Res 13:3013–3022. https://doi.org/10.2147/jpr.S279902
Xu X, Yuan Z, Zhang S, Li G, Zhang G (2022) Regulation of TRPV1 channel in monosodium urate-induced gouty arthritis in mice. Inflamm Res 71:485–495. https://doi.org/10.1007/s00011-022-01561-7
Yen YT, Tu PH, Chen CJ, Lin YW, Hsieh ST, Chen CC (2009) Role of acid-sensing ion channel 3 in sub-acute-phase inflammation. Mol Pain 5:1. https://doi.org/10.1186/1744-8069-5-1
Zhao K, An R, Xiang Q, Li G, Wang K, Song Y, Liao Z, Li S, Hua W, Feng X, Wu X, Zhang Y, Das A, Yang C (2021) Acid-sensing ion channels regulate nucleus pulposus cell inflammation and pyroptosis via the NLRP3 inflammasome in intervertebral disc degeneration. Cell Prolif 54:e12941. https://doi.org/10.1111/cpr.12941
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors participated in the study and supported the publication. Z. G. wrote the manuscript and designed the study. Y. Z. performed research, analyzed the data, and prepared figures. M. L. and Z. S. conducted material preparation and data. L. H. and L. G. designed the study. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All protocols involving animal experiments were approved by the Institutional Animal Care and Use Committee of China Pharmaceutical University (Permission Number: CPU2019-05–001). All protocols were performed following the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, Z., Miao, L., Zhang, S. et al. The role of acid-sensing ion channels in monosodium urate-induced gouty pain in mice. Pflugers Arch - Eur J Physiol 476, 101–110 (2024). https://doi.org/10.1007/s00424-023-02862-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-023-02862-4