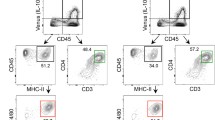
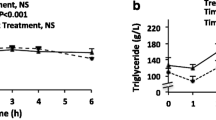
Abstract
Proper food intake is important for maintaining good health in humans. Chocolate is known to exert anti-inflammatory effects; however, the mechanisms remain unclear. In this study, we aimed to investigate the effects of cocoa butter intake on gut immunity in rats and rabbits. Cocoa butter intake increased the lymph flow, cell density, and IL-1β, IL-6 and IL-10 levels in mesenteric lymph. Clodronate, a macrophage depletion compound, significantly enhanced the release of all cytokines. The immunoreactivities of macrophage markers CD68 and F4/80 in the jejunal villi were significantly decreased with clodronate. Piceatannol, a selective cell surface ATP synthase inhibitor significantly reduced the cocoa butter intake-mediated releases of IL-1β, IL-6 and IL-10. The immunoreactivities of cell surface ATP synthase were observed in rat jejunal villi. Shear stress stimulation on the myofibroblast cells isolated from rat jejunum released ATP and carbon dioxide depended with H+ release. In rabbit in vivo experiments, cocoa butter intake increased the concentrations of ATP and H+ in the portal vein. The in vitro experiments with isolated cells of rat jejunal lamina propria the pH of 3.0 and 5.0 in the medium released significantly IL-1β and IL-6. ATP selectively released IL-10. These findings suggest that cocoa butter intake regulates the gut immunity through the release and transport of IL-1β, IL-6, and IL-10 into mesenteric lymph vessels in a negative feedback system. In addition, the H+ and ATP released from cell surface ATP synthase in jejunal villi play key roles in the cocoa butter intake–mediated regulation of gut immunity.







Similar content being viewed by others
Data availability
All relevant data are available from the corresponding author on request.
References
Bamias G, Cominelli F (2016) Cytokines and intestinal inflammation. Curr Opin Gastroenterol 32:437–442
Chien S (2007) Mechanotransduction and endothelial cell homeostasis: the wisdom of the cell. Am J Physiol Heart Circ Physiol 292:H1209–H1224
Choy EH, Beneetti FD, Takeuchi T, Hashizume M, John MR, Kishimoto T (2020) Translating IL-6 biology into effective treatments. Nat Rev Rheumatol 16:335–345
Ding H, Li Y, Li X, Liu X, Chen S, Liu M, Zeng H (2020) Treatment with 7% and 10% CO2 enhanced expression of IL-1β, TNF-α, and IL-6 in hypoxic cultures of human whole blood. J Int Med Res 48:300060520912105
Dos Anjos CA (2017) F4/80 as a major macrophage marker: the case of the peritoneum and spleen. Results Probl Cell Differ 62:161–179
Furuya K, Sokabe M, Furuya S (2005) Characteristics of subepithelial fibroblasts as a mechano-sensor in the intestine: cell-shape-dependent ATP release and P2Y1 signaling. J Cell Sci 118:3289–3304
Hayashi M, Watanabe-Asaka T, Nagashio S, Kaidoh M, Yokoyama Y, Maejima D, Kajihara R, Amari R, Arai N, Kawai Y, Ohhashi T (2021) Water intake accelerates ATP release from myofibroblast cells in rats: ATP-mediated podoplanin-dependent control for physiological function and immunity. Am J Physiol Gastrointest Liver Physiol 320:G54–G65
Hirano S, Zhou Q, Furuyama A, Kanno S (2017) Differential regulation of IL-1β and IL-6 in murine macrophages. Inflammation 40:1933–1943
Jancic CC, Cabrini M, Gabelloni ML, Rodrigues CR, Salamone G, Trevani AS, Geffner J (2012) Lower extracellular pH stimulates the production of IL-1β by human monocytes. Cytokine 57:258–268
Kaibara E (1712) Youjoukun, Fukuoka, Fukuokahan, 1712
Kawai Y, Kaidoh M, Ohhashi T (2008) MDA-MB-231 produces ATP-mediated ICAM-1-dependent facilitation of the attachment of carcinoma cells to human lymphatic endothelial cells. Am J Physiol Cell Physiol 295:C1123–C1132
Kawai Y, Yokoyama Y, Kaidoh M, Ohhashi T (2010) Shear stress-induced ATP-mediated endothelial constitutive nitric oxide synthase expression in human lymphatic endothelial cells. Am J Physiol Cell Physiol 298:C647–C655
Kawai Y, Yoshida K, Kaidoh M, Yokoyama Y, Ohhashi T (2012) Shear stress-mediated F1/Fo ATP synthase-dependent CO2 gas excretion from human pulmonary arteriolar endothelial cells. J Cell Physiol 227:2059–2068
Kim SH, Lee HY, Jang YS (2015) Expression of the ATP-gated P2X7 receptor on M cells and its modulating role in the mucosal immune environment. Immune New 15:44–49
Latz E, Xiao TS, Stutz A (2013) Activation and regulation of the inflammasomes. Nat Rev Immunol 3:397–411
Lippi D (2013) Chocolate in history: food, medicine, medi-food. Nutrients 5:1573–1584
McKay DM, Mancini NL, Shearer J, Shutt T (2020) Perturbed mitochondrial dynamics, an emerging aspect of epithelial-microbe interactions. Am J Physiol Gastrointest Liver Physiol 318:G748–G762
Miura S, Sekizuka E, Nagata H, Oshio C, Minamitani H, Suematsu M, Suzuki M, Hamada Y, Kobayashi H, Tsuchiya M (1987) Increased lymphocyte transport by lipid absorption in rat mesenteric lymphatics. Am J Physiol Gastrointest Liver Physiol 253:G596–G600
Moreno SG (2018) Depleting macrophages in vivo with clodronate liposomes. Methods Mol Biol 84:259–262
Nagashio S, Ajima K, Maejima D, Sanjo H, Kajihara R, Hayashi M, Watanabe-Asaka T, Kaidoh M, Yokoyama Y, Taki S, Kawai Y, Ohhashi T (2019) Water intake increases mesenteric lymph flow and the total flux of albumin, long-chain fatty acids, and IL-22 in rats: new concept of absorption in jejunum. Am J Physiol Gastrointest Liver Physiol 316:G155–G165
Pang G, Couch L, Batey R, Clancy R, Cripps A (1994) GM-CSF, IL-1alpha, IL-1beta, IL-6, IL-10, ICAM-1 and VCAM-1 gene expression and cytokine production in human duodenal fibroblasts stimulated with lipopolysaccharide, IL-1alpha and TNF-alpha. Clin Exp Immunol 96:437–443
Rashed R, Zaki MAM, Mohamed NAW, Mansou OM, Refaey FAHE (2021) Prognostic value of tumor associated macrophage markers CD163 and CD68 immunohistochemistry in classical Hodgkin lymphoma. Clin Lab. https://doi.org/10.7754/Clin.Lab.2020.200920
Saraiva M, O’Garra A (2010) The regulation of IL-10 production by immune cells. Nat Rev Immunol 10:170–181
Schnell A, Huang L, Singer M, Singaraju A, Barilla RM, Regan BML, Bollhagen A, Thakore PI, Dionne D, Delorey TM, Pawlak M, Horste GMZ, Rozenblatt-Rosen O, Irizarry RA, Regev A, Kucchroo VK (2021) Stem-like intestinal Th17 cells give rise to pathogenic effector T cells during autoimmunity. Cell 184:6281–6298
Thomson AB, Schoeller C, Keelean M, Smith L, Clandinin MT (1993) Lipid absorption: passing through the unstirred layers, brush-border membrane, and beyond. Can J Physiol Pharmacol 71:531–555
Verna R (2013) The history and science of chocolate. Malays J Pathol 35:111–121
Watanabe-Asaka T, Hayashi M, Maejima D, Kawai Y, Ohhashi T (2021) From digestion and absorption to innate immunity and health care: water and food intake may contribute to IL-22 in ILC3-dependent mucosal immunity in the jejunum. J Physiol Sci 71:31
Wei HX, Wang B, Li B (2020) IL-10 and IL-22 in mucosal immunity: driving protection and pathology. Front Immunol. https://doi.org/10.3389/fimmu.2020.01315
Xie Q, Shen WW, Zhong J, Huang C, Zhang C, Li J (2014) Lipopolysaccharide/adenosine triphosphate induces IL-1β and IL-18 secretion through NLRP3 inflammasome in RAW264.7 murine macrophage cells. Int J Mol Med 34:341–349
Yamamoto K, Shimizu N, Obi S, Kumagaya S, Taketani Y, Kamiya A, Ando J (2007) Involvement of cell surface ATP synthase in flow-induced ATP release by vascular endothelial cells. Am J Physiol Heart Circ Physiol 293:H1646–H1653
Zhang L, Wei X, Zhang R, Petitte JN, Si D, Li Z, Cheng J, Du M (2019) Design and development of a novel peptide for treating intestinal inflammation. Front Immunol. https://doi.org/10.3389/fimmu.2019.01841
Acknowledgements
We thank Editage for English language editing and their support to non-native English speakers.
Funding
The Department of Innovation of Medical and Health Sciences Research at Shinshu University School of Medicine was established and supported financially by the donation of BOURBON, Co., Ltd, Kashiwazaki, Niigata, Japan and Aizawa Hospital, Matsumoto, Nagano, Japan. The authors declare that this study received funding from BOURBON Co. Ltd. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
Author information
Authors and Affiliations
Contributions
T.O. designed the experiments, analyzed the data, constructed all figures, and wrote the manuscript. Y.K., M.H., T-W. A., and N.A. designed the experiments, analyzed the data, and revised the manuscript. K.A., R.K., M.T., N.K., D.M., Y.Y., and M.K., performed the experiments, and analyzed data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study and all experimental protocols were approved by the Institutional Animal Care and Use Committee of Shinshu University (1st April, 2019).
Consent for publication
All authors approved the final version of the manuscript and the publication of this manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arai, N., Kajihara, R., Takasaka, M. et al. Cell surface ATP synthase-released H+ and ATP play key roles in cocoa butter intake-mediated regulation of gut immunity through releases of cytokines in rat. Pflugers Arch - Eur J Physiol 475, 945–960 (2023). https://doi.org/10.1007/s00424-023-02822-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-023-02822-y