Abstract
Cardiac fibroblasts play an important role in cardiac matrix turnover and are involved in cardiac fibrosis development. Ca2+ is a driving belt in this phenomenon. This study evaluates the functional expression and contribution of the Ca2+-activated channel TRPM4 in atrial fibroblast phenotype. Molecular and electrophysiological investigations were conducted in human atrial fibroblasts in primary culture and in atrial fibroblasts obtained from wild-type and transgenic mice with disrupted Trpm4 gene (Trpm4−/−). A typical TRPM4 current was recorded on human cells (equal selectivity for Na+ and K+, activation by internal Ca2+, voltage sensitivity, conductance of 23.2 pS, inhibition by 9-phenanthrol (IC50 = 6.1 × 10−6 mol L−1)). Its detection rate was 13% on patches at days 2–4 in culture but raised to 100% on patches at day 28. By the same time, a cell growth was observed. This growth was smaller when cells were maintained in the presence of 9-phenanthrol. Similar cell growth was measured on wild-type mice atrial fibroblasts during culture. However, this growth was minimized on Trpm4−/− mice fibroblasts compared to control animals. In addition, the expression of alpha smooth muscle actin increased during culture of atrial fibroblasts from wild-type mice. This was not observed in Trpm4−/− mice fibroblasts. It is concluded that TRPM4 participates in fibroblast growth and could thus be involved in cardiac fibrosis.






Similar content being viewed by others
Data availability
The dataset generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adapala RK, Thoppil RJ, Luther DJ, Paruchuri S, Meszaros JG, Chilian WM, Thodeti CK (2013) TRPV4 channels mediate cardiac fibroblast differentiation by integrating mechanical and soluble signals. J Mol Cell Cardiol 54:45–52
Barbet G, Demion M, Moura IC, Serafini N, Léger T, Vrtovsnik F, Monteiro RC, Guinamard R, Kinet JP, Launay P (2008) The calcium-activated nonselective cation channel TRPM4 is essential for the migration but not the maturation of dendritic cells. Nat Immunol 9:1148–1156
Bers DM, Patton CW, Nuccitelli R (2010) A practical guide to the preparation of Ca2+ buffers. Methods Cell Biol 99:1–26
Bianchi B, Ozhathil LC, Medeiros-Domingo A, Gollob MH, Abriel H (2018) Four TRPM4 cation channel mutations found in cardiac conduction diseases lead to altered protein stability. Front Physiol 9:177
Burris SK, Wang Q, Bulley S, Neeb ZP, Jaggar JH (2015) 9-Phenanthrol inhibits recombinant and arterial myocyte TMEM16A channels. Br J Pharmacol 172:2459–2468
Burstein B, Libby E, Calderone A, Nattel S (2008) Differential behaviors of atrial versus ventricular fibroblasts: a potential role for platelet-derived growth factor in atrial-ventricular remodeling differences. Circulation 117:1630–1641
Chacar S, Farès N, Bois P, Faivre JF (2017) Basic signaling in cardiac fibroblasts. J Cell Physiol 232:725–730
Chatelier A, Mercier A, Tremblier B, Thériault O, Moubarak M, Benamer N, Corbi P, Bois P, Chahine M, Faivre JF (2012) A distinct de novo expression of Nav1.5 sodium channels in human atrial fibroblasts differentiated into myofibroblasts. J Physiol 590:4307–4319
Cho CH, Kim E, Lee YS, Yarishkin O, Yoo JC, Park JY, Hong SG, Hwang EM (2014) Depletion of 14-3-3γ reduces the surface expression of transient receptor potential melastatin 4b (TRPM4b) channels and attenuates TRPM4b-mediated glutamate-induced neuronal cell death. Mol Brain 7:52
Demion M, Bois P, Launay P, Guinamard R (2007) TRPM4, a Ca2+-activated nonselective cation channel in mouse sino-atrial node cells. Cardiovasc Res 73:531–538
Demion M, Thireau J, Gueffier M, Finan A, Khoueiry Z, Cassan C, Serafini N, Aimond F, Granier M, Pasquié JL, Launay P, Richard S (2014) Trpm4 gene invalidation leads to cardiac hypertrophy and electrophysiological alterations. PLoS One 9:e115256
Ding XQ, Ban T, Liu ZY, Lou J, Tang LL, Wang JX, Chu WF, Zhao D, Song BL, Zhang ZR (2017) Transient receptor potential melastatin 4 (TRPM4) contributes to high salt diet-mediated early-stage endothelial injury. Cell Physiol Biochem 41:835–848
Du J, Xie J, Zhang Z, Tsujikawa H, Fusco D, Silverman D, Liang B, Yue L (2010) TRPM7-mediated Ca2+ signals confer fibrogenesis in human atrial fibrillation. Circ Res 106:992–1003
Echeverría C, Montorfano I, Cabello-Verrugio C, Armisén R, Varela D, Simon F (2015) Suppression of transient receptor potential melastatin 4 expression promotes conversion of endothelial cells into fibroblasts via transforming growth factor/activin receptor-like kinase 5 pathway. J Hypertens 33:981–992
El Chemaly A, Guinamard R, Demion M, Fares N, Jebara V, Faivre JF, Bois P (2006) A voltage-activated proton current in human cardiac fibroblasts. Biochem Biophys Res Commun 340:512–516
El Chemaly A, Norez C, Magaud C, Bescond J, Chatelier A, Fares N, Findlay I, Jayle C, Becq F, Faivre JF, Bois P (2014) ANO1 contributes to angiotensin-II-activated Ca2+-dependent Cl- current in human atrial fibroblasts. J Mol Cell Cardiol 68:12–19
Farès N, Bois P, Lenfant J, Potreau D (1998) Characterization of a hyperpolarization-activated current in dedifferentiated adult rat ventricular cells in primary culture. J Physiol 506:73–82
Feng J, Armillei MK, Yu AS, Liang BT, Runnels LW, Yue L (2019) Ca2+ signaling in cardiac fibroblasts and fibrosis-associated heart diseases. J Cardiovasc Dev Dis 6:34
Gonzales AL, Garcia ZI, Amberg GC, Earley S (2010) Pharmacological inhibition of TRPM4 hyperpolarizes vascular smooth muscle. J Cardiovasc Dev Dis 299:1195–1202
Grand T, Demion M, Norez C, Mettey Y, Launay P, Becq F, Bois P, Guinamard R (2008) Br J Pharmacol 153:697–1705
Gueffier M, Zintz J, Lambert K, Finan A, Aimond F, Chakouri N, Hédon C, Granier M, Launay P, Thireau J, Richard S, Demion M (2017) The TRPM4 channel is functionally important for the beneficial cardiac remodeling induced by endurance training. J Muscle Res Cell Motil 38:3–16
Guinamard R, Rahmati M, Lenfant J, Bois P (2002) Characterization of a Ca2+-activated nonselective cation channel during dedifferentiation of cultured rat ventricular cardiomyocytes. J Membr Biol 188:127–135
Guinamard R, Chatelier A, Demion M, Potreau D, Patri S, Rahmati M, Bois P (2004) Functional characterization of a Ca(2+)-activated non-selective cation channel in human atrial cardiomyocytes. J Physiol 558:75–83
Guinamard R, Sallé L, Simard C (2011) The non-selective monovalent cationic channels TRPM4 and TRPM5. Adv Exp Med Biol 704:147–171
Guinamard R, Hof T, Del Negro CA (2014) The TRPM4 channel inhibitor 9-phenanthrol. Br J Pharmacol 171:1600–1613
Guinamard R, Bouvagnet P, Hof T, Liu H, Simard C, Sallé L (2015) TRPM4 in cardiac electrical activity. Cardiovasc Res 108:21–30
Harada M, Luo X, Qi XY, Tadevosyan A, Maguy A, Ordog B, Ledoux J, Kato T, Naud P, Voigt N, Shi Y, Kamiya K, Murohara T, Kodama I, Tardif JC, Schotten U, Van Wagoner DR, Dobrev D, Nattel S (2012) Transient receptor potential canonical-3 channel-dependent fibroblast regulation in atrial fibrillation. Circulation 126:2051–2064
Hartmann AK, Aranda Lopez P, Zajac M, Freichel M, Schild H, Radsak MP, Stassen M (2017) 9-Phenanthrol enhances the generation of an CD8+ T cell response following transcutaneous immunization with imiquimod in mice. J Dermatol Sci 87:260–267
Hof T, Simard C, Rouet R, Sallé L, Guinamard R (2013) Implication of the TRPM4 non-selective cation channel in mammalian sinus rhythm. Heart Rhythm 10:1683–1689
Hof T, Sallé L, Coulbault L, Richer R, Alexandre J, Rouet R, Manrique A, Guinamard R (2016) TRPM4 non-selective cation channels influence action potentials in rabbit Purkinje fibres. J Physiol 594:295–306
Inoue R, Kurahara LH, Hiraishi K (2019) TRP channels in cardiac and intestinal fibrosis. Semin Cell Dev Biol 94:40–49
Kecskés M, Jacobs G, Kerselaers S, Syam N, Menigoz A, Vangheluwe P, Freichel M, Flockerzi V, Voets T, Vennekens R (2015) The Ca(2+)-activated cation channel TRPM4 is a negative regulator of angiotensin II-induced cardiac hypertrophy. Basic Res Cardiol 110:43
Kim BJ, Kim SY, Lee S, Jeon JH, Matsui H, Kwon YK, Kim SJ, So I (2012) The role of transient receptor potential channel blockers in human gastric cancer cell viability. Canadian J Physiol Pharmacol 90:175–186
Klesen A, Jakob D, Emig R, Kohl P, Ravens U, Peyronnet R (2018) Cardiac fibroblasts: active players in (atrial) electrophysiology? Herzschrittmachertherapie Elektrophysiol 29:62–69
Koivumäki JT, Clark RB, Belke D, Kondo C, Fedak PW, Maleckar MM, Giles WR (2014) Na(+) current expression in human atrial myofibroblasts: identity and functional roles. Front Physiol 5:275
Kovacic JC, Dimmeler S, Harvey RP, Finkel T, Aikawa E, Krenning G, Baker AH (2019) Endothelial to mesenchymal transition in cardiovascular disease: JACC state-of-the-art review. J Am Coll Cardiol 73:190–209
Kruse M, Pongs O (2014) TRPM4 channels in the cardiovascular system. Curr Opin Pharmacol 15:68–73
Launay P, Fleig A, Perraud AL, Scharenberg AM, Penner R, Kinet JP (2002) TRPM4 is a Ca2+-activated nonselective cation channel mediating cell membrane depolarization. Cell 109:397–407
Loh KP, Ng G, Yu CY, Fhu CK, Yu D, Vennekens R, Nilius B, Soong TW, Liao P (2014) TRPM4 inhibition promotes angiogenesis after ischemic stroke. Pflugers Arch 466:563–576
Louault C, Benamer N, Faivre JF, Potreau D, Bescond J (2008) Implication of connexins 40 and 43 in functional coupling between mouse cardiac fibroblasts in primary culture. Biochim Biophys Acta 1778:2097–2104
Mathar I, Jacobs G, Kecskes M, Menigoz A, Philippaert K, Vennekens R (2014a) Trpm4. Handb Exp Pharmacol 222:461–487
Mathar I, Kecskes M, Van der Mieren G, Jacobs G, Camacho Londoño JE, Uhl S, Flockerzi V, Voets T, Freichel M, Nilius B, Herijgers P, Vennekens R (2014b) Increased β-adrenergic inotropy in ventricular myocardium from Trpm4-/- mice. Circ Res 114:283–294
Meszaros JG, Gonzalez AM, Endo-Mochizuki Y, Villegas S, Villarreal F, Brunton LL (2000) Identification of G protein-coupled signaling pathways in cardiac fibroblasts: cross talk between G(q) and G(s). Am J Physiol Cell Physiol 278:C154–C162
Monteilh-Zoller MK, Hermosura MC, Nadler MJ, Scharenberg AM, Penner R, Fleig A (2003) TRPM7 provides an ion channel mechanism for cellular entry of trace metal ions. J Gen Physiol 121:49–60
Nakajima H, Nakajima HO, Salcher O, Dittiè AS, Dembowsky K, Jing S, Field LJ (2000) Atrial but not ventricular fibrosis in mice expressing a mutant transforming growth factor-beta(1) transgene in the heart. Circ Res 86:571–579
Nelson P, Ngoc Tran TD, Zhang H, Zolochevska O, Figueiredo M, Feng JM, Gutierrez DL, Xiao R, Yao S, Penn A, Yang LJ, Cheng H (2013) Transient receptor potential melastatin 4 channel controls calcium signals and dental follicle stem cell differentiation. Stem Cells 31:167–177
Nilius B, Prenen J, Droogmans G, Voets T, Vennekens R, Freichel M, Wissenbach U, Flockerzi V (2003) Voltage dependence of the Ca2+-activated cation channel TRPM4. J Biol Chem 278:30813–30820
Pérez CA, Huang L, Rong M, Kozak JA, Preuss AK, Zhang H, Max M, Margolskee RF (2002) A transient receptor potential channel expressed in taste receptor cells. Nat Neurosci 5:1169–1176
Poulet C, Künzel S, Büttner E, Lindner D, Westermann D, Ravens U (2016) Altered physiological functions and ion currents in atrial fibroblasts from patients with chronic atrial fibrillation. Physiol Rep 4:e12681
Rose RA, Hatano N, Ohya S, Imaizumi Y, Giles WR (2007) C-type natriuretic peptide activates a non-selective cation current in acutely isolated rat cardiac fibroblasts via natriuretic peptide C receptor-mediated signalling. J Physiol 580:255–274
Sagredo AI, Sagredo EA, Pola V, Echeverría C, Andaur R, Michea L, Stutzin A, Simon F, Marcelain K, Armisén R (2019) TRPM4 channel is involved in regulating epithelial to mesenchymal transition, migration, and invasion of prostate cancer cell lines. J Cell Physiol 234:2037–2050
Schmitz C, Perraud AL, Johnson CO, Inabe K, Smith MK, Penner R, Kurosaki T, Fleig A, Scharenberg AM (2003) TRPM7 provides an ion channel mechanism for cellular entry of trace metal ions. Cell 114:191–200
Simard C, Hof T, Keddache Z, Launay P, Guinamard R (2013) The TRPM4 non-selective cation channel contributes to the mammalian atrial action potential. J Mol Cell Cardiol 59:11–19
Veress R, Baranyai D, Hegyi B, Kistamás K, Dienes C, Magyar J, Bányász P, Nánási PP, Szentandrássy N, Horváth B (2018) Transient receptor potential melastatin 4 channel inhibitor 9-phenanthrol inhibits K+ but not Ca2+ currents in canine ventricular myocytes. Can J Physiol Pharmacol 96:1022–1029
Wang J, Takahashi K, Piao H, Qu P, Naruse K (2013) 9-Phenanthrol, a TRPM4 inhibitor, protects isolated rat hearts from ischemia-reperfusion injury. PLoS One 8:e70587
Yu Y, Chen S, Xiao C, Jia Y, Guo J, Jiang J, Liu P (2014) TRPM7 is involved in angiotensin II induced cardiac fibrosis development by mediating calcium and magnesium influx. Cell Calcium 55:252–260
Yu CX, Zhang YY, Wu XY, Tang HX, Liang XQ, Xue ZM, Xue YD, Li J, Zhu H, Huo R, Ban T (2019) Transient receptor potential melastatin 4 contributes to early-stage endothelial injury induced by arsenic trioxide. Toxicol Lett 312:98–108
Yue Z, Zhang Y, Xie J, Jiang J, Yue L (2013) Transient receptor potential (TRP) channels and cardiac fibrosis. Curr Top Med Chem 13:270–282
Zhang X, Zhang T, Wu J, Yu X, Zheng D, Yang F, Li T, Wang L, Zhao Y, Dong S, Zhong X, Fu S, Xu CQ, Lu F, Zhang WH (2014) Calcium sensing receptor promotes cardiac fibroblast proliferation and extracellular matrix secretion. Cell Physiol Biochem 33:557–568
Zou ZG, Rios FJ, Montezano AC, Touyz RM (2019) TRPM7, magnesium, and signaling. Int J Mol Sci 20:1877
Acknowledgments
The authors thank Marie Demion (INSERM U1046, Montpellier, France) for providing the mouse strain used in this study and Claudine Combes for technical assistance in human fibroblast isolation experiments. They also thank Vladimir Saplacan, Dimitrios Buklas, Julien Desgue, and Gerard Babatasi, cardiac surgeons, CHU Caen.
Funding
This work was done with a financial support (RIN ENTRAC) of « Région Normandie », France. Christophe Simard received a fellowship from “Région Normandie,” France. This work was conducted as part of the FHU REMOD-VHF project.
Author information
Authors and Affiliations
Contributions
C. Simard, C. Magaud, and R. Adjlane were in charge of cell isolation and culture. C. Simard and R. Guinamard performed the single-channel patch-clamp recordings. C. Simard, C. Magaud, and Q. Dupas were in charge of the biochemical approaches. L. Sallé, A. Manrique, and P. Bois participated in data analysis and editing of the paper. C. Simard, C Magaud, JF. Faivre, and R. Guinamard were in charge of the design of the experiments and writing of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Studies using human atrial appendages were made after the approval of local medical ethics committee (Comité de Protection des Personnes Nord Ouest III, Caen, France, ref.: DC-2013-1967) and patient written informed consent.
Experiments using mouse atrial cells were carried out in strict accordance with the European Commission Directive 2010/63/EU for animal care. The study was also conducted with authorization for animal experimentation #14-98 from the local DDPP (Direction Départementale de la Protection des Populations).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Figure 1.
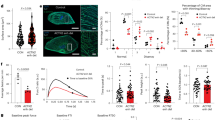
Expression and localization of smooth muscle actin (α-SMA), CD31 and vimentin in primary culture of human cardiac fibroblasts. A. Expression of CD31 in human umbilical vein endothelial cell line (HUVEC) by immunolabeling. Left panel: omission of the primary antibody; Right panel: detection of CD31 in HUVEC cells. Cells were incubated for 2 h with the secondary antibody (1:250, chicken anti-mouse alexa 488, Thermo Fisher Scientific). B. Immunostaining experiments performed on primary culture of human atrial fibroblasts after 12 days in culture to characterize expression of α-SMA, CD31 (green channels) and vimentin (red channel). Upper panel: example of double immunofluorescence staining of α-SMA and vimentin in primary culture of human cardiac fibroblast. Lower panel: Example of double immunofluorescence staining of CD31 and vimentin in primary culture of human cardiac fibroblast. Note the absence of expression of the endothelial cell marker CD31. DAPI staining (blue) was used to mark the nuclei. Images were obtained using confocal microscopy; Bar = 100 μm. (PNG 193201 kb)
Rights and permissions
About this article
Cite this article
Simard, C., Magaud, C., Adjlane, R. et al. TRPM4 non-selective cation channel in human atrial fibroblast growth. Pflugers Arch - Eur J Physiol 472, 1719–1732 (2020). https://doi.org/10.1007/s00424-020-02476-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-020-02476-0