Abstract
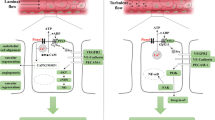
The calcium-sensing receptor (CaR) is widely expressed throughout the entire cardiovascular system and is capable of activating signaling pathways in different cells. Alongside calcium, the CaR also responds to physiological polycations such as putrescine underlining a participation in physiological and pathophysiological processes. Here, we aimed to determine mechanisms as to how CaR activation affects the contractile responsiveness of ventricular cardiomyocytes under basal and stimulated conditions. For that purpose, cardiac myocytes from 3-month-old male Wistar rats were isolated, and the acute effects of an antagonist (NPS2390), agonists (putrescine and gadolinium), or of downregulation of the CaR by siRNA on cell shortening were recorded in a cell-edge-detection system. In addition, experiments were performed on muscle stripes and Langendorff preparations. Mechanistic insights were taken from calcium transients of beating fura-2 AM-loaded cardiomyocytes and western blots. Isolated ventricular cardiomyocytes constitutively express CaR. The expression in the atria is less pronounced. Acute inhibition of CaR reduced basal cell shortening of ventricular myocytes at nearly physiological levels of extracellular calcium. Inhibition of CaR strongly reduced contractility of ventricular muscle stripes but not of atria. Activation of CaR by putrescine and gadolinium influences the contractile responsiveness of isolated cardiomyocytes. Increased calcium mobilization from the sarcoplasmic reticulum via an IP3-dependent mechanism was responsible for amplified systolic calcium transients and a subsequent improvement in cell shortening. Alongside with these effects, activation of CaR increased relaxation velocity of the cells. In conclusion, ventricular CaR expression affects contractile parameters of ventricular heart muscle cells and modifies electromechanical coupling of cardiomyocytes.







Similar content being viewed by others
References
Anwar A, Schluter KD, Heger J, Piper HM, Euler G (2008) Enhanced SERCA2a expression improves contractile performance of ventricular cardiomyocytes of rat under adrenergic stimulation. Pflugers Arch – Eur J Physiol 457:485–491
Barrabés JA, Garcia-Dorado D, Agulló L, Rodríguez-Sinovas A, Padilla F, Trobo L, Soler-Soler J (2006) Intracoronary infusion of Gd3+ into ischemic region does not suppress phase Ib ventricular arrhythmias after coronary occlusion in swine. Am J Physiol Heart Circ Physiol 290:H2344–H2350
Bergeron RJ, Wiegand J, Weimar WR, Snyder PS (1998) Polyamine analogue antiarrhythmics. Pharmacol Res 38:367–380
Brown EM, Gamba G, Riccardi D, Lombardi M, Butters R, Kifor O, Sun A, Hediger MA, Lytton J, Herbert SC (1993) Cloning and characterization of an extracellular Ca(2+)-sensing receptor from bovine parathyroid. Nature 366(6455):575–580
Canaff L, Petit JL, Kisiel M, Watson PH, Gascon-Barré M, Hendy GN (2001) Extracellular calcium-sensing receptor is expressed in rat hepatocytes: coupling to intracellular calcium mobilization and stimulation of bile flow. J Biol Chem 276:4070–4079
Colyer J (1998) Phosphorylation states of phospholamban. Ann N Y Acad Sci 853:79–91
Garrett JE, Tamir H, Kifor O, Simin RT, Rogers KV, Mithal A, Gagel RF, Brown EM (1995) Calcitonin-secreting cells of the thyroid express an extracellular calcium receptor gene. Endocrinology 136:5202–5211
Geibel J, Sritharan K, Geibel R, Geibel P, Persing JS, Seeger A, Roepke TK, Deichstetter M, Prinz C, Cheng SX, Martin D, Herbert SC (2006) Calcium-sensing receptor abrogates secretagogue- induced increases in intestinal net fluid secretion by enhancing cyclic nucleotide destruction. Proc Natl Acad Sci U S A 103:9390–9397
Grohè C, van Eickels M, Wenzel S, Meyer R, Degenhardt H, Doevendans PA, Heinemann MP, Ross G, Schlüter K-D (2004) Sex-specific differences in ventricular expression and function of parathyroid hormone-related peptide. Cardiovasc Res 61:307–316
Harris P (1982) Polyamine metabolism in myocardial hypertrophy. Eur Heart J Suppl A 3:73–74
Huang C, Handlogten ME, Miller RT (2002) Parallel activation of phosphatidylinositol 4-kinase and phospholipase C by the extracellular calcium-sensing receptor. J Biol Chem 277:20293–20300
Ladilov Y, Efe O, Schäfer C, Rother B, Kasseckert S, Abdallah Y, Meuter K, Schlüter K-D, Piper HM (2003) Reoxygenation-induced rigor-type contracture. J Mol Cell Cardiol 35:1481–1490
Langer M, Luttecke D, Schluter KD (2003) Mechanism of the positive contractile effect of nitric oxide on rat ventricular cardiomyocytes with positive force/frequency relationship. Pflugers Arch-Eur J Physiol 447:289–297
McConkey DJ, Orrenius S (1997) The role of calcium in the regulation of apoptosis. Biochem Biophys Res Commun 239:357–366
Molostvov G, James S, Fletcher S, Bennett J, Lehnert H, Bland R, Zehnder D (2007) Extracellular calcium-sensing receptor is functionally expressed in human artery. Am J Physiol Ren Physiol 293:F946–F955
Nemeth EF, Delmar EG, Heaton WL, Miller MA, Lambert LD, Conklin RL, Gowen M, Gleason JG, Bhatnagar PK, Fox J (2001) Calcilytic compounds: potent and selective Ca2+ receptor antagonists that stimulate secretion of parathyroid hormone. J Pharmacol Exp Ther 299:323–331
Piper HM, Millar BC, McDermott BJ (1989) The negative inotropic effect of neuropeptide Y on the ventricular cardiomyocyte. Naunyn-Schmiedeberg’s Arch Pharmacol 340:333–337
Purroy J, Spurr NK (2002) Molecular genetics of calcium sensing in bone cells. Hum Mol Genet 11:2377–2384
Quinn SJ, Ye CP, Diaz R, Kifor O, Bai M, Vassilev P, Brown E (1997) The Ca2+-sensing receptor: a target for polyamines. Am J Physiol 273:C1315–C1323
Schlüter K-D, Piper HM (2005) Isolation and culture of adult ventricular cardiomyocytes. In: Dhein S et al (eds) Practical methods in cardiovascular research. Springer, Berlin-Heidelberg, pp 557–567
Schlüter K, Katzer C, Frischkopf K, Wenzel S, Taimor G, Piper HM (2000) Expression, release, and biological activity of parathyroid hormone-related peptide from coronary endothelial cells. Circ Res 86:946–951
Smajilovic S, Tfelt-Hansen J (2008) Novel role of the calcium-sensing receptor in blood pressure modulation. Hypertension 52:994–1000
Tagliavini S, Genedani S, Bertolini A, Bazzani C (1991) Ischemia- and reperfusion-induced arrhythmias are prevented by putrescine. Eur J Pharmacol 194:7–10
Tfelt-Hansen J, Hansen JL, Smajilovic S, Terwilliger EF, Haunso S, Sheikh SP (2006) Calcium receptor is functionally expressed in rat neonatal ventricular cardiomyocytes. Am J Physiol Heart Circ Physiol 290:H1165–H1171
Wang JW, Yazawa K, Hao LY, Onoue Y, Kameyama M (2007) Verrucotoxin inhibits KATP channels in cardiac myocytes through a muscarinic M3 receptor-PKC pathway. Eur J Pharmacol 563:172–179
Wang M, Yao Y, Kuang D, Hampson DR (2006) Activation of family C G-protein-coupled receptors by the tripeptide glutathione. J Biol Chem 281:8864–8870
Wang R, Xu C, Zhao W, Zhang J, Cao K, Yang B, Wu L (2003) Calcium and polyamine regulated calcium-sensing receptors in cardiac tissues. Eur J Biochem 270:2680–2688
Wang Y, Bukoski RD (1998) Distribution of the perivascular nerve Ca2+ receptor in rat arteries. Br J Pharmacol 125:1397–1404
Wang YG, Dedkova EN, Ji X, Blatter LA, Lipsius SL (2005) Phenylephrine acts via IP3-dependent intracellular NO release to stimulate L-type Ca2+ current in cat atrial myocytes. J Physiol 567(Pt 1):143–157
Ward DT, Riccardi D (2002) Renal physiology of the extracellular calcium-sensing receptor. Pflugers Arch 445:169–176
Weston AH, Absi M, Ward DT, Ohanian J, Dodd RH, Dauban P, Petrel C, Ruat M, Edwards G (2005) Evidence in favor of a calcium-sensing receptor in arterial endothelial cells: studies with calindol and Calhex 231. Circ Res 97:391–398
Wilkins BJ, Molkentin JD (2004) Calcium-calcineurin signaling in the regulation of cardiac hypertrophy. Biochem Biophys Res Commun 322:1178–1191
Zayzafoon M (2006) Calcium/calmodulin signaling controls osteoblast growth and differentiation. J Cell Biochem 97:56–70
Zhang WH, Fu SB, Lu FH, Wu B, Gong DM, Pan ZW, Lv YJ, Zhao YJ, Li QF, Wang R, Yang BF, Xu CQ (2006) Involvement of calcium-sensing receptor in ischemia/reperfusion-induced apoptosis in rat cardiomyocytes. Biochem Biophys Res Commun 347:872–881
Zhao YJ, Xu CQ, Zhang WH, Zhang L, Bian SL, Huang Q, Sun HL, Li QF, Zhang YQ, Tian Y, Wang R, Yang BF, Li WM (2007) Role of polyamines in myocardial ischemia/reperfusion injury and their interactions with nitric oxide. Eur J Pharmacol 562:236–246
Zhao YJ, Zhang WH, Xu CQ, Li HZ, Wang LN, Li H, Sun YH, Lin Y, Han LP, Zhang L, Tian Y, Wang R, Yang BF, Li WM (2009) Involvement of the ornithine decarboxylase/polyamine system in precondition-induced cardioprotection through an interaction with PKC in rat hearts. Mol Cell Biochem 332:135–144
Ziegelstein RC, Xiong Y, He C, Hu Q (2006) Expression of a functional extracellular calcium-sensing receptor in human aortic endothelial cells. Biochem Biophys Res Commun 342:153–163
Acknowledgments
The authors thank Nadine Woitasky, Peter Volk, and Daniela Schreiber for excellent technical support. The data of this study are in part results of the thesis of Elena Dyukova.
Funding
This study was supported by the DAAD (cooperation program of the universities of Giessen, Germany and Kazan, Russia).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
(PPTX 141 kb)
Supplementary Fig. 2
(PPTX 99 kb)
Supplementary Fig. 3
(PPTX 70 kb)
Supplementary Fig. 4
(PPTX 68 kb)
Supplementary Fig. 5
(PPTX 113 kb)
Rights and permissions
About this article
Cite this article
Schreckenberg, R., Dyukova, E., Sitdikova, G. et al. Mechanisms by which calcium receptor stimulation modifies electromechanical coupling in isolated ventricular cardiomyocytes. Pflugers Arch - Eur J Physiol 467, 379–388 (2015). https://doi.org/10.1007/s00424-014-1498-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-014-1498-y