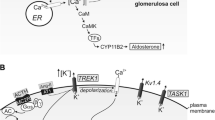
Abstract
Aldosterone, secreted by adrenal glomerulosa cells, allows the adaptation of the vertebrate organism to a wide range of physiological and pathological stimuli including acute haemodynamic challenges and long-term changes in dietary sodium and potassium intake. Most of the extracellular signals are mediated by cytosolic Ca2+ signal deriving from Ca2+ release, store-operated and/or voltage-gated Ca2+ influx. Mitochondria in glomerulosa cells play a fundamental role in generating and modulating the final biological response. These organelles not only house several enzymes of aldosterone biosynthesis but also—in a Ca2+-dependent manner—provide NADPH for the function of these enzymes. Moreover, mitochondria, constituting a high portion of cytoplasmic volume and displaying a uniquely low-threshold Ca2+ sequestering ability, shape and thus modulate the decoding of the complex cytosolic Ca2+ response. The unusual features of mitochondrial Ca2+ signalling that permit such an integrative function in adrenal glomerulosa cells are hereby described.




Similar content being viewed by others
References
Bartter FC, Barbour BH, Carr AA, Delea CS (1964) On the role of potassium and of the central nervous system in the regulation of aldosterone secretion. In: Baulieu EE, Robel P (eds) Aldosterone. Blackwell, Oxford, pp 221–242
Baughman JM, Perocchi F, Girgis HS et al (2011) Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476:341–345
Berridge MJ (1990) Calcium oscillations. J Biol Chem 265:9583–9586
Berridge MJ (1993) Inositol trisphosphate and calcium signalling. Nature 361:315–325
Berridge MJ, Lipp P, Bootman MD (2000) The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol 1:11–21
Bezprozvanny I, Watras J, Ehrlich BE (1991) Bell-shaped calcium-response curves of Ins(1,4,5)P3− and calcium-gated channels from endoplasmic reticulum of cerebellum. Nature 351:751–754
Boyd JE, Palmore WP, Mulrow PJ (1971) Role of potassium in the control of aldosterone secretion in the rat. Endocrinology 88:556–565
Brandenburger Y, Kennedy ED, Python CP et al (1996) Possible role for mitochondrial calcium in angiotensin II- and potassium-stimulated steroidogenesis in bovine adrenal glomerulosa cells. Endocrinology 137:5544–5551
Camello-Almaraz C, Gomez-Pinilla PJ, Pozo MJ, Camello PJ (2006) Mitochondrial reactive oxygen species and Ca2+ signaling. Am J Physiol Cell Physiol 291:C1082–C1088
Camello-Almaraz C, Salido GM, Pariente JA, Camello PJ (2002) Role of mitochondria in Ca2+ oscillations and shape of Ca2+ signals in pancreatic acinar cells. Biochem Pharmacol 63:283–292
Challet C, Maechler P, Wollheim CB, Ruegg UT (2001) Mitochondrial calcium oscillations in C2C12 myotubes. J Biol Chem 276:3791–3797
Collins TJ, Lipp P, Berridge MJ, Bootman MD (2001) Mitochondrial Ca2+ uptake depends on the spatial and temporal profile of cytosolic Ca2+ signals. J Biol Chem 276:26411–26420
Crompton M, Moser R, Ludi H, Carafoli E (1978) The interrelations between the transport of sodium and calcium in mitochondria of various mammalian tissues. Eur J Biochem 82:25–31
Csordás G, Thomas AP, Hajnóczky G (1999) Quasi-synaptic calcium signal transmission between endoplasmic reticulum and mitochondria. EMBO J 18:96–108
Csordás G, Várnai G et al (2010) Imaging interorganelle contacts and local calcium dynamics at the ER-mitochondrial interface. Mol Cell 39:121–132
Czirják G, Enyedi P (2002) TASK-3 dominates the background potassium conductance in rat adrenal glomerulosa cells. Mol Endocrinol 16:621–629
Czirják G, Fischer T, Spät A, Lesage F, Enyedi P (2000) TASK (TWIK-related acid-sensitive K+ channel) is expressed in glomerulosa cells of rat adrenal cortex and inhibited by angiotensin II. Mol Endocrinol 14:863–874
De Stefani D, Raffaello A, Teardo E, Szabo I, Rizzuto R (2011) A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature 476:336–340
Di Capite J, Ng SW, Parekh AB (2009) Decoding of cytoplasmic Ca2+ oscillations through the spatial signature drives gene expression. Curr Biol 19:853–858
Dluhy RG, Axelrod L, Underwood RH, Williams GH (1972) Studies of the control of plasma aldosterone concentration in normal man. II. Effect of dietary potassium and acute potassium infusion. J Clin Invest 51:1950–1957
Duchen MR (2004) Mitochondria in health and disease: perspectives on a new mitochondrial biology. Mol Aspects Med 25:365–451
Dupont G, Combettes L, Bird GS, Putney JW (2011) Calcium oscillations. Cold Spring Harb Perspect Biol 3:pii: a004226
Enyedi P, Szabadkai G, Horváth A, Szilágyi GL, Spät A (1994) Inositol 1,4,5-trisphosphate receptor subtypes in adrenal glomerulosa cells. Endocrinology 134:2354–2359
Foreman MA, Smith J, Publicover SJ (2006) Characterisation of serum-induced intracellular Ca2+ oscillations in primary bone marrow stromal cells. J Cell Physiol 206:664–671
Foskett JK, White C, Cheung KH, Mak DO (2007) Inositol trisphosphate receptor Ca2+ release channels. Physiol Rev 87:593–658
Fülöp L, Szanda G, Enyedi B, Várnai P, Spät A (2011) The effect of OPA1 on mitochondrial Ca2+ signaling. PLoS One 6:e25199
Giacomello M, Drago I, Bortolozzi M et al (2010) Ca2+ hot spots on the mitochondrial surface are generated by Ca2+ mobilization from stores, but not by activation of store-operated Ca2+ channels. Mol Cell 38:280–290
Gunter TE, Pfeiffer DR (1990) Mechanisms by which mitochondria transport calcium. Am J Physiol 258:C755–C786
Hajnóczky G, Csordás G, Hunyady L et al (1992) Angiotensin-II inhibits Na+/K+ pump in rat adrenal glomerulosa cells: possible contribution to stimulation of aldosterone production. Endocrinology 130:1637–1644
Hajnóczky G, Robb-Gaspers LD, Seitz MB, Thomas AP (1995) Decoding of cytosolic calcium oscillations in the mitochondria. Cell 82:415–424
Hajnóczky G, Thomas AP (1997) Minimal requirements for calcium oscillations driven by the IP3 receptor. EMBO J 16:3533–3543
Hattori M, Suzuki AZ, Higo T et al (2004) Distinct roles of inositol 1,4,5-trisphosphate receptor types 1 and 3 in Ca2+ signaling. J Biol Chem 279:11967–11975
Iino M (1990) Biphasic Ca2+ dependence of inositol 1,4,5-trisphosphate- induced Ca release in smooth muscle cells of the guinea pig taenia caeci. J Gen Physiol 95:1103–1122
Ishii K, Hirose K, Iino M (2006) Ca2+ shuttling between endoplasmic reticulum and mitochondria underlying Ca2+ oscillations. EMBO Rep 7:390–396
Jiang D, Zhao L, Clapham DE (2009) Genome-wide RNAi screen identifies Letm1 as a mitochondrial Ca2+/H+ antiporter. Science 326:144–147
Kaftan EJ, Xu T, Abercrombie RF, Hille B (2000) Mitochondria shape hormonally induced cytoplasmic calcium oscillations and modulate exocytosis. J Biol Chem 275:25465–25470
Kirichok Y, Krapivinsky G, Clapham DE (2004) The mitochondrial calcium uniporter is a highly selective ion channel. Nature 427:360–364
Koncz P, Szanda G, Rajki A, Spät A (2006) Reactive oxygen species, Ca2+ signaling and mitochondrial NAD(P)H level in adrenal glomerulosa cells. Cell Calcium 40:347–357
Korzeniowski MK, Szanda G, Balla T, Spät A (2009) Store-operated Ca2+ influx and subplasmalemmal mitochondria. Cell Calcium 46:49–55
Lupo D, Vollmer C, Deckers M et al (2011) Mdm38 is a 14-3-3-like receptor and associates with the protein synthesis machinery at the inner mitochondrial membrane. Traffic 12:1457–1466
Maechler P, Kennedy ED, Wang HY, Wollheim CB (1998) Desensitization of mitochondrial Ca2+ and insulin secretion responses in the beta cell. J Biol Chem 273:20770–20778
Mammucari C, Patron M, Granatiero V, Rizzuto R (2011) Molecules and roles of mitochondrial calcium signaling. Biofactors 37:219–227
Marsault R, Murgia M, Pozzan T, Rizzuto R (1997) Domains of high Ca2+ beneath the plasma membrane of living A7r5 cells. EMBO J 16:1575–1581
McCormack JG, Halestrap AP, Denton RM (1990) Role of calcium ions in regulation of mammalian intramitochondrial metabolism. Physiol Rev 70:391–425
Meyer T, Stryer L (1988) Molecular model for receptor-stimulated calcium spiking. Proc Natl Acad Sci USA 85:5051–5055
Michels G, Khan IF, Endres-Becker J et al (2009) Regulation of the human cardiac mitochondrial Ca2+ uptake by 2 different voltage-gated Ca2+ channels. Circulation 119:2435–2443
Montero M, Alonso MT, Carnicero E et al (2000) Chromaffin-cell stimulation triggers fast millimolar mitochondrial Ca2+ transients that modulate secretion. Nat Cell Biol 2:57–61
Montero M, Lobaton CD, Moreno A, Alvarez J (2002) A novel regulatory mechanism of the mitochondrial Ca2+ uniporter revealed by the p38 mitogen-activated protein kinase inhibitor SB202190. FASEB J 16:1955–1957
Moreau B, Nelson C, Parekh AB (2006) Biphasic regulation of mitochondrial Ca2+ uptake by cytosolic Ca2+ concentration. Curr Biol 16:1672–1677
Moreau B, Parekh AB (2008) Ca2+-dependent inactivation of the mitochondria Ca2+ uniporter involves proton flux through the ATP synthase. Curr Biol 18:855–859
Nakazaki M, Ishihara H, Kakei M et al (1998) Repetitive mitochondrial Ca2+ signals synchronize with cytosolic Ca2+ oscillations in the pancreatic beta-cell line, MIN6. Diabetologia 41:279–286
Nowikovsky K, Froschauer EM, Zsurka G et al (2004) The LETM1/YOL027 gene family encodes a factor of the mitochondrial K+ homeostasis with a potential role in the Wolf-Hirschhorn syndrome. J Biol Chem 279:30307–30315
Nussdorfer GG (1980) Cytophysiology of the adrenal zona glomerulosa. Int Rev Cytol 64:307–368
Parekh AB (2008) Ca2+ microdomains near plasma membrane Ca2+ channels: impact on cell function. J Physiol 586:3043–3054
Perocchi F, Gohil VM, Girgis HS et al (2010) MICU1 encodes a mitochondrial EF hand protein required for Ca2+ uptake. Nature 467:291–296
Petersen OH, Wakui M (1990) Oscillating intracellular Ca2+ signals evoked by activation of receptors linked to inositol lipid hydrolysis: mechanism of generation. J Membr Biol 118:93–105
Pinton P, Leo S, Wieckowski MR, Di Benedetto G, Rizzuto R (2004) Long-term modulation of mitochondrial Ca2+ signals by protein kinase C isozymes. J Cell Biol 165:223–232
Pitter JG, Maechler P, Wollheim CB, Spät A (2002) Mitochondria respond to Ca2+ already in the submicromolar range: correlation with redox state. Cell Calcium 31:97–104
Pralong WF, Hunyady L, Várnai P, Wollheim CB, Spät A (1992) Pyridine nucleotide redox state parallels production of aldosterone in potassium-stimulated adrenal glomerulosa cells. Proc Natl Acad Sci USA 89:132–136
Pralong WF, Spät A, Wollheim CB (1994) Dynamic pacing of cell metabolism by intracellular Ca2+. J Biol Chem 269:27310–27314
Quinn SJ, Cornwall MC, Williams GH (1987) Electrical properties of isolated rat adrenal glomerulosa and fasciculata cells. Endocrinology 120:903–914
Rizzuto R, Pinton P, Carrington W et al (1998) Close contacts with the endoplasmic reticulum as determinants of mitochondrial Ca2+ responses. Science 280:1763–1766
Robb-Gaspers LD, Burnett P, Rutter GA, Denton RM, Rizzuto R, Thomas AP (1998) Integrating cytosolic calcium signals into mitochondrial metabolic responses. EMBO J 17:4987–5000
Rohács T, Bagó A, Deák F, Hunyady L, Spät A (1994) Capacitative Ca2+ influx in adrenal glomerulosa cells. Possible role in angiotensin II response. Am J Physiol Cell Physiol 267:C1246–C1252
Rohács T, Nagy G, Spät A (1997) Cytoplasmic Ca2+ signalling and reduction of mitochondrial pyridine nucleotides in adrenal glomerulosa cells in response to K+, angiotensin II and vasopressin. Biochem J 322:785–792
Rohács T, Tory K, Dobos A, Spät A (1997) Intracellular calcium release is more efficient than calcium influx in stimulating mitochondrial NAD(P)H formation in adrenal glomerulosa cells. Biochem J 328:525–528
Rydstrom J, da Cruz AT, Ernster L (1970) Factors governing the kinetics and steady state of the mitochondrial nicotinamide nucleotide transhydrogenase system. Eur J Biochem 17:56–62
Spät A, Bradford PG, McKinney JS, Rubin RP, Putney JW Jr (1986) A saturable receptor for 32P-inositol-1,4,5-triphosphate in hepatocytes and neutrophils. Nature 319:514–516
Spät A, Fabiato A, Rubin RP (1986) Binding of inositol trisphosphate by a liver microsomal fraction. Biochem J 233:929–932
Spät A, Fülöp L, Koncz P, Szanda G (2008) When is high-Ca2+ microdomain required for mitochondrial Ca2+ uptake? Acta Physiol (Oxf) 195:139–147
Spät A, Fülöp L, Szanda G (2012) The role of mitochondrial Ca2+ and NAD(P)H in the control of aldosterone secretion. Cell Calcium. doi:10.1016/j.ceca.2012.01.009
Spät A, Hunyady L (2004) Control of aldosterone secretion: a model for convergence in cellular signaling pathways. Physiol Rev 84:489–539
Spät A, Pitter JG (2004) The effect of cytoplasmic Ca2+ signal on the redox state of mitochondrial pyridine nucleotides. Molec cell Endocrin 215:115–118
Spät A, Szanda G, Csordás G, Hajnóczky G (2008) High- and low-calcium-dependent mechanisms of mitochondrial calcium signalling. Cell Calcium 44:51–63
Straub SV, Giovannucci DR, Yule DI (2000) Calcium wave propagation in pancreatic acinar cells: functional interaction of inositol 1,4,5-trisphosphate receptors, ryanodine receptors, and mitochondria. J Gen Physiol 116:547–559
Szanda G, Koncz P, Várnai P, Spät A (2006) Mitochondrial Ca2+ uptake with and without the formation of high-Ca2+ microdomains. Cell Calcium 40:527–538
Szanda G, Rajki A, Gallego-Sandin S, Garcia-Sancho J, Spät A (2009) Effect of cytosolic Mg2+ on mitochondrial Ca2+ signaling. Pflügers Archiv European J Physiol 457:941–954
Taylor CW, Laude AJ (2002) IP3 receptors and their regulation by calmodulin and cytosolic Ca2+. Cell Calcium 32:321–334
Taylor CW, Rahman T, Tovey SC, Dedos SG, Taylor EJ, Velamakanni S (2009) IP3 receptors: some lessons from DT40 cells. Immunol Rev 231:23–44
Várnai P, Petheö GL, Makara JK, Spät A (1998) Electrophysiological study on the high K+ sensitivity of rat glomerulosa cells. Pflügers Archiv European J Physiol 435:429–431
Vinson GP, Whitehouse B, Hinson J (1992) The adrenal cortex. Prentice Hall, Englewood Cliffs, pp 65–139, 07632
Waldeck-Weiermair M, Jean-Quartier C, Rost R et al (2011) Leucine zipper EF hand-containing transmembrane protein 1 (Letm1) and uncoupling proteins 2 and 3 (UCP2/3) contribute to two distinct mitochondrial Ca2+ uptake pathways. J Biol Chem 286:28444–28455
Walsh C, Barrow S, Voronina S, Chvanov M, Petersen OH, Tepikin A (2009) Modulation of calcium signalling by mitochondria. Biochim Biophys Acta 1787:1374–1382
Wiederkehr A, Szanda G, Akhmedov D et al (2011) Mitochondrial matrix calcium is an activating signal for hormone secretion. Cell Metab 13:601–611
Zhang S, Fritz N, Ibarra C, Uhlen P (2011) Inositol 1,4,5-trisphosphate receptor subtype-specific regulation of calcium oscillations. Neurochem Res 36:1175–1185
Zhou YD, Fang XF, Cui ZJ (2009) UVA-induced calcium oscillations in rat mast cells. Cell Calcium 45:18–28
Zimmermann B, Walz B (1999) The mechanism mediating regenerative intercellular Ca2+ waves in the blowfly salivary gland. EMBO J 18:3222–3231
Acknowledgements
This work was supported by grants from the Hungarian Scientific Research Fund (OTKA TS-049851, TS-040865 and NK-72661) and the Council for Medical Research (ETT 0007/2006 and 008–09).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published as part of the Special Issue on Cell-specific roles of mitochondrial Ca2+ handling
Rights and permissions
About this article
Cite this article
Spät, A., Szanda, G. Special features of mitochondrial Ca2+ signalling in adrenal glomerulosa cells. Pflugers Arch - Eur J Physiol 464, 43–50 (2012). https://doi.org/10.1007/s00424-012-1086-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-012-1086-y