Abstract
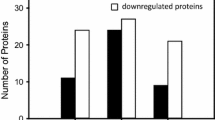
Myosin plays a key role in the structure and function of cardiac muscle. Three myosin isoenzymes (V1, V2, and V3) with different ATPase activities have been identified in mammalian ventricles based on their heavy chain constituents. The relative amount of myosin isoenzymes changes under physiological and pathological conditions. Until now, myosin isoenzymes have frequently been determined using either tube gel (nondenaturing) polyacrylamide gel electrophoresis (PAGE), or gradient or uniform sodium dodecyl sulfate (denaturing) PAGE. Both methods have disadvantages, e.g., a long running time. We developed, therefore, a uniform, nondenaturing PAGE with slab minigel format for analyzing the myosin isoenzymes in normoxic and stunned rabbit hearts. In normoxic hearts of adult rabbits, V3 predominated over V1 (46 vs 41%). In turn, in the stunned hearts, V1 predominated over V3 (70 vs 30%), and the heterodimeric V2 was not anymore detectable. This alteration appears to result from a selective loss of myosin heavy chain (MHC)-β. In parallel, the biochemical markers troponin I and creatine kinase were increased in the stunned hearts. We suggest that alterations of myosin isoenzymes in stunned myocardium can be monitored with native PAGE. The present analysis of myosin isoenzyme appears thus as a new tool for evaluating defects in MHC dimer formation in postischemic hearts.




Similar content being viewed by others
References
Alpert NA, Mulieri LA, Litten RZ (1979) Functional significance of altered myosin adenosine triphosphatase activity in enlarged hearts. Am J Cardiol 44:947–953
Bolli R (1990) Mechanism of myocardial “stunning”. Circulation 82:723–738
Burkhoff D (1990) Myocardial energetics and the postischemic heart. Ann Thorac Surg 49:525–527
Cummins P, Lambert SJ (1986) Myosin transitions in the bovine and human heart. A developmental and anatomical study of heavy and light chain subunits in the atrium and ventricle. Circ Res 58(6):846–858
Esser KA, Boluyt MO, White TP (1988) Separation of cardiac myosin heavy chain by gradient SDS-PAGE. Am J Physiol 255:659–663
Garcia Pomblum SC, Pomblum VJ, Gams E, Reiser PJ, Schipke JD (2003) Electrophoretic separation of ventricular myosin isoenzymes using a native polyacrylamide minigel system. Cell Biochem Biophys 38(1):33–40
Hein S, Scheffold T, Schaper J (1995) Ischemia induces early changes to cytoskeletal and contractile proteins in diseased human myocardium. J Thorac Cardiovasc Surg 110(1):89–98
Hoh JFY, McGrath PA, Hale PT (1978) Electrophoretic analysis of multiple forms of rat cardiac myosin: effects of hypophysectomy and thyroxine replacement. J Mol Cell Cardiol 10:1053–1076
Hoh JFY, Yeoh GPS, Thomas MAW, Higginbottom L (1979) Structural differences in the heavy chains of rat ventricular myosin isoenzymes. FEBS Lett 97(2):330–334
Holubarsch C, Goulette RP, Litten RZ, Martin BJ, Mulieri LA, Alpert NR (1985) The economy of isometric force development, myosin isoenzyme pattern and myofibrillar ATPase activity in normal and hypothyroid rat myocardium. Circ Res 56:78–86
Katz AM (1992) Physiology of the heart, 2nd edn. Raven, New York
Lamers JMJ (1999) Preconditioning and limitation of stunning: one step closer to the protected protein(s)? Cardiovasc Res 42:571–575
Litten RZ, Martin BJ, Buchthal RH, Nagai R, Low RB, Alpert NR (1985) Heterogeneity of myosin isozyme content of rabbit heart. Circ Res 57(3):406–414
Lompre AM, Schwartz K, d’albis A, Lacombe G, van Thiem N, Swynghedauw B (1979) Myosin isoenzyme redistribution in chronic heart overload. Nature 282(5734):105–107
Marban E, Kitakaze M, Chacko VP, Pike MM (1988) Ca2+ transients in perfused hearts revealed by gated 19F NMR spectroscopy. Circ Res 63:673–678
McDonough JL, Arrell DK, Van Eyk JE (1999) Troponin I degradation and covalent complex formation accompanies myocardial ischemia/reperfusion injury. Circ Res 84(1):9–20
McNally EM, Kraft R, Bravo-Zehnder M, Taylor A, Leinwand LA (1989) Full-length rat alpha and beta cardiac myosin heavy chain sequences. J Mol Biol 210:665–671
Morkin E (1993) Regulation of myosin heavy chain genes in the heart. Circulation 87(5):1451–1460
O’Neill L, Holbrook NJ, Fargnoli J, Lakatta EG (1991) Progressive changes from young adult age to senescence in mRNA for rat cardiac myosin heavy chain genes. Cardioscience 2(1):1–5
Pope B, Hoh JFY, Weeds A (1980) The ATPase activities of the rat cardiac myosin isoenzymes. FEBS Lett 118:205–208
Reiser PJ, Portman MA, Ning XH, Moravec CS (2001) Human cardiac myosin heavy chain isoforms in fetal and failing adult atria and ventricles. Am J Physiol 280(4):H1814–H1820
Reiser PJ, Kline WO (1998) Electrophoretic separation and quantitation of cardiac myosin heavy chain in eight mammalian species. Am J Physiol 274:H1048–H1053
Rupp H, Dietz K (1991) Mathematical models of myosin heterodimer formation in the rat heart during thyroid hormone alterations. Circ Res 68(1):27–37
Schipke JD, Sunderdiek U, Korbmacher B, Schwanke U, Arnold G (1995) Utilization of oxygen by the contractile apparatus is disturbed during reperfusion of post-ischemic myocardium. Eur Heart J 16:1476–1481
Schmitz-Spanke S, Seyfried E, Schwanke U, Korbmacher B, Sunderdiek U, Winter J et al (2002) The isolated rabbit heart: comparison between five different modifications. Herz 27(8):803–813
Schwartz K, Lecarpentier Y, Martin JL, Lompre AM, Mercadier JJ, Swynghedauw B (1981) Myosin isoenzymic distribution correlates with speed of myocardial contraction. J Mol Cell Cardiol 13(12):1071–1075
Swynghedauw B (1989) Remodelling of the heart in response to chronic mechanical overload. Eur Heart J 10:935–943
Takeda N, Nakamura I, Ohkubo T, Hatanaka T, Nagano M (1988) Effects of physical training on the myocardium of streptozotocin-induced diabetic rats. Basic Res Cardiol 83(5):525–530
Toyo-oka T, Ross J Jr (1981) Ca2+ sensitivity change and troponin loss in cardiac natural actomyosin after coronary occlusion. Am J Physiol 240(5):H704–H708
Tulp Ab, Verwood D, Neefjes J (1999) Electromigration for separations of proteins complexes. J Chromatogr B 722:141–152
Van Eyk JE, Murphy AM (2001) The role of troponin abnormalities as a cause for stunned myocardium. Coron Artery Dis 12(5):343–347
Wade ME, Herb RA, Powers SK, Criswell D (1999) Exercise and beta-adrenergic regulation of rat cardiac myosin isoforms. J Sports Med Phys Fitness 39:42–46
Westfall MV, Solaro RJ (1992) Alterations in myofibrillar function and protein profiles after complete global ischemia in rat hearts. Circ Res 70(2):302–313
Whalen RG, Schwartz K, Bouveret P, Sell SM, Gros F (1979) Contractile protein isozymes in muscle development: identification of an embryonic form of myosin heavy chain. Proc Natl Acad Sci USA 76(10):5197–5201
Yamashita H, Sugiura S, Serizawa T et al (1992) Sliding velocity of isolated rabbit cardiac myosin correlates with isozyme distribution. Am J Physiol 263:H464–H472
Acknowledgement
Reading and correcting of the manuscript by Dr. C. Heke is greatly appreciated. Mrs. S.C. Garcia was supported by a fellowship from the German Academic Exchange Service (DAAD) and had a lay-off of the Federal University of Santa Maria/Brazil. The study was supported in part by a grant from the German Research Foundation (DFG; Schi 201/13-1).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garcia, S.C., Pomblum, V.J., Gams, E. et al. Reduction in β-myosin heavy chains in stunned myocardium as assessed by nondenaturing gel electrophoresis. Pflugers Arch - Eur J Physiol 454, 937–943 (2007). https://doi.org/10.1007/s00424-007-0268-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-007-0268-5