Abstract
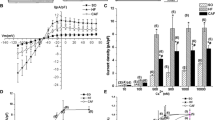
Sodium current INa plays an important role in the pacemaker activity of the sinoatrial node (SAN). However, expression profiles of corresponding sodium channel subunits in normal SAN remain unclear. And little is known about expression alteration of sodium channel in SAN under heart failure (HF) condition. We assessed SAN function and expression of Nav1.1, Nav1.2, Nav1.3, Nav1.5, Nav1.6, and Nav1.7 in sham-operated rats and rats subjected to abdominal arteriovenous shunt (volume overload)-induced HF. Immunohistochemistry, Western blot, and quantitative real-time reverse transcriptase PCR analysis were used to quantify sodium channel subunit protein and mRNA expression in the SAN. Intrinsic heart rate declined and sinus node recovery time was prolonged in HF rats, indicating suppressed SAN pacemaker function. In rat SAN, Nav1.1 and Nav1.6 were the primary subunits, Nav1.5 and Nav1.7 were weakly expressed, and Nav1.2 and Nav1.3 were not found to be present. HF significantly decreased SAN sodium channel expression at both the protein and mRNA levels (Nav1.1 by 61 and 71%, Nav1.6 by 49 and 46%, respectively). In conclusion, Nav1.1 and Nav1.6 are the dominant subunits in rat SAN, and downregulation of Nav1.1 and Nav1.6 expression contributes to HF-induced SAN dysfunction. These findings provide additional information about molecular basis of disease-related impairment of SAN function.







Similar content being viewed by others
References
Jose AD, Collison D (1970) The normal range and determinants of the intrinsic heart rate in man. Cardiovasc Res 4:160–167
Satoh H (2003) Sino-atrial nodal cells of mammalian hearts: ionic currents and gene expression of pacemaker ionic channels. J Smooth Muscle Res 39:175–193
Verkerk AO, Wilders R, Coronel R, Ravesloot JH, Verheijck EE (2003) Ionic remodeling of sinoatrial node cells by heart failure. Circulation 108:760–766
Zicha S, Fernandez-Velasco M, Lonardo G, L’Heureux N, Nattel S (2005) Sinus node dysfunction and hyperpolarization-activated (HCN) channel subunit remodeling in a canine heart failure model. Cardiovasc Res 66:472–481
Goldin AL (2001) Resurgence of sodium channel research. Annu Rev Physiol 63:871–894
Benson DW, Wang DW, Dyment M, Knilans TK, Fish FA, Strieper MJ, Rhodes TH, George AL Jr (2003) Congenital sick sinus syndrome caused by recessive mutations in the cardiac sodium channel gene (SCN5A). J Clin Invest 112:1019–1028
Maier SK, Westenbroek RE, Yamanushi TT, Dobrzynski H, Boyett MR, Catterall WA, Scheuer T (2003) An unexpected requirement for brain-type sodium channels for control of heart rate in the mouse sinoatrial node. Proc Natl Acad Sci USA 100:3507–3512
Lei M, Jones SA, Liu J, Lancaster MK, Fung SS, Dobrzynski H, Camelliti P, Maier SK, Noble D, Boyett MR (2004) Requirement of neuronal- and cardiac-type sodium channels for murine sinoatrial node pacemaking. J Physiol 559:835–848
Flaim SF, Minteer WJ, Zelis R (1980) Acute effects of arterio-venous shunt on cardiovascular hemodynamics in rat. Pflugers Arch 385:203–209
Grimm D, Holmer SR, Riegger GA, Kromer EP (1999) Effects of beta-receptor blockade and angiotensin II type I receptor antagonism in isoproterenol-induced heart failure in the rat. Cardiovasc Pathol 8:315–323
Mulder P, Barbier S, Chagraoui A, Richard V, Henry JP, Lallemand F, Renet S, Lerebours G, Mahlberg-Gaudin F, Thuillez C (2004) Long-term heart rate reduction induced by the selective I(f) current inhibitor ivabradine improves left ventricular function and intrinsic myocardial structure in congestive heart failure. Circulation 109:1674–1679
Dent MR, Singal T, Dhalla NS, Tappia PS (2004) Expression of phospholipase D isozymes in scar and viable tissue in congestive heart failure due to myocardial infarction. J Cell Mol Med 8:526–536
Elvan A, Wylie K, Zipes DP (1996) Pacing-induced chronic atrial fibrillation impairs sinus node function in dogs. Electrophysiological remodeling. Circulation 94:2953–2960
Dobrzynski H, Rothery SM, Marples DD, Coppen SR, Takagishi Y, Honjo H, Tamkun MM, Henderson Z, Kodama I, Severs NJ, Boyett MR (2000) Presence of the Kv1.5 K(+) channel in the sinoatrial node. J Histochem Cytochem 48:769–780
Musa H, Dobrzynski H, Berry Z, Abidi F, Cass CE, Young JD, Baldwin SA, Boyett MR (2002) Immunocytochemical demonstration of the equilibrative nucleoside transporter rENT1 in rat sinoatrial node. J Histochem Cytochem 50:305–309
Jacquet R, Hillyer J, Landis WJ (2005) Analysis of connective tissues by laser capture microdissection and reverse transcriptase-polymerase chain reaction. Anal Biochem 337:22–34
Antoni H, Bocker D, Eickhorn R (1988) Sodium current kinetics in intact rat papillary muscle: measurements with the loose-patch-clamp technique. J Physiol 406:199–213
ten Velde I, de Jonge B, Verheijck EE, van Kempen MJ, Analbers L, Gros D, Jongsma HJ (1995) Spatial distribution of connexin43, the major cardiac gap junction protein, visualizes the cellular network for impulse propagation from sinoatrial node to atrium. Circ Res 76:802–811
Boyett MR, Dobrzynski H, Lancaster MK, Jones SA, Honjo H, Kodama I (2003) Sophisticated architecture is required for the sinoatrial node to perform its normal pacemaker function. J Cardiovasc Electrophysiol 14:104–106
Kucera JP, Rohr S, Rudy Y (2002) Localization of sodium channels in intercalated disks modulates cardiac conduction. Circ Res 91:1176–1182
Lei M, Goddard C, Liu J, Leoni AL, Royer A, Fung SS, Xiao G, Ma A, Zhang H, Charpentier F, Vandenberg JI, Colledge WH, Grace AA, Huang CL (2005) Sinus node dysfunction following targeted disruption of the murine cardiac sodium channel gene Scn5a. J Physiol 567:387–400
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Dhar Malhotra J, Chen C, Rivolta I, Abriel H, Malhotra R, Mattei LN, Brosius FC, Kass RS, Isom LL (2001) Characterization of sodium channel alpha- and beta-subunits in rat and mouse cardiac myocytes. Circulation 103:1303–1310
Maier SK, Westenbroek RE, Schenkman KA, Feigl EO, Scheuer T, Catterall WA (2002) An unexpected role for brain-type sodium channels in coupling of cell surface depolarization to contraction in the heart. Proc Natl Acad Sci USA 99:4073–4078
Haufe V, Cordeiro JM, Zimmer T, Wu YS, Schiccitano S, Benndorf K, Dumaine R (2005) Contribution of neuronal sodium channels to the cardiac fast sodium current INa is greater in dog heart Purkinje fibers than in ventricles. Cardiovasc Res 65:117–127
Baruscotti M, Westenbroek R, Catterall WA, DiFrancesco D, Robinson RB (1997) The newborn rabbit sino-atrial node expresses a neuronal type I-like Na+ channel. J Physiol 498 (Pt 3):641–648
Rush AM, Craner MJ, Kageyama T, Dib-Hajj SD, Waxman SG, Ranscht B (2005) Contactin regulates the current density and axonal expression of tetrodotoxin-resistant but not tetrodotoxin-sensitive sodium channels in DRG neurons. Eur J Neurosci 22:39–49
Opthof T, Coronel R, Rademaker HM, Vermeulen JT, Wilms-Schopman FJ, Janse MJ (2000) Changes in sinus node function in a rabbit model of heart failure with ventricular arrhythmias and sudden death. Circulation 101:2975–2980
Sanders P, Kistler PM, Morton JB, Spence SJ, Kalman JM (2004) Remodeling of sinus node function in patients with congestive heart failure: reduction in sinus node reserve. Circulation 110:897–903
Tomaselli GF, Marban E (1999) Electrophysiological remodeling in hypertrophy and heart failure. Cardiovasc Res 42:270–283
Li D, Melnyk P, Feng J, Wang Z, Petrecca K, Shrier A, Nattel S (2000) Effects of experimental heart failure on atrial cellular and ionic electrophysiology. Circulation 101:2631–2638
Han W, Chartier D, Li D, Nattel S (2001) Ionic remodeling of cardiac Purkinje cells by congestive heart failure. Circulation 104:2095–2100
Acknowledgements
We would like to acknowledge Dr. Linlin Zhang for his help with the Western blot experiment. In addition, we are also grateful to Dr. Lisa Zhang for revising the English of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Du, Y., Huang, X., Wang, T. et al. Downregulation of neuronal sodium channel subunits Nav1.1 and Nav1.6 in the sinoatrial node from volume-overloaded heart failure rat. Pflugers Arch - Eur J Physiol 454, 451–459 (2007). https://doi.org/10.1007/s00424-007-0216-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-007-0216-4