Abstract
Purpose
Chyle leak resulting from thoracic duct (TD) injury poses significant morbidity and mortality challenges. We assessed the feasibility of using near-infrared (NIR) indocyanine green (ICG) imaging for intraoperative fluorescence TD lymphography during minimal access esophagectomy (MAE) in a semiprone position with inguinal nodal injection of ICG dye.
Methods
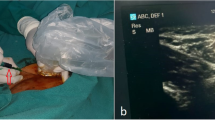
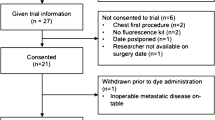
Ninety-nine patients with esophageal or gastroesophageal junctional cancer undergoing MAE received inguinal node injections of 2.5 mg ICG dye (total 5 mg) under sonographic guidance during anesthesia induction. Stryker’s 1688 AIM HD system was used in 76 cases, Karl Storz OPAL 1 S in 20, and in three cases the Karl Storz Rubina.
Results
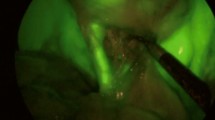
In 93 patients (94%), the TD was clearly delineated along its entire length; it was not visualized in 6 patients (6%). Fluorescence guidance facilitated TD ligation in 16 cases, while 3 cases required clipping of duct tributaries for oncological considerations. Twenty-eight patients exhibited minor duct variations. Fluorescence was sustained throughout surgery (median observation time 60 min post-injection; range 30–330). No patient experienced any chyle leak within 30 days post-surgery and no adverse reactions to ICG was evident.
Conclusions
Intraoperative fluorescence TD lymphography using ICG during MAE in a semiprone position with inguinal nodal injection proved safe, feasible, and effective, allowing clear visualization of the TD in almost all cases. This approach aids safe ligation and reduces chyle leak risk. It offers real-time imaging of TD anatomy and variations, providing valuable feedback to surgeons for managing TD injuries during MAE procedures and represents an excellent educational tool.




Similar content being viewed by others
Data Availability
The data is available with the corresponding author and can be shared upon a suitable request.
References
Rindani R, Martin CJ, Cox MR (1999) Transhiatal versus Ivor-Lewis oesophagectomy: is there a difference? Aust N Z J Surg 69(3):187–194. https://doi.org/10.1046/j.1440-1622.1999.01520.x
Hulscher JB, Tijssen JG, Obertop H, van Lanschot JJ (2001) Transthoracic versus transhiatal resection for carcinoma of the esophagus: a meta-analysis. Ann Thorac Surg 72(1):306–313. https://doi.org/10.1016/s0003-4975(00)02570-4
Schild HH, Strassburg CP, Welz A, Kalff J (2013) Treatment options in patients with chylothorax. Dtsch Arztebl Int 110(48):819–826. https://doi.org/10.3238/arztebl.2013.0819
Brinkmann S, Schroeder W, Junggeburth K, Gutschow CA, Bludau M, Hoelscher AH, Leers JM (2016) Incidence and management of chylothorax after Ivor Lewis esophagectomy for cancer of the esophagus. J Thorac Cardiovasc Surg 151(5):1398–1404. https://doi.org/10.1016/j.jtcvs.2016.01.030
Stange S, Sziklavari Z (2021) Modern treatment options for postoperative chylothorax: a systematic review. Pneumologie 75(6):439–446. https://doi.org/10.1055/a-1172-7288
Nusrath S, Thammineedi SR, Saksena AR, Patnaik SC, Reddy P, Usofi Z, Kumar S (2022) Thoracic duct lymphography by near-infrared indocyanine green fluorescence imaging in thoracic surgery. A review Indian J Surg Oncol 13(2):415–420. https://doi.org/10.1007/s13193-022-01493-y
Shen Y, Feng M, Khan MA, Wang H, Tan L, Wang Q (2014) A simple method minimizes chylothorax after minimally invasive esophagectomy. J Am Coll Surg 218(1):108–112. https://doi.org/10.1016/j.jamcollsurg.2013.09.014
Shackcloth MJ, Poullis M, Lu J, Page RD (2001) Preventing of chylothorax after oesophagectomy by routine pre-operative administration of oral cream. Eur J Cardiothorac Surg 20(5):1035–1036. https://doi.org/10.1016/s1010-7940(01)00928-9
Du ZS, Li XY, Luo HS, Wu SX, Zheng CP, Li ZY, Fu JH (2019) Preoperative administration of olive oil reduces chylothorax after minimally invasive esophagectomy. Ann Thorac Surg 107(5):1540–1543. https://doi.org/10.1016/j.athoracsur.2018.10.053
Thammineedi SR, Patnaik SC, Shuka S, Nusrath S (2021) Fluorescence-guided cancer surgery-a new paradigm. J Surg Oncol 123(8):1679–1698. https://doi.org/10.1002/jso.26469
Matsutani T, Hirakata A, Nomura T, Hagiwara N, Matsuda A, Yoshida H, Uchida E (2014) Transabdominal approach for chylorrhea after esophagectomy by using fluorescence navigation with indocyanine green. Case Rep Surg 2014:464017. https://doi.org/10.1155/2014/464017
Kamiya K, Unno N, Konno H (2009) Intraoperative indocyanine green fluorescence lymphography, a novel imaging technique to detect a chyle fistula after an esophagectomy: report of a case. Surg Today 39(5):421–424. https://doi.org/10.1007/s00595-008-3852-1
Chang TI, Chen YS, Huang SC (2014) Intraoperative indocyanine green fluorescence lymphography to detect chylous leakage sites after congenital heart surgery. J Thorac Cardiovasc Surg 148(2):739–740. https://doi.org/10.1016/j.jtcvs.2014.03.021
Kaburagi T, Takeuchi H, Oyama T, Nakamura R, Takahashi T, Wada N, Saikawa Y, Kamiya S, Tanaka M, Wada T, Kitagawa Y (2013) Intraoperative fluorescence lymphography using indocyanine green in a patient with chylothorax after esophagectomy: Report of a case. Surg Today 43(2):206–210. https://doi.org/10.1007/s00595-012-0391-6
Bassi M, Vannucci J, Venuta F, De Giacomo T (2020) Effectiveness of indocyanine green fluorescence for the identification of thoracic duct in recurrent idiopathic chylothorax. Interact Cardiovasc Thorac Surg 31(2):284. https://doi.org/10.1093/icvts/ivaa080
Vecchiato M, Martino A, Sponza M, Uzzau A, Ziccarelli A, Marchesi F, Petri R (2020) Thoracic duct identification with indocyanine green fluorescence during minimally invasive esophagectomy with patient in prone position. Dis Esophagus 33(12). https://doi.org/10.1093/dote/doaa030
Varshney VK, Nayar R, Soni SC, Selvakumar B, Garg PK, Varshney P, Khera PS (2022) Intra-nodal indocyanine green injection to delineate thoracic duct during minimally invasive esophagectomy. J Gastrointest Surg 26(8):1559–1565. https://doi.org/10.1007/s11605-022-05341-w
Benson RC, Kues HA (1978) Fluorescence properties of indocyanine green as related to angiography. Phys Med Biol 23(1):159–163. https://doi.org/10.1088/0031-9155/23/1/017
Thammineedi SR, Patnaik SC, Shuka S, Nusrath S (2022) Indocyanine green fluorescent thoracic duct lymphography by inguinal nodal injection approach for identifying thoracic duct and chyle leak: a case report. Indian J Surg. https://doi.org/10.1007/s12262-022-03619-6
Ashitate Y, Tanaka E, Stockdale A, Choi HS, Frangioni JV (2011) Near-infrared fluorescence imaging of thoracic duct anatomy and function in open surgery and video-assisted thoracic surgery. J Thorac Cardiovasc Surg 142(1):31-8.e1–2. https://doi.org/10.1016/j.jtcvs.2011.03.004
Steffey MA, Mayhew PD (2018) Use of direct near-infrared fluorescent lymphography for thoracoscopic thoracic duct identification in 15 dogs with chylothorax. Vet Surg 47(2):267–276. https://doi.org/10.1111/vsu.12740
Chakedis J, Shirley LA, Terando AM, Skoracki R, Phay JE (2018) Identification of the thoracic duct using indocyanine green during cervical lymphadenectomy. Ann Surg Oncol 25(12):3711–3717. https://doi.org/10.1245/s10434-018-6690-4
Barnes TG, MacGregor T, Sgromo B, Maynard ND, Gillies RS (2022) Near infra-red fluorescence identification of the thoracic duct to prevent chyle leaks during oesophagectomy. Surg Endosc 36(7):5319–5325. https://doi.org/10.1007/s00464-021-08912-1
Tokumaru S, Kitazawa M, Nakamura S, Koyama M, Soejima Y (2022) Intraoperative visualization of morphological patterns of the thoracic duct by subcutaneous inguinal injection of indocyanine green in esophagectomy for esophageal cancer. Ann Gastroenterol Surg 6(6):873–879. https://doi.org/10.1002/ags3.12594
Coratti F, Barbato G, Cianchi F (2021) Thoracic duct identification with indocyanine green fluorescence: a simplified method. Dis Esophagus 34(3). https://doi.org/10.1093/dote/doaa130 doaa130
Author information
Authors and Affiliations
Contributions
SR—acquisition of data, analysis and interpretation of data, drafting of manuscript, critical revision of manuscript. SP—acquisition of data, analysis and interpretation of data, drafting of manuscript, critical revision of manuscript. SS—study conception and design, drafting of manuscript, critical revision of manuscript. PR—drafting of manuscript, critical revision of manuscript. YV—interpretation of data, drafting of manuscript, critical revision of manuscript. SN—study conception and design, acquisition of data, analysis and interpretation of data, drafting of manuscript, critical revision of manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thammineedi, S.R., Patnaik, S.C., Reddy, P. et al. Impact of fluorescent thoracic duct lymphography via intranodal approach in minimal access esophageal cancer surgery. Langenbecks Arch Surg 408, 426 (2023). https://doi.org/10.1007/s00423-023-03162-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-03162-2