Abstract
Purpose
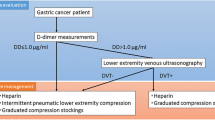
This study aimed to investigate the clinical efficacy of lower-extremity ultrasonography screening with early intervention for deep venous thrombosis (DVT) on the incidence of venous thromboembolism (VTE) after minimally invasive surgery (MIS) for gastric cancer (GC).
Methods
Between January 2012 and December 2019, 1070 patients were diagnosed with both clinical and pathological stage I–III GC and underwent MIS at our institution. Routine ultrasonographic screening for DVT in lower extremities is performed before MIS. Patients diagnosed with DVT were preoperatively administered anticoagulant therapy. Enoxaparin was routinely administrated after surgery irrespective of the presence of DVT. The incidence of postoperative symptomatic VTE was examined retrospectively.
Results
A total of 74 (6.9%) patients were preoperatively diagnosed with DVT. Multivariate analyses revealed that age > 70 years (p = 0.015), female sex (p < 0.001), and positive serum D-dimer test (p < 0.001) were significant and independent risk factors for preoperative DVT. The incidence of symptomatic postoperative VTE was 1 (0.09%); symptomatic VTE developed in one patient among patients without DVT, whereas no patient with DVT developed VTE.
Conclusions
Preoperative DVT screening using lower-extremity ultrasonography followed by preoperative anticoagulant therapy should be considered as a useful strategy to safely perform MIS for GC without increasing the incidence of VTE.


Similar content being viewed by others
Data availability
All data are presented in this manuscript.
Code availability
Not applicable.
References
Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC (2013) Epidemiology of cancer-associated venous thrombosis. Blood 122(10):1712–1723. https://doi.org/10.1182/blood-2013-04-460121
Li A, Garcia DA, Lyman GH, Carrier M (2019) Direct oral anticoagulant (DOAC) versus low-molecular-weight heparin (LMWH) for treatment of cancer associated thrombosis (CAT): a systematic review and meta-analysis. Thromb Res 173:158–163. https://doi.org/10.1016/j.thromres.2018.02.144
Crawford F, Andras A, Welch K, Sheares K, Keeling D (2016) Chappell FM (2016) D-dimer test for excluding the diagnosis of pulmonary embolism. Cochrane Database Syst Rev 8:Cd010864. https://doi.org/10.1002/14651858.CD010864.pub2
Rogers SO Jr, Kilaru RK, Hosokawa P, Henderson WG, Zinner MJ, Khuri SF (2007) Multivariable predictors of postoperative venous thromboembolic events after general and vascular surgery: results from the patient safety in surgery study. J Am Coll Surg 204(6):1211–1221. https://doi.org/10.1016/j.jamcollsurg.2007.02.072
Tapson VF (2004) Acute pulmonary embolism. Cardiol Clin 22(3):353-365,v. https://doi.org/10.1016/j.ccl.2004.04.002
Hata T, Ikeda M, Miyata H, Nomura M, Gotoh M, Sakon M, Yamamoto K, Wakabayashi G, Seto Y, Mori M, Doki Y (2019) Frequency and risk factors for venous thromboembolism after gastroenterological surgery based on the Japanese National Clinical Database (516 217 cases). Ann Gastroenterol Surg 3(5):534–543. https://doi.org/10.1002/ags3.12275
Sakon M, Maehara Y, Yoshikawa H, Akaza H (2006) Incidence of venous thromboembolism following major abdominal surgery: a multi-center, prospective epidemiological study in Japan. J Thromb Haemost 4(3):581–586. https://doi.org/10.1111/j.1538-7836.2006.01786.x
Clagett GP, Anderson FA Jr, Geerts W, Heit JA, Knudson M, Lieberman JR, Merli GJ, Wheeler HB (1998) Prevention of venous thromboembolism. Chest 114(5 Suppl):531s–560s. https://doi.org/10.1378/chest.114.5_supplement.531s
Kim W, Kim HH, Han SU, Kim MC, Hyung WJ, Ryu SW, Cho GS, Kim CY, Yang HK, Park DJ, Song KY, Lee SI, Ryu SY, Lee JH, Lee HJ (2016) Decreased morbidity of laparoscopic distal gastrectomy compared with open distal gastrectomy for stage I gastric cancer: short-term outcomes from a multicenter randomized controlled trial (KLASS-01). Ann Surg 263(1):28–35. https://doi.org/10.1097/sla.0000000000001346
Hyung WJ, Yang HK, Han SU, Lee YJ, Park JM, Kim JJ, Kwon OK, Kong SH, Kim HI, Lee HJ, Kim W, Ryu SW, Jin SH, Oh SJ, Ryu KW, Kim MC, Ahn HS, Park YK, Kim YH, Hwang SH, Kim JW, Cho GS (2019) A feasibility study of laparoscopic total gastrectomy for clinical stage I gastric cancer: a prospective multi-center phase II clinical trial, KLASS 03. Gastric Cancer 22(1):214–222. https://doi.org/10.1007/s10120-018-0864-4
Kim HH, Han SU, Kim MC, Kim W, Lee HJ, Ryu SW, Cho GS, Kim CY, Yang HK, Park DJ, Song KY, Lee SI, Ryu SY, Lee JH, Hyung WJ (2019) Effect of laparoscopic distal gastrectomy vs open distal gastrectomy on long-term survival among patients with stage I gastric cancer: the KLASS-01 randomized clinical trial. JAMA Oncol 5(4):506–513. https://doi.org/10.1001/jamaoncol.2018.6727
Katai H, Mizusawa J, Katayama H, Takagi M, Yoshikawa T, Fukagawa T, Terashima M, Misawa K, Teshima S, Koeda K, Nunobe S, Fukushima N, Yasuda T, Asao Y, Fujiwara Y, Sasako M (2017) Short-term surgical outcomes from a phase III study of laparoscopy-assisted versus open distal gastrectomy with nodal dissection for clinical stage IA/IB gastric cancer: Japan Clinical Oncology Group Study JCOG0912. Gastric Cancer 20(4):699–708. https://doi.org/10.1007/s10120-016-0646-9
Katai H, Mizusawa J, Katayama H, Morita S, Yamada T, Bando E, Ito S, Takagi M, Takagane A, Teshima S, Koeda K, Nunobe S, Yoshikawa T, Terashima M, Sasako M (2020) Survival outcomes after laparoscopy-assisted distal gastrectomy versus open distal gastrectomy with nodal dissection for clinical stage IA or IB gastric cancer (JCOG0912): a multicentre, non-inferiority, phase 3 randomised controlled trial. Lancet Gastroenterol Hepatol 5(2):142–151. https://doi.org/10.1016/s2468-1253(19)30332-2
Katai H, Mizusawa J, Katayama H, Kunisaki C, Sakuramoto S, Inaki N, Kinoshita T, Iwasaki Y, Misawa K, Takiguchi N, Kaji M, Okitsu H, Yoshikawa T, Terashima M (2019) Single-arm confirmatory trial of laparoscopy-assisted total or proximal gastrectomy with nodal dissection for clinical stage I gastric cancer: Japan Clinical Oncology Group study JCOG1401. Gastric Cancer 22(5):999–1008. https://doi.org/10.1007/s10120-019-00929-9
Shinohara T, Satoh S, Kanaya S, Ishida Y, Taniguchi K, Isogaki J, Inaba K, Yanaga K, Uyama I (2013) Laparoscopic versus open D2 gastrectomy for advanced gastric cancer: a retrospective cohort study. Surg Endosc 27(1):286–294. https://doi.org/10.1007/s00464-012-2442-x
Nakauchi M, Suda K, Kadoya S, Inaba K, Ishida Y, Uyama I (2016) Technical aspects and short- and long-term outcomes of totally laparoscopic total gastrectomy for advanced gastric cancer: a single-institution retrospective study. Surg Endosc 30(10):4632–4639. https://doi.org/10.1007/s00464-015-4726-4
Uyama I, Suda K, Satoh S (2013) Laparoscopic surgery for advanced gastric cancer: current status and future perspectives. J Gastric Cancer 13(1):19–25. https://doi.org/10.5230/jgc.2013.13.1.19
Nguyen NT, Cronan M, Braley S, Rivers R, Wolfe BM (2003) Duplex ultrasound assessment of femoral venous flow during laparoscopic and open gastric bypass. Surg Endosc 17(2):285–290. https://doi.org/10.1007/s00464-002-8812-z
Suda K, Man IM, Ishida Y, Kawamura Y, Satoh S, Uyama I (2015) Potential advantages of robotic radical gastrectomy for gastric adenocarcinoma in comparison with conventional laparoscopic approach: a single institutional retrospective comparative cohort study. Surg Endosc 29(3):673–685. https://doi.org/10.1007/s00464-014-3718-0
Shibasaki S, Suda K, Nakauchi M, Nakamura K, Kikuchi K, Inaba K, Uyama I (2020) Non-robotic minimally invasive gastrectomy as an independent risk factor for postoperative intra-abdominal infectious complications: a single-center, retrospective and propensity score-matched analysis. World J Gastroenterol 26(11):1172–1184. https://doi.org/10.3748/wjg.v26.i11.1172
(2021) Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer 24(1):1–21. https://doi.org/10.1007/s10120-020-01042-y
Shibasaki S, Suda K, Nakauchi M, Nakamura T, Kadoya S, Kikuchi K, Inaba K, Uyama I (2018) Outermost layer-oriented medial approach for infrapyloric nodal dissection in laparoscopic distal gastrectomy. Surg Endosc 32(4):2137–2148. https://doi.org/10.1007/s00464-018-6111-6
Nakamura K, Suda K, Suzuki A, Nakauchi M, Shibasaki S, Kikuchi K, Nakamura T, Kadoya S, Inaba K, Uyama I (2018) Intracorporeal isosceles right triangle-shaped anastomosis in totally laparoscopic distal gastrectomy. Surg Laparosc Endosc Percutan Tech 28(3):193–201. https://doi.org/10.1097/sle.0000000000000535
Uyama I, Kanaya S, Ishida Y, Inaba K, Suda K, Satoh S (2012) Novel integrated robotic approach for suprapancreatic D2 nodal dissection for treating gastric cancer: technique and initial experience. World J Surg 36(2):331–337. https://doi.org/10.1007/s00268-011-1352-8
Shibasaki S, Suda K, Nakauchi M, Kikuchi K, Kadoya S, Ishida Y, Inaba K, Uyama I (2017) Robotic valvuloplastic esophagogastrostomy using double flap technique following proximal gastrectomy: technical aspects and short-term outcomes. Surg Endosc 31(10):4283–4297. https://doi.org/10.1007/s00464-017-5489-x
Shibasaki S, Suda K, Nakauchi M, Nakamura K, Tanaka T, Kikuchi K, Inaba K, Uyama I (2020) Impact of the Endoscopic Surgical Skill Qualification System on the safety of laparoscopic gastrectomy for gastric cancer. Surg Endosc. https://doi.org/10.1007/s00464-020-08102-5
(2011) Guidelines for the diagnosis, treatment and prevention of pulmonary thromboembolism and deep vein thrombosis (JCS 2009). Circ J 75(5):1258–1281. https://doi.org/10.1253/circj.cj-88-0010
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213. https://doi.org/10.1097/01.sla.0000133083.54934.ae
Katayama H, Kurokawa Y, Nakamura K, Ito H, Kanemitsu Y, Masuda N, Tsubosa Y, Satoh T, Yokomizo A, Fukuda H, Sasako M (2016) Extended Clavien-Dindo classification of surgical complications: Japan Clinical Oncology Group postoperative complications criteria. Surg Today 46(6):668–685. https://doi.org/10.1007/s00595-015-1236-x
Hata T, Yasui M, Ikeda M, Miyake M, Ide Y, Okuyama M, Ikenaga M, Kitani K, Morita S, Matsuda C, Mizushima T, Yamamoto H, Murata K, Sekimoto M, Nezu R, Mori M, Doki Y (2019) Efficacy and safety of anticoagulant prophylaxis for prevention of postoperative venous thromboembolism in Japanese patients undergoing laparoscopic colorectal cancer surgery. Ann Gastroenterol Surg 3(5):568–575. https://doi.org/10.1002/ags3.12279
Tanizawa Y, Bando E, Kawamura T, Tokunaga M, Makuuchi R, Iida K, Nanri K, Yoneyama M, Terashima M (2017) Prevalence of deep venous thrombosis detected by ultrasonography before surgery in patients with gastric cancer: a retrospective study of 1140 consecutive patients. Gastric Cancer 20(5):878–886. https://doi.org/10.1007/s10120-016-0677-2
Wada T, Fujiwara H, Morita S, Fukagawa T, Katai H (2017) Incidence of and risk factors for preoperative deep venous thrombosis in patients undergoing gastric cancer surgery. Gastric Cancer 20(5):872–877. https://doi.org/10.1007/s10120-017-0690-0
Lee KW, Bang SM, Kim S, Lee HJ, Shin DY, Koh Y, Lee YG, Cha Y, Kim YJ, Kim JH, Park DJ, Kim HH, Oh D, Lee JS (2010) The incidence, risk factors and prognostic implications of venous thromboembolism in patients with gastric cancer. J Thromb Haemost 8(3):540–547. https://doi.org/10.1111/j.1538-7836.2009.03731.x
Dranitsaris G, Jelincic V, Choe Y (2012) Meta-regression analysis to indirectly compare prophylaxis with dalteparin or enoxaparin in patients at high risk for venous thromboembolic events. Clin Appl Thromb Hemost 18(3):233–242. https://doi.org/10.1177/1076029611426869
Sakon M, Kobayashi T, Shimazui T (2010) Efficacy and safety of enoxaparin in Japanese patients undergoing curative abdominal or pelvic cancer surgery: results from a multicenter, randomized, open-label study. Thromb Res 125(3):e65-70. https://doi.org/10.1016/j.thromres.2009.09.009
Jung YJ, Seo HS, Park CH, Jeon HM, Kim JI, Yim HW, Song KY (2018) Venous thromboembolism incidence and prophylaxis use after gastrectomy among Korean patients with gastric adenocarcinoma: the PROTECTOR randomized clinical trial. JAMA Surg 153(10):939–946. https://doi.org/10.1001/jamasurg.2018.2081
Bell BR, Bastien PE, Douketis JD (2015) Prevention of venous thromboembolism in the Enhanced Recovery After Surgery (ERAS) setting: an evidence-based review. Can J Anaesth 62(2):194–202. https://doi.org/10.1007/s12630-014-0262-2
Talec P, Gaujoux S, Samama CM (2016) Early ambulation and prevention of post-operative thrombo-embolic risk. J Visc Surg 153(6s):S11-s14. https://doi.org/10.1016/j.jviscsurg.2016.09.002 (1037-1052)
Rabe E, Partsch H, Morrison N, Meissner MH, Mosti G, Lattimer CR, Carpentier PH, Gaillard S, Jünger M, Urbanek T, Hafner J, Patel M, Wu S, Caprini J, Lurie F, Hirsch T (2020) Risks and contraindications of medical compression treatment - a critical reappraisal An international consensus statement. Phlebology 35(7):447–460. https://doi.org/10.1177/0268355520909066
Badireddy M, Mudipalli VR (2020) Deep Venous Thrombosis Prophylaxis StatPearls. StatPearls Publishing Copyright © 2020, StatPearls Publishing LLC., Treasure Island
Lee CH, Lin LJ, Cheng CL, Kao Yang YH, Chen JY, Tsai LM (2010) Incidence and cumulative recurrence rates of venous thromboembolism in the Taiwanese population. J Thromb Haemost 8(7):1515–1523. https://doi.org/10.1111/j.1538-7836.2010.03873.x
Cui G, Wang X, Yao W, Li H (2013) Incidence of postoperative venous thromboembolism after laparoscopic versus open colorectal cancer surgery: a meta-analysis. Surg Laparosc Endosc Percutan Tech 23(2):128–134. https://doi.org/10.1097/SLE.0b013e3182827cef
van der Graaf F, van den Borne H, van der Kolk M, de Wild PJ, Janssen GW, van Uum SH (2000) Exclusion of deep venous thrombosis with D-dimer testing–comparison of 13 D-dimer methods in 99 outpatients suspected of deep venous thrombosis using venography as reference standard. Thromb Haemost 83(2):191–198
Bates SM, Jaeschke R, Stevens SM, Goodacre S, Wells PS, Stevenson MD, Kearon C, Schunemann HJ, Crowther M, Pauker SG, Makdissi R, Guyatt GH (2012) Diagnosis of DVT: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141(2 Suppl):e351S-e418S. https://doi.org/10.1378/chest.11-2299
Mumoli N, Mastroiacovo D, Giorgi-Pierfranceschi M, Pesavento R, Mochi M, Cei M, Pomero F, Mazzone A, Vitale J, Ageno W, Dentali F (2018) Ultrasound elastography is useful to distinguish acute and chronic deep vein thrombosis. J Thromb Haemost 16(12):2482–2491. https://doi.org/10.1111/jth.14297
Matsuoka Y, Morimatsu H (2019) Incidence rates of postoperative pulmonary embolisms in symptomatic and asymptomatic patients, detected by diagnostic images - a single-center retrospective study. Circ J 83(2):432–440. https://doi.org/10.1253/circj.CJ-18-0729
Hope WW, Demeter BL, Newcomb WL, Schmelzer TM, Schiffern LM, Heniford BT, Sing RF (2007) Postoperative pulmonary embolism: timing, diagnosis, treatment, and outcomes. Am J Surg 194(6):814–818. https://doi.org/10.1016/j.amjsurg.2007.08.014 (discussion 818-819)
Verso M, Agnelli G (2003) Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J Clin Oncol 21(19):3665–3675. https://doi.org/10.1200/jco.2003.08.008
Acknowledgements
The authors would like to thank MARUZEN-YUSHODO Co., Ltd. (https://kw.maruzen.co.jp/kousei-honyaku/) for the English language editing.
Author information
Authors and Affiliations
Contributions
All the authors have fully met the ICMJE authorship criteria as follows. Study conception and design, Kazumitsu Suzuki, Susumu Shibasaki, Ichiro Uyama, and Koichi Suda; acquisition of the data, Kazumitsu Suzuki, Kenichi Nakamura, Tsuyoshi Tanaka, and Kenji Kikuchi; analysis and interpretation of the data, Kazumitsu Suzuki, Susumu Shibasaki, Koichi Suda, Masaya Nakauchi, Shingo Akimoto, and Kazuki Inaba; drafting of the manuscript, Kazumitsu Suzuki, Susumu Shibasaki, and Koichi Suda; critical revision of the manuscript, Susumu Shibasaki, Ichiro Uyama, and Koichi Suda. All authors read and approved the final manuscript. All authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. The present study was approved by the Institutional Review Board of Fujita Health University (HM18-409).
Consent to participate
Informed consent for this study was obtained through an opt-out method.
Consent for publication
Not applicable.
Conflict of interest
Kazumitsu Suzuki, Susumu Shibasaki, Masaya Nakauchi, Kenichi Nakamura, Shingo Akimoto, Tsuyoshi Tanaka, Kenji Kikuchi, Kazuki Inaba, Koichi Suda, and Ichiro Uyama have no commercial association with or financial involvement that might pose a conflict of interest in connection with the submitted article. Ichiro Uyama has received lecture fees from Intuitive Surgical, Inc., outside of the submitted work. Koichi Suda, Tsuyoshi Tanaka, and Kenji Kikuchi have been funded by Medicaroid, Inc. in relation to the Collaborative Laboratory for Research and Development in Advanced Surgical Technology, Fujita Health University. Koichi Suda has also received advisory fees from Medicaroid, Inc., outside of the submitted work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Suzuki, K., Shibasaki, S., Nakauchi, M. et al. Impact of routine preoperative sonographic screening with early intervention for deep venous thrombosis in lower extremities on preventing postoperative venous thromboembolism in patients with gastric cancer scheduled for minimally invasive surgery. Langenbecks Arch Surg 407, 597–608 (2022). https://doi.org/10.1007/s00423-021-02315-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-021-02315-5