Abstract
Purpose
To give an overview over cell therapeutic approaches to immunosuppression in clinical kidney transplantation. A focus is on myeloid suppressor cell therapy by mitomycin C-induced cells (MICs).
Methods
Literature review with an emphasis on already existing therapies.
Results
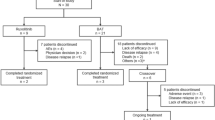
Several cell therapeutic approaches to immunosuppression and donor-specific unresponsiveness are now being tested in early phase I and phase II trials in clinical kidney transplantation. Cell products such as regulatory T cells or regulatory macrophages, or other myeloid suppressor cell therapies, may either consist of donor-specific, third-party, or autologous cell preparations. Major problems are the identification of the suppressive cell populations and their expansion to have sufficient amount of cells to achieve donor unresponsiveness (e.g., with regulatory T cells). We show a simple and safe way to establish donor unresponsiveness in living-donor kidney transplantation by MIC therapy. A phase I clinical trial is now under way to test the safety and efficacy of this cell therapeutic approach.
Conclusions
Cell therapeutic approaches to immunosuppression after kidney transplantation may revolutionize clinical transplantation in the future.



Similar content being viewed by others
References
Wolfe RA, Ashby VB, Milford EL, Ojo AO, Ettenger RE, Agodoa LY, Held PJ, Port FK (1999) Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant. N Engl J Med 341(23):1725–1730
Rana A, Gruessner A, Agopian VG, Khalpey Z, Riaz IB, Kaplan B, Halazun KJ, Busuttil RW, Gruessner RW (2015) Survival benefit of solid-organ transplant in the United States. JAMA Surg 150(3):252–259
Billingham RE, Brent L, Medawar PB (1953) Actively acquired tolerance of foreign cells. Nature 172(4379):603–606
Opelz G, Graver B, Terasaki PI (1981) Induction of high kidney graft survival rate by multiple transfusion. Lancet 1(8232):1223–1225
Marti HP, Henschkowski J, Laux G, Vogt B, Seiler C, Opelz G, Frey FJ (2006) Effect of donor-specific transfusions on the outcome of renal allografts in the cyclosporine era. Transpl Int 19(1):19–26
Schmetterer KG, Neunkirchner A, Pickl WF (2012) Naturally occurring regulatory T cells: markers, mechanisms, and manipulation. FASEB J 26(6):2253–2276
Liu W, Putnam AL, Xu-Yu Z, Szot GL, Lee MR, Zhu S, Gottlieb PA, Kapranov P, Gingeras TR, de St F, Groth B et al (2006) CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J Exp Med 203(7):1701–1711
Shevach EM (2009) Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity 30(5):636–645
Mahnke K, Bedke T, Enk AH (2007) Regulatory conversation between antigen presenting cells and regulatory T cells enhance immune suppression. Cell Immunol 250(1–2):1–13
Schaier M, Seissler N, Schmitt E, Meuer S, Hug F, Zeier M, Steinborn A (2012) DR(high+)CD45RA(−)-Tregs potentially affect the suppressive activity of the total Treg pool in renal transplant patients. PLoS One 7(3), e34208
Schaier M, Seissler N, Becker LE, Schaefer SM, Schmitt E, Meuer S, Hug F, Sommerer C, Waldherr R, Zeier M et al (2013) The extent of HLA-DR expression on HLA-DR(+) Tregs allows the identification of patients with clinically relevant borderline rejection. Transpl Int 26(3):290–299
Hsu SM, Mathew R, Taylor AW, Stein-Streilein J (2014) Ex-vivo tolerogenic F4/80(+) antigen-presenting cells (APC) induce efferent CD8(+) regulatory T cell-dependent suppression of experimental autoimmune uveitis. Clin Exp Immunol 176(1):37–48
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392(6673):245–252
Broichhausen C, Riquelme P, Geissler EK, Hutchinson JA (2012) Regulatory macrophages as therapeutic targets and therapeutic agents in solid organ transplantation. Curr Opin Organ Transplant 17(4):332–342
Hutchinson JA, Geissler EK (2015) Now or never? The case for cell-based immunosuppression in kidney transplantation. Kidney Int
Steinman RM (2012) Decisions about dendritic cells: past, present, and future. Annu Rev Immunol 30:1–22
Andre S, Tough DF, Lacroix-Desmazes S, Kaveri SV, Bayry J (2009) Surveillance of antigen-presenting cells by CD4+ CD25+ regulatory T cells in autoimmunity: immunopathogenesis and therapeutic implications. Am J Pathol 174(5):1575–1587
Kawai T, Cosimi AB, Spitzer TR, Tolkoff-Rubin N, Suthanthiran M, Saidman SL, Shaffer J, Preffer FI, Ding R, Sharma V et al (2008) HLA-mismatched renal transplantation without maintenance immunosuppression. N Engl J Med 358(4):353–361
Kawai T, Sachs DH, Sprangers B, Spitzer TR, Saidman SL, Zorn E, Tolkoff-Rubin N, Preffer F, Crisalli K, Gao B et al (2014) Long-term results in recipients of combined HLA-mismatched kidney and bone marrow transplantation without maintenance immunosuppression. Am J Transplant 14(7):1599–1611
Tang Q, Bluestone JA (2013) Regulatory T-cell therapy in transplantation: moving to the clinic. Cold Spring Harb Perspect Med 3(11)
Wood KJ, Bushell A, Hester J (2012) Regulatory immune cells in transplantation. Nat Rev Immunol 12(6):417–430
Gallon L, Traitanon O, Yu Y, Shi B, Leventhal JR, Miller J, Mas V, L X, Mathew JM (2015) Differential effects of calcineurin and mammalian target of rapamycin inhibitors on alloreactive Th1, Th17, and Regulatory T Cells. Transplantation
Scandling JD, Busque S, Shizuru JA, Lowsky R, Hoppe R, Dejbakhsh-Jones S, Jensen K, Shori A, Strober JA, Lavori P et al (2015) Chimerism, graft survival, and withdrawal of immunosuppressive drugs in HLA matched and mismatched patients after living donor kidney and hematopoietic cell transplantation. Am J Transplant 15(3):695–704
Leventhal JR, Elliott MJ, Yolcu ES, Bozulic LD, Tollerud DJ, Mathew JM, Konieczna I, Ison MG, Galvin J, Mehta J et al (2015) Immune reconstitution/immunocompetence in recipients of kidney plus hematopoietic stem/facilitating cell transplants. Transplantation 99(2):288–298
Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, Sun X, Chen J, Yang S, Cai J et al (2012) Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA 307(11):1169–1177
Nauta AJ, Fibbe WE (2007) Immunomodulatory properties of mesenchymal stromal cells. Blood 110(10):3499–3506
Schliesser U, Streitz M, Sawitzki B (2012) Tregs: application for solid-organ transplantation. Curr Opin Organ Transplant 17(1):34–41
Juvet SC, Whatcott AG, Bushell AR, Wood KJ (2014) Harnessing regulatory T cells for clinical use in transplantation: the end of the beginning. Am J Transplant 14(4):750–763
van der Net JB, Bushell A, Wood KJ, Harden PN (2015) Regulatory T cells: first steps of clinical application in solid organ transplantation. Transpl Int
Putnam AL, Brusko TM, Lee MR, Liu W, Szot GL, Ghosh T, Atkinson MA, Bluestone JA (2009) Expansion of human regulatory T-cells from patients with type 1 diabetes. Diabetes 58(3):652–662
Sagoo P, Ali N, Garg G, Nestle FO, Lechler RI, Lombardi G (2011) Human regulatory T cells with alloantigen specificity are more potent inhibitors of alloimmune skin graft damage than polyclonal regulatory T cells. Sci Transl Med 3(83):83ra42
Edozie FC, Nova-Lamperti EA, Povoleri GA, Scotta C, John S, Lombardi G, Afzali B (2014) Regulatory T-cell therapy in the induction of transplant tolerance: the issue of subpopulations. Transplantation 98(4):370–379
Brunstein CG, Miller JS, Cao Q, McKenna DH, Hippen KL, Curtsinger J, Defor T, Levine BL, June CH, Rubinstein P et al (2011) Infusion of ex vivo expanded T regulatory cells in adults transplanted with umbilical cord blood: safety profile and detection kinetics. Blood 117(3):1061–1070
Di Ianni M, Falzetti F, Carotti A, Terenzi A, Castellino F, Bonifacio E, Del Papa B, Zei T, Ostini RI, Cecchini D et al (2011) Tregs prevent GVHD and promote immune reconstitution in HLA-haploidentical transplantation. Blood 117(14):3921–3928
Trzonkowski P, Bieniaszewska M, Juscinska J, Dobyszuk A, Krzystyniak A, Marek N, Mysliwska J, Hellmann A (2009) First-in-man clinical results of the treatment of patients with graft versus host disease with human ex vivo expanded CD4+CD25+CD127- T regulatory cells. Clin Immunol 133(1):22–26
Geissler EK (2012) The ONE Study compares cell therapy products in organ transplantation: introduction to a review series on suppressive monocyte-derived cells. Transplant Res 1(1):11
Safinia N, Vaikunthanathan T, Fraser H, Scotta C, Lechler R, Lombardi G (2014) A GMP Treg expansion protocol restores Treg suppressor function in end-stage liver disease; implications for adoptive transfer therapy. Gut 63:A92–A93
Ferrer IR, Hester J, Bushell A, Wood KJ (2014) Induction of transplantation tolerance through regulatory cells: from mice to men. Immunol Rev 258(1):102–116
Wu T, Zhao Y, Zhao Y (2014) The roles of myeloid-derived suppressor cells in transplantation. Expert Rev Clin Immunol 10(10):1385–1394
Rosborough BR, Raich-Regue D, Turnquist HR, Thomson AW (2014) Regulatory myeloid cells in transplantation. Transplantation 97(4):367–379
Hutchinson JA, Riquelme P, Geissler EK, Fandrich F (2011) Human regulatory macrophages. Methods Mol Biol 677:181–192
Hutchinson JA, Riquelme P, Sawitzki B, Tomiuk S, Miqueu P, Zuhayra M, Oberg HH, Pascher A, Lutzen U, Janssen U et al (2011) Cutting edge: immunological consequences and trafficking of human regulatory macrophages administered to renal transplant recipients. J Immunol 187(5):2072–2078
Steinman RM, Banchereau J (2007) Taking dendritic cells into medicine. Nature 449(7161):419–426
Obeid M, Tesniere A, Ghiringhelli F, Fimia GM, Apetoh L, Perfettini JL, Castedo M, Mignot G, Panaretakis T, Casares N et al (2007) Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med 13(1):54–61
Jiga LP, Bauer TM, Chuang JJ, Opelz G, Terness P (2004) Generation of tolerogenic dendritic cells by treatment with mitomycin C: inhibition of allogeneic T-cell response is mediated by downregulation of ICAM-1, CD80, and CD86. Transplantation 77(11):1761–1764
Jiga LP, Ehser S, Kleist C, Opelz G, Terness P (2007) Inhibition of heart allograft rejection with mitomycin C-treated donor dendritic cells. Transplantation 83(3):347–350
Terness P, Oelert T, Ehser S, Chuang JJ, Lahdou I, Kleist C, Velten F, Hammerling GJ, Arnold B, Opelz G (2008) Mitomycin C-treated dendritic cells inactivate autoreactive T cells: toward the development of a tolerogenic vaccine in autoimmune diseases. Proc Natl Acad Sci U S A 105(47):18442–18447
Kleist C, Sandra-Petrescu F, Jiga L, Dittmar L, Mohr E, Greil J, Mier W, Becker LE, Lang P, Opelz G et al (2015) Generation of suppressive blood cells for control of allograft rejection. Clin Sci (Lond) 128(9):593–607
Radu CA, Kiefer J, Horn D, Kleist C, Dittmar L, Sandra F, Rebel M, Ryssel H, Koellensperger E, Gebhard MM et al (2012) Mitomycin-C-treated peripheral blood mononuclear cells (PBMCs) prolong allograft survival in composite tissue allotransplantation. J Surg Res 176(2):e95–e101
Liu L, Wang F, Zheng Y, Yuan X, Wang D, Zeng W, He X, Wang C, Deng S (2014) Pretreatment of transfused donor splenocytes and allografts with mitomycin C attenuates acute rejection in heart transplantation in mice. Transplant Proc 46(4):1169–1174
Funding
The TOL-1 study is funded by a grant from the German government (EXIST-Forschungstransfer: TolerogenixX, 03EFB BW56).
Conflicts of Interest
None.
Ethical approval
All procedures will perform in the planned TOL-1 study involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent will obtain from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding authors
Additional information
All authors drafted and critically revised the manuscript.
Rights and permissions
About this article
Cite this article
Morath, C., Schmitt, A., Zeier, M. et al. Cell therapy for immunosuppression after kidney transplantation. Langenbecks Arch Surg 400, 541–550 (2015). https://doi.org/10.1007/s00423-015-1313-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-015-1313-z