Abstract
Purpose
This randomized, controlled, single-blinded multicenter study evaluated the efficacy of latest-generation fibrin sealant containing synthetic aprotinin as fibrinolysis inhibitor as supportive treatment for hemostasis after elective partial hepatectomy.
Methods
Adult subjects undergoing resection of at least one liver segment were assigned to treatment with fibrin sealant or manual compression with a surgical gauze swab if persistent oozing necessitated additional hemostatic measures after primary control of arterial and venous bleeding. The primary outcome measure was the proportion of subjects with intraoperative hemostasis at 4 min after start of randomized treatment application. Secondary efficacy outcome measures included intraoperative hemostasis at 6, 8, and 10 min, intra- and postoperative rebleedings, transfusion requirements, and drainage volume.
Results
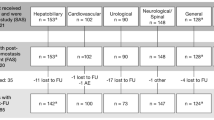
Seventy subjects were randomized. Hemostasis at 4 min was achieved in 29/35 (82.9 %) fibrin sealant subjects compared with 13/35 (37.1 %) control subjects (p < 0.001). Significantly more fibrin sealant subjects achieved hemostasis at 6 (p < 0.001), 8 (p = 0.028), and 10 min (p = 0.017). The number of rebleedings was low in both study arms. Transfusion requirements and 48-h drainage volumes were similar between the study arms. No adverse events related to study treatment were reported.
Conclusions
Fibrin sealant was shown to be safe and superior to manual compression in the control of parenchymal bleeding after hepatic resection. The use of synthetic aprotinin as fibrinolysis inhibitor further improves the safety margin of fibrin sealant by eliminating the risk of transmission of bovine spongiform encephalopathy and other bovine pathogens.


Similar content being viewed by others
References
Jarnagin WR, Gonen M, Fong Y, DeMatteo RP, Ben-Porat L, Little S et al (2002) Improvement in perioperative outcome after hepatic resection: analysis of 1,803 consecutive cases over the past decade. Ann Surg 236:397–407
Imamura H, Seyama Y, Kokudo N, Maema A, Sugawara Y, Sano K et al (2003) One thousand fifty-six hepatectomies without mortality in 8 years. Arch Surg 138:1198–1206
Poon RT, Fan ST, Lo CM, Liu CL, Lam CM, Yuen WK et al (2004) Improving perioperative outcome expands the role of hepatectomy in management of benign and malignant hepatobiliary diseases: analysis of 1222 consecutive patients from a prospective database. Ann Surg 240:698–708
Jones RM, Moulton CE, Hardy KJ (1998) Central venous pressure and its effect on blood loss during liver resection. Br J Surg 85:1058–1060
Smyrniotis V, Farantos C, Kostopanagiotou G, Arkadopoulos N (2005) Vascular control during hepatectomy: review of methods and results. World J Surg 29:1384–1396
Ishizaki Y, Yoshimoto J, Miwa K, Sugo H, Kawasaki S (2006) Safety of prolonged intermittent pringle maneuver during hepatic resection. Arch Surg 141:649–653
Alkozai EM, Lisman T, Porte RJ (2009) Bleeding in liver surgery: prevention and treatment. Clin Liver Dis 13:145–154
Poon RT (2007) Current techniques of liver transection. HPB 9:166–173
Gurusamy KS, Pamecha V, Sharma D, Davidson BR (2009) Techniques for liver parenchymal transection in liver resection. Cochrane Database Syst Rev 2009CD006880
Kohno H, Nagasue N, Chang YC, Taniura H, Yamanoi A, Nakamura T (1992) Comparison of topical hemostatic agents in elective hepatic resection: a clinical prospective randomized trial. World J Surg 16:966–969
Chapman WC, Clavien PA, Fung J, Khanna A, Bonham A (2000) Effective control of hepatic bleeding with a novel collagen-based composite combined with autologous plasma: results of a randomized controlled trial. Arch Surg 135:1200–1204
Schwartz M, Madariaga J, Hirose R, Shaver TR, Sher L, Chari R et al (2004) Comparison of a new fibrin sealant with standard topical hemostatic agents. Arch Surg 139:1148–1154
Frilling A, Stavrou GA, Mischinger HJ, de Hemptinne B, Rokkjaer M, Klempnauer J et al (2005) Effectiveness of a new carrier-bound fibrin sealant versus argon beamer as haemostatic agent during liver resection: a randomized prospective trial. Langenbecks Arch Surg 390:114–120
Noun R, Elias D, Balladur P, Bismuth H, Parc R, Lasser P et al (1996) Fibrin glue effectiveness and tolerance after elective liver resection: a randomized trial. Hepato-Gastroenterology 43:221–224
Eder F, Meyer F, Nestler G, Halloul Z, Lippert H (2005) Sealing of the hepatic resection area using fibrin glue reduces significant amount of postoperative drain fluid. World J Gastroenterol 11:5984–5987
Briceño J, Naranjo A, Ciria R, Díaz-Nieto R, Sánchez-Hidalgo JM, Luque A et al (2010) A prospective study of the efficacy of clinical application of a new carrier-bound fibrin sealant after liver resection. Arch Surg 145(5):482–488
Figueras J, Llado L, Miro M, Ramos E, Torras J, Fabregat J et al (2007) Application of fibrin glue sealant after hepatectomy does not seem justified. Ann Surg 245(4):536–542
Rousou J, Levitsky S, Gonzalez-Lavin L, Cosgrove D, Magilligan D, Weldon C et al (1989) Randomized clinical trial of fibrin sealant in patients undergoing resternotomy or reoperation after cardiac operations. A multicenter study. J Thorac Cardiovasc Surg 97:194–203
Lowe J, Luber J, Levitsky S, Hantak E, Montgomery J, Schiestl N et al (2007) Evaluation of the topical hemostatic efficacy and safety of TISSEEL VH S/D fibrin sealant compared with currently licensed TISSEEL VH in patients undergoing cardiac surgery: a phase 3, randomized, double-blind clinical study. J Cardiovasc Surg (Torino) 48:323–331
Saha SP, Muluk S, Schenk W 3rd, Burks SG, Grigorian A, Ploder B et al (2011) Use of fibrin sealant as a hemostatic agent in expanded polytetrafluoroethylene graft placement surgery. Ann Vasc Surg 25:813–822
Saha SP, Muluk S, Schenk W 3rd, Dennis JW, Ploder B, Grigorian A et al (2012) A prospective randomized study comparing fibrin sealant to manual compression for the treatment of anastomotic suture-hole bleeding in expanded polytetrafluoroethylene grafts. J Vasc Surg 56:134–141
Wichmann BA, Hill ID (1982) Algorithm AS 183: An efficient and portable pseudo-random number generator. Appl Statist 31:188–190
McLeod AI (1985) Remark AS R58 A remark on Algorithm AS 183. An efficient and portable pseudo-random number generator. J R Stat Soc Ser C Appl Stat 34:198–200
StatXact 8 PROCs Manual for SAS Users, Cytel Inc, Cambridge, 2007
Fischer L, Seiler CM, Broelsch CE, de Hemptinne B, Klempnauer J, Mischinger H-J et al (2011) Hemostatic efficacy of TachoSil in liver resection compared with argon beam coagulator treatment: an open, randomized, prospective, multicenter, parallel-group trial. Surgery 149:48–55
Katkhouda N (2008) Application of fibrin glue after hepatectomy might still be justified. Letter to the editor. Ann Surg 247:399–400
Capussotti L, Ferrero A, Viganò L, Sgotto E, Muratore A, Polastri R (2006) Bile leakage and liver resection. Arch Surg 141:690–694
Shippert RD (2005) A study of time-dependent operating room fees and how to save $100 000 by using time-saving products. Am J Cosmetic Surg 22(1):25–34
Beierlein W, Scheule AM, Antoniadis G, Braun C, Schosser R (2000) An immediate, allergic skin reaction to aprotinin after reexposure to fibrin sealant. Transfusion 40(3):302–305
Scheule AM, Beierlein W, Wendel HP, Eckstein FS, Heinemann MK, Ziemer G (1998) Fibrin sealant, aprotinin, and immune response in children undergoing operations for congenital heart disease. J Thorac Cardiovasc Surg 115:883–889
Horowitz B, Prince AM, Horowitz MS, Watklevicz C (1993) Viral safety of solvent-detergent treated blood products. In: Brown F (ed) Virological safety aspects of plasma derivatives. Dev Biol Stand. Karger, Basel, 81:147–161
Acknowledgments
The following principal investigators and centers participated in the study (in alphabetical order): Hüseyin Bektas, MD, Medical University of Hannover, Clinic for General, Visceral and Transplant Surgery, D-30625 Hannover, Germany; Markus Buechler, MD who as principal investigator followed Jan Schmidt, MD, University of Heidelberg Surgical Clinic, Clinic for General, Visceral and Transplant Surgery, D-69120 Heidelberg, Germany; Sven Jonas, MD, University Clinic of Leipzig, Clinic for Visceral, Transplant, Thorax and Vascular Surgery, D-04103 Leipzig, Germany; Gernot Kaiser, MD, University Clinic of Essen, Clinic for General and Transplant Surgery, D-45147 Essen, Germany; Silvio Nadalin, MD, Eberhard Karls University of Tuebingen, Clinic for General, Visceral and Transplant Surgery, D-72076 Tuebingen, Germany; Daniel Seehofer, MD, Charité-Medical University of Berlin, Clinic for General, Visceral and Transplant Surgery, D-13353 Berlin, Germany; and Utz Settmacher, MD, University Clinic of Jena, Clinic for General, Visceral and Vascular Surgery, D-07747 Jena, Germany. This clinical research was sponsored by Baxter Innovations GmbH, Vienna, Austria.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The study was registered under ClinicalTrials.gov identifier NCT01244425.
Rights and permissions
About this article
Cite this article
Bektas, H., Nadalin, S., Szabo, I. et al. Hemostatic efficacy of latest-generation fibrin sealant after hepatic resection: a randomized controlled clinical study. Langenbecks Arch Surg 399, 837–847 (2014). https://doi.org/10.1007/s00423-014-1227-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-014-1227-1