Abstract
Background
Radiofrequency ablation (RFA) is an inherent part of curative treatment within a multimodal therapy concept of malignant liver tumors. The biggest problem is the high rate of local recurrences in tumors with a diameter of more than 3 cm because of the high variability and poor reproducibility of the zone of ablation. No imaging modality facilitates monitoring during neither intraoperativ nor percutaneous RFA. This experimental study describes and compares an in vitro and in vivo porcine model by its electro-physiological parameters with the aim of monitoring RFA procedures.
Materials and methods
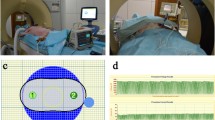
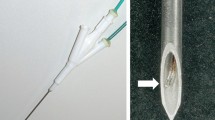
RFA was performed in a perfused in vitro porcine (one RFA per liver) and in vivo porcine model (24 animals) with three different RFA systems (Rita XL 5 cm, Rita XLi 7 cm, LeVeen 5 cm). In the in vivo model, percutaneous placement of the RFA device was guided by native and contrast-enhanced CT scan. The electro-physical parameters during RFA were online (in real time) recorded by a dedicated software. After the RFA, the livers were explanted, sliced, and measured according to the consensus technique.
Results
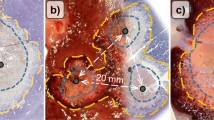
The delivered energy was in vivo versus in vitro: Rita XL 238 ± 135 kJ versus 135 ± 53 kJ (p = 0.247); Rita XLi 711 ± 180 kJ versus 159 ± 54 (p = 0.016) and with LeVeen 212 ± 71 kJ (in vivo). The LeVeen system was inconsistent in the in vitro model. This correlates to an energy consumption per ml of necrosis in vivo versus in vitro Rita XL of 8 ± 3 kJ/ml versus 6.4 ± 3.9 kJ/ml (p = 0.537), Rita XLi of 10 ± 6 kJ/ml versus 1.8 ± 0.2 kJ/ml (p = 0.016), and LeVeen of 14.0 ± 12 kJ/ml (in vivo). The volume of ablation was in vivo versus in vitro Rita XL 30 ± 10 ml versus 26 ± 17 ml (p = 0.329), Rita XLi 90 ± 58 ml versus 88 ± 21 ml (p = 0.905), and LeVeen 22 ± 11 ml versus 50 ± 12 ml (p = 0.04). The impedance during RFA were in vivo versus in vitro Rita XL 39 ± 4 Ω versus 50 ± 14 Ω (p < 0.247), Rita XLi 33 ± 5 Ω versus 61 ± 16 Ω (p = 0.016) and LeVeen 31 ± 2 Ω (in vivo).
Conclusion
The volume of ablation showed analogue data in vivo and in vitro. The delivered energy and energy consumption was in vivo up to five times (Rita XLi) higher than in vitro and the impedance in vivo was always lower than in vitro. These differences observed between in vivo and in vitro were more pronounced than previously described. Thus the use of an in vitro model for research of the RFA technique must be challenged. The large deployment of the Rita XLi was a problem for percutaneous positioning of the device without direct contact to liver surface or major vessels in 80-kg pigs and to a lesser extent in in vitro liver originating from 130- to 140-kg pigs. Modern RFA systems which generate large volume of tissue necrosis can therefore only be adequately tested in a porcine model with a liver weight of at least 1.5–2 kg. Alternatively, a bovine liver model (with a liver weight up to 10 kg) should be developed in the future.



Similar content being viewed by others
References
Mulier S, Ni Y, Jamart J, Ruers T, Marchal G, Michel L (2005) Local recurrence after hepatic radiofrequency coagulation: multivariate meta-analysis and review of contributing factors. Ann Surg 242(2):158–171. doi:10.1097/01.sla.0000171032.99149.fe
Stippel DL, Brochhagen HG, Arenja M, Hunkemoller J, Holscher AH, Beckurts KT (2004) Variability of size and shape of necrosis induced by radiofrequency ablation in human livers: a volumetric evaluation. Ann Surg Oncol 11(4):420–425. doi:10.1245/ASO.2004.09.012
Leen E, Kumar S, Khan SA, Low G, Ong KO, Tait P, Averkiou M (2009) Contrast-enhanced 3D ultrasound in the radiofrequency ablation of liver tumors. World J Gastroenterol 15(3):289–299. doi:10.3748/wjg.15.289
Lepetit-Coiffe M, Quesson B, Seror O, Dumont E, Le Bail B, Moonen CT, Trillaud H (2006) Real-time monitoring of radiofrequency ablation of rabbit liver by respiratory-gated quantitative temperature MRI. J Magn Reson Imaging 24(1):152–159. doi:10.1002/jmri.20605
Seror O, Lepetit-Coiffe M, Quesson B, Trillaud H, Moonen CT (2006) Quantitative magnetic resonance temperature mapping for real-time monitoring of radiofrequency ablation of the liver: an ex vivo study. Eur Radiol 16(10):2265–2274. doi:10.1007/s00330-006-0210-9
Stippel DL, Bangard C, Prenzel K, Yavuzyasar S, Fischer JH, Holscher AH (2008) Which parameters are needed for targeting a multitined radiofrequency device—an approach to a simple algorithm. Langenbecks Arch Surg. doi:10.1007/s00423-088-0306-6
Solazzo SA, Liu Z, Lobo SM, Ahmed M, Hines-Peralta AU, Lenkinski RE, Goldberg SN (2005) Radiofrequency ablation: importance of background tissue electrical conductivity—an agar phantom and computer modeling study. Radiology 236(2):495–502. doi:10.1148/radiol.2362040965
Mulier S, Ni Y, Frich L, Burdio F, Denys AL, De Wispelaere JF, Dupas B, Habib N, Hoey M, Jansen MC et al (2007) Experimental and clinical radiofrequency ablation: proposal for standardized description of coagulation size and geometry. Ann Surg Oncol 14(4):1381–1396. doi:10.1245/s10434-006-9033-9
Ng KK, Lam CM, Poon RT, Shek TW, Yu WC, To JY, Wo YH, Lau CP, Tang TC, Ho DW et al (2005) Porcine liver: morphologic characteristics and cell viability at experimental radiofrequency ablation with internally cooled electrodes. Radiology 235(2):478–486. doi:10.1148/radiol.2352040425
Grundmann RT, Hermanek P, Merkel S, Germer CT, Hauss J, Henne-Bruns D, Herfarth K, Hopt UT, Junginger T, Klar E et al (2008) Diagnosis and treatment of colorectal liver metastases—workflow. Zentralbl Chir 133(3):267–284. doi:10.1055/s-2008-1076796
Itoh T, Orba Y, Takei H, Ishida Y, Saitoh M, Nakamura H, Meguro T, Horita S, Fujita M, Nagashima K (2002) Immunohistochemical detection of hepatocellular carcinoma in the setting of ongoing necrosis after radiofrequency ablation. Mod Pathol 15(2):110–115. doi:10.1038/modpathol.3880501
Brieger J, Pereira PL, Trubenbach J, Schenk M, Krober SM, Schmidt D, Aube C, Claussen CD, Schick F (2003) In vivo efficiency of four commercial monopolar radiofrequency ablation systems: a comparative experimental study in pig liver. Invest Radiol 38(10):609–616. doi:10.1097/01.rli.0000077122.70885.94
Germer CT, Buhr HJ, Isbert C (2005) Nonoperative ablation for liver metastases. Possibilities and limitations as a curative treatment. Chirurg 76(6):552–554, 556–563. doi:10.1007/s00104-005-1047-5
Scott DJ, Fleming JB, Watumull LM, Lindberg G, Tesfay ST, Jones DB (2002) The effect of hepatic inflow occlusion on laparoscopic radiofrequency ablation using simulated tumors. Surg Endosc 16(9):1286–1291. doi:10.1007/s004640080167
Bitsch RG, Dux M, Helmberger T, Lubienski A (2006) Effects of vascular perfusion on coagulation size in radiofrequency ablation of ex vivo perfused bovine livers. Invest Radiol 41(4):422–427. doi:10.1097/01.rli.0000201231.60420.a2
Author information
Authors and Affiliations
Corresponding author
Additional information
Best of Abstracts—Chirurgisches Forum 2009, Deutsche Gesellschaft für Chirurgie.
Rights and permissions
About this article
Cite this article
Wahba, R., Bangard, C., Kleinert, R. et al. Electro-physiological parameters of hepatic radiofrequency ablation—a comparison of an in vitro versus an in vivo porcine liver model. Langenbecks Arch Surg 394, 503–509 (2009). https://doi.org/10.1007/s00423-009-0475-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-009-0475-y