Abstract
Background and aims
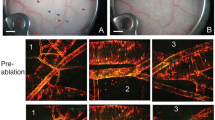
Neovascularization plays a pivotal role in tissue engineering and tissue regeneration. However, reliable technologies to visualize and quantify blood vessel networks in target tissue areas are still pending. In this work, we introduce a new method which allows comparing vascularization levels in normal and tissue-engineered skin.
Materials and methods
Normal skin was isolated, and vascular dermal regeneration was analyzed based on tissue transillumination and computerized digital segmentation. For tissue-engineered skin, a bilateral full skin defect was created in a nude mouse model and then covered with a commercially available scaffold for dermal regeneration. After 3 weeks, the whole skin (including scaffold for dermal regeneration) was harvested, and vascularization levels were analyzed.
Results
The blood vessel network in the skin was better visualized by transillumination than by radio-angiographic studies, the gold standard for angiographies. After visualization, the whole vascular network was digitally segmented showing an excellent overlapping with the original pictures. Quantification over the digitally segmented picture was performed, and an index of vascularization area (VAI) and length (VLI) of the vessel network was obtained in target tissues. VAI/VLI ratio was calculated to obtain the vessel size index.
Conclusions
We present a new technique which has several advantages compared to others, as animals do not require intravascular perfusions, total areas of interest can be quantitatively analyzed at once, and the same target tissue can be processed for further experimental analysis.







Similar content being viewed by others
References
MacNeil S (2007) Progress and opportunities for tissue-engineered skin. Nature 445:874–880
Shepherd BR, Enis RE, Wang F, Suarez Y, Pober JS, Schechner JS (2006) Vascularization and engraftment of a human skin substitute using circulating progenitor cell-derived endothelial cells. FASEB J 10:1739–1741
Laschke MW, Harder Y, Amon M, Martin I, Farhadi J, Ring A, Torio-Padron N, Schramm R, Rucker M, Junker D, Haufel JM, Carvalho C, Germann G, Vollmar B, Menger MD (2006) Angiogenesis in tissue engineering: breathing life into constructed tissue substitutes. Tissue Eng 12:2093–2104
Brey EM, Uriel S, Greisler HP, McIntire LV (2005) Therapeutic neovascularization: contributions from bioengineering. Tissue Eng 11:567–584
Machens HG, Grzybowski S, Bucsky B, Spanholtz T, Niedworok C, Maichle A, Stoeckelhuber B, Condurache A, Liu F, Egana JT, Kaum M, Mailender P, Aacht T (2006) A technique to detect and to quantify fasciocutaneous blood vessels in small laboratory animals ex vivo. J Surg Res 131:91–96
Mc Donald DM, Choyke PL (2003) Imaging of angiogenesis: from microscope to clinic. Nat Med 9:713–725
Bergeron L, Tang M, Morris SF (2006) A review of vascular injection techniques for the study of perforator flaps. Plast Reconstr Surg 117:2050–2057
Condurache A, Aach T (2005a) Vessel segmentation in angiograms using hysteresis thresholding. Proceedings of the Ninth IAPR Conference on Machine Vision Applications. 269–272
Condurache A, Aach T, Grzybowsky S, Machens HG (2005b) Vessel segmentation and analysis in laboratory skin transplant micro-angiograms. Proceedings of the Eighteenth IEEE Symposium on Computer-Based Medical Systems. 21–26
Rucker M, Laschke MW, Junker D, Carvalho C, Tavassol F, Mulhaupt R, Gelrich NC, Menger MD (2008) Vascularization and biocompatibility of scaffolds consisting of different calcium phosphate compounds. J Biomed Mater Res (in press)
Acknowledgments
The authors would like to thank Dr. Ralf Werner and Dr. Felipe Opazo for their critical discussion of the work and Dr. Ignacio Bazán for his help during the manuscript preparation. This work was supported by the University of Luebeck (Grant to HGM).
Conflict of interest statement
None disclosed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Egaña, J.T., Condurache, A., Lohmeyer, J.A. et al. Ex vivo method to visualize and quantify vascular networks in native and tissue engineered skin. Langenbecks Arch Surg 394, 349–356 (2009). https://doi.org/10.1007/s00423-008-0333-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-008-0333-3