Abstract
Background
Animal experiments reveal significant gender differences in the immunological response to surgical trauma. This raises the possibility that gender differences may also exist in patients after major abdominal surgery.
Patients and methods
This prospective study included 40 patients (20 men, 20 women) with colorectal diseases requiring surgical intervention. To evaluate the immune response to surgery circulating lymphocyte populations and natural killer cells were determined by flow-cytometry, and IL-6 serum levels were measured by enzyme-linked immunosorbent assay. Blood samples were taken before and on days 1, 2, and 5 after surgery.
Results
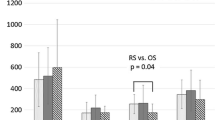
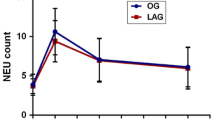
Despite comparable preoperative cell counts we detected significant postoperative gender differences regarding B-lymphocyte, T-lymphocyte, T-helper cell counts, and NK cell counts. While only a short, insignificant depression of these immune competent cells was detected in women, men suffered long-lasting (5 days) depression of these cells. Furthermore, women showed a more pronounced immediate (day 1) proinflammatory response (circulating IL-6) after abdominal surgery.
Conclusions
Significant immunological gender differences following major abdominal surgery were observed in this prospective clinical study. Our findings support the experimental observations of better posttraumatic immune competence in women than in men. These gender differences may be of relevance for short- and long-term results after surgery for colorectal diseases. Future studies will address the use of sex-steroids and/or their antagonists as a therapeutic option for the improvement in perioperative immune dysfunction in patients with major surgery.





Similar content being viewed by others
References
Wichmann MW, Zellweger R, DeMaso CM, Ayala A, Chaudry IH (1996) Enhanced immune responses in females, as opposed to decreased responses in males following haemorrhagic shock and resuscitation. Cytokine 8:853–863
Zellweger R, Wichmann MW, Ayala A, Stein S, DeMaso CM, Chaudry IH (1997) Females in proestrus state maintain splenic immune functions and tolerate sepsis better than males. Crit Care Med 25:106–110
Wichmann MW, Ayala A, Chaudry IH (1997) Male sex-steroids are responsible for depressing macrophage immune function after trauma-hemorrhage. Am J Physiol 273:C1335–C1340
Wichmann MW, Zellweger R, DeMaso CM, Ayala A, Chaudry IH (1996) Mechanisms of immunosuppression in males following trauma-hemorrhage: critical role of testosterone. Arch Surg 131:1186–1192
Angele MK, Knöferl MW, Ayala A, Albina JE, Cioffi WG, Bland KI, Chaudry IH (1999) Testosterone and estrogen regulate pro- and antiinflammatory cytokine release by macrophages following trauma-hemorrhage. Am J Physiol 277:C35–C42
Messingham KA, Messingham KA, Shirazi M, Duffner LA, Duffner LA, Emanuele MA, Kovacs EJ, Kovacs EJ, Kovacs EJ, Kovacs EJ (2001) Testosterone receptor blockade restores cellular immunity in male mice after burn injury. J Endocrinol 169:299–308
Angele MK, Schwacha MG, Ayala A, Chaudry IH (2000) Effect of gender and sex on immune responses following shock. Shock 14:81–90
Bone RC (1992) Toward an epidemiology and natural history of SIRS (systemic inflammatory response syndrome). JAMA 268:3452–3455
Offner PJ, Moore EE, Biffl WL (1999) Male gender is a risk factor for major infections after surgery. Arch Surg 134:935–938; discussion 938–940
Oberholzer A, Keel M, Zellweger R, Steckholzer U, Trentz O, Ertel W (2000) Incidence of septic complications and multiple organ failure in severely injured patients is sex specific. J Trauma 48:932–937
Majetschak M, Christensen B, Obertacke U, Waydhas C, Schindler AE, Nast-Kolb D, Schade FU (2000) Sex differences in posttraumatic cytokine release of endotoxin-stimulated whole blood: relationship to the development of severe sepsis. J Trauma 48:832–840
Schröder J, Kahlke V, Staubach KH, Zabel P, Stüber F (1998) Gender differences in human sepsis. Arch Surg 133:1200–1205
Wichmann MW, Inthorn D, Andress HJ, Schildberg FW (2000) Incidence and mortality of severe sepsis in surgical intensive care patients: the influence of patient gender on disease process and outcome. Intensive Care Med 26:167–172
Albers MJIJ, de Gast-Bakker DAH, van Dam NAM, Madern GC, Tibboel D (2002) Male sex predisposes the newborn surgical patient to parenteral nutrition-associated cholestasis and to sepsis. Arch Surg 137:789–793
Center for Disease Control (1992) Mortality patterns – United States, 1989. Morbidity Mortality Weekly Report (MMWR) 41:121–125
Salo M (1992) Effects of anaesthesia and surgery on the immune response. Acta Anaesthesiol Scand 36:201–220
Shigemitsu Y, Saito T, Kinoshita T, Kobayashi M (1992) Influence of surgical stress on bactericidal activity of neutrophils and complications of infection in patients with esophageal cancer. J Surg Oncol 50:90–97
Wichmann MW, Muller C, Hornung HM, Lau-Werner U, Schildberg FW (2001) Gender differences in long-term survival of patients with colorectal cancer. Br J Surg 88:1092–1098
McGowan JE, Barnes MW, Finland N (1975) Bacteremia at Boston City Hospital: occurrence and mortality during 12 selected years (1935–1972) with special reference to hospital-acquired cases. J Infect Dis 132:316–335
Bone RC (1992) Toward an epidemiology and natural history of SIRS (systemic inflammatory response syndrome). JAMA 268:3452–3455
Wichmann MW, Inthorn D, Andress HJ, Schildberg FW (2000) Incidence and mortality of severe sepsis in surgical intensive care patients: the influence of patient gender on disease process and outcome. Intensive Care Med 26:167–172
Ordemann J, Jacobi CA, Schwenk W, Stösslein R, Müller JM (2001) Cellular and humoral inflammatory response after laparoscopic and conventional colorectal resections. Results of a prospective randomized trial. Surg Endosc 15:600–608
Lennard TWJ, Shenton BK, Borzotta A, Donnelly PK, White M, Gerrie LM, Proud G, Taylor RMR (1985) The influence of surgical operations on components of the human immune system. Br J Surg 72:771–776
Tartter PI (1988) Preoperative lymphocyte subsets and infectious complications after colorectal cancer surgery. Surgery 103:226–230
Tang CL, Eu KW, Tai BC, Soh JGS, Machin D, Seow-Choen F (2001) Randomized clinical trial of the effect of open versus laparoscopically assisted colectomy on systemic immunity in patients with colorectal cancer. Br J Surg 88:801–807
Tartter PI, Steinberg B, Barron DM, Martinelli G (1987) The prognostic significance of natural killer cytotoxicity in patients with colorectal cancer. Arch Surg 122:1264–1268
Acknowledgements
Part of this work was presented at the 119th Annual Meeting of the German Surgical Society, Berlin, May 2002.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wichmann, M.W., Müller, C., Meyer, G. et al. Different immune responses to abdominal surgery in men and women. Langenbecks Arch Surg 387, 397–401 (2003). https://doi.org/10.1007/s00423-002-0346-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-002-0346-2