Abstract
Purpose
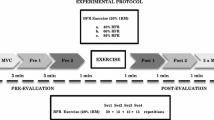
Constant blood flow occlusion (BFO) superimposed on aerobic exercise can impair muscle function and exercise tolerance; however, no study has investigated the effect of intermittent BFO on the associated responses. Fourteen participants (n = 7 females) were recruited to compare neuromuscular, perceptual, and cardiorespiratory responses to shorter (5:15s, occlusion-to-release) and longer (10:30s) BFO applied during cycling to task failure.
Methods
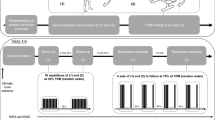
In randomized order, participants cycled to task failure (task failure 1) at 70% of peak power output with (i) shorter BFO, (ii) longer BFO, and (iii) no BFO (Control). Upon task failure in the BFO conditions, BFO was removed, and participants continued cycling until a second task failure (task failure 2). Maximum voluntary isometric knee contractions (MVC) and femoral nerve stimuli were performed along with perceptual measures at baseline, task failure 1, and task failure 2. Cardiorespiratory measures were recorded continuously across the exercises.
Results
Task failure 1 was longer in Control than 5:15s and 10:30s (P < 0.001), with no differences between the BFO conditions. At task failure 1, 10:30s elicited a greater decline in twitch force compared to 5:15s and Control (P < 0.001). At task failure 2, twitch force remained lower in 10:30s than Control (P = 0.002). Low-frequency fatigue developed to a greater extent in 10:30s compared to Control and 5:15s (P < 0.047). Dyspnea and Fatigue were greater for Control than 5:15s and 10:30s at the end of task failure 1 (P < 0.002).
Conclusion
Exercise tolerance during BFO is primarily dictated by the decline in muscle contractility and accelerated development of effort and pain.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abe T, Fujita S, Nakajima T et al (2010) Effects of low-intensity cycle training with restricted leg blood flow on thigh muscle volume and VO2max in young men. J Sports Sci Med 9:452–458
Aboodarda SJ, Mira J, Floreani M et al (2018) Effects of endurance cycling training on neuromuscular fatigue in healthy active men. Part II: Corticospinal excitability and voluntary activation. Eur J Appl Physiol 118:2295–2305. https://doi.org/10.1007/S00421-018-3951-7
Aboodarda SJ, Iannetta D, Emami N et al (2020) Effects of pre-induced fatigue vs. concurrent pain on exercise tolerance, neuromuscular performance and corticospinal responses of locomotor muscles. J Physiol 598:285–302. https://doi.org/10.1113/jp278943
Allen DG, Lamb GD, Westerblad H (2008) Skeletal muscle fatigue: cellular mechanisms. Physiol Rev 88:287–332. https://doi.org/10.1152/physrev.00015.2007
Amann M, Eldridge MW, Lovering AT et al (2006) Arterial oxygenation influences central motor output and exercise performance via effects on peripheral locomotor muscle fatigue in humans. J Physiol 575:937–952. https://doi.org/10.1113/jphysiol.2006.113936
Amann M, Blain GM, Proctor LT et al (2010) Group III and IV muscle afferents contribute to ventilatory and cardiovascular response to rhythmic exercise in humans. J Appl Physiol 109:966–976. https://doi.org/10.1152/JAPPLPHYSIOL.00462.2010
Amann M, Sidhu S, Weavil J et al (2015) Autonomic responses to exercise: group III/IV muscle afferents and fatigue. Auton Neurosci 188:19–23. https://doi.org/10.1016/j.autneu.2014.10.018
Babault N, Desbrosses K, Fabre M et al (2006) Neuromuscular fatigue development during maximal concentric and isometric knee extensions. J Appl Physiol 100:780–785. https://doi.org/10.1152/japplphysiol.00737.2005
Bakeman R (2005) Recommended effect size statistics for repeated measures designs. Behav Res Methods 37:379–384. https://doi.org/10.3758/bf03192707
Borg G (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14:377–381. https://doi.org/10.1249/00005768-198205000-00012
Borg G (1998) Borg’s perceived exertion and pain scales. Human Kinetics, Champaign, IL
Borg E, Borg G, Larsson K et al (2010) An Index for breathlessness and leg fatigue. Scand J Med Sci Sports 20:544–650. https://doi.org/10.1111/j.1600-0838.2009.00985.x
Broxterman RM, Craig JC, Smith JR et al (2015) Influence of blood flow occlusion on the development of peripheral and central fatigue during small muscle mass handgrip exercise. J Physiol 593:4043–4054. https://doi.org/10.1113/jp270424
Canestri R, Franco-Alvarenga PE et al (2021) Effects of experimentally induced muscle pain on endurance performance: a proof-of-concept study assessing neurophysiological and perceptual responses. Psychophysiology. https://doi.org/10.1111/psyp.13810
Centner C, Wiegel P, Gollhofer A, König D (2019) Effects of blood flow restriction training on muscular strength and hypertrophy in older individuals: a systematic review and meta-analysis. Sports Med 49:95–108. https://doi.org/10.1007/s40279-018-0994-1
Cohen J (1988) Statistical power analysis for the behavioural sciences. Lawrence Erlbaum Associates, Hillsdale, NJ
Conceição MS, Gáspari AF, Ramkrapes APB et al (2018) Anaerobic metabolism induces greater total energy expenditure during exercise with blood flow restriction. PLoS ONE 13:e0194776. https://doi.org/10.1371/JOURNAL.PONE.0194776
Copithorne DB, Rice CL (2019) The effect of blood flow occlusion during acute low-intensity isometric elbow flexion exercise. Eur J Appl Physiol 119:587–595. https://doi.org/10.1007/S00421-019-04088-8
Davies MJ, Benson AP, Cannon DT et al (2017) Dissociating external power from intramuscular exercise intensity during intermittent bilateral knee-extension in humans. J Physiol 595:6673–6686. https://doi.org/10.1113/jp274589
de Almeida AR, Jazayeri D, Yeung ST et al (2022) The effects of pain induced by blood flow occlusion in one leg on exercise tolerance and corticospinal excitability and inhibition of the contralateral leg in males. Appl Physiol Nutr Metab 47:632–648. https://doi.org/10.1139/apnm-2021-0597
de Oliveira MFM, Caputo F, Corvino RB, Denadai BS (2016) Short-term low-intensity blood flow restricted interval training improves both aerobic fitness and muscle strength. Scand J Med Sci Sports 26:1017–1025. https://doi.org/10.1111/sms.12540
Doyle-Baker D, Temesi J, Medysky ME et al (2018) An innovative ergometer to measure neuromuscular fatigue immediately after cycling. Med Sci Sports Exerc 50:375–387. https://doi.org/10.1249/mss.0000000000001427
Enoka RM, Duchateau J (2016) Translating fatigue to human performance. Med Sci Sports Exerc 48:2228–2238. https://doi.org/10.1249/mss.0000000000000929
Fiorenza M, Hostrup M, Gunnarsson TP et al (2019) Neuromuscular fatigue and metabolism during high-intensity intermittent exercise. Med Sci Sports Exerc 51:1642–1652. https://doi.org/10.1249/mss.0000000000001959
Fitts RH (2008) The cross-bridge cycle and skeletal muscle fatigue. J Appl Physiol 104:551–558. https://doi.org/10.1152/japplphysiol.01200.2007
Gandevia SC (1992) Some central and peripheral factors affecting human motoneuronal output in neuromuscular fatigue. Sports Med Int J Appl Med Sci Sport Exerc 13:93–98. https://doi.org/10.2165/00007256-199213020-00004
Hammer SM, Alexander AM, Didier KD, Barstow TJ (2020) Influence of blood flow occlusion on muscular recruitment and fatigue during maximal-effort small muscle-mass exercise. J Physiol 598:4293–4306. https://doi.org/10.1113/JP279925
Hughes L, Paton B, Rosenblatt B et al (2017) Blood flow restriction training in clinical musculoskeletal rehabilitation: a systematic review and meta-analysis. Br J Sports Med 51:1003–1011. https://doi.org/10.1136/bjsports-2016-097071
Hureau TJ, Romer LM, Amann M (2018) The ‘sensory tolerance limit’: a hypothetical construct determining exercise performance? Eur J Sport Sci 18:13–24. https://doi.org/10.1080/17461391.2016.1252428
Hureau TJ, Broxterman RM, Weavil JC et al (2022) On the role of skeletal muscle acidosis and inorganic phosphates as determinants of central and peripheral fatigue: a 31P-MRS study. J Physiol 600:3096–81. https://doi.org/10.1113/jp283036
Iannetta D, Zhang J, Murias J, Aboodarda S (2022) Neuromuscular and perceptual mechanisms of fatigue accompanying task failure in response to moderate-, heavy-, severe-, and extreme-intensity cycling. J Appl Physiol 133:323–334. https://doi.org/10.1152/JAPPLPHYSIOL.00764.2021
Jessee M, Mattocks K, Buckner S et al (2018) Mechanisms of blood flow restriction: the new testament. Tech Orthop 33:72–79. https://doi.org/10.1097/BTO.0000000000000252
Kaufman MP, Hayes SG (2002) The exercise pressor reflex. Clin Auton Res 12:429–439. https://doi.org/10.1007/S10286-002-0059-1
Keramidas M, Kounalakis S, Geladas N (2012) The effect of interval training combined with thigh cuffs pressure on maximal and submaximal exercise performance. Clin Physiol Funct Imaging 32:205–213. https://doi.org/10.1111/j.1475-097x.2011.01078.x
Kluger B, Krupp L, Enoka R (2013) Fatigue and fatigability in neurologic illnesses: proposal for a unified taxonomy. Neurology 80:409–416. https://doi.org/10.1212/wnl.0b013e31827f07be
Krüger RL, Aboodarda SJ, Jaimes LM et al (2019) Cycling performed on an innovative ergometer at different intensities–durations in men: neuromuscular fatigue and recovery kinetics. Appl Physiol Nutr Metab 44:1320–1328. https://doi.org/10.1139/apnm-2018-0858
Loenneke JP, Wilson GJ, Wilson JM (2010) A mechanistic approach to blood flow occlusion. Int J Sports Med 31:1–4. https://doi.org/10.1055/S-0029-1239499
Mancora S (2010) Effort: Perception of. In: Encyclopedia of perception of effort. Los Angeles: Sage Publications Inc., Thousand Oaks
Manini T, Clark B (2009) Blood flow restricted exercise and skeletal muscle health. Exerc Sport Sci Rev 37:78–85. https://doi.org/10.1097/jes.0b013e31819c2e5c
McClean Z, Iannetta D, MacInnis M, Aboodarda SJ (2022) Shorter high-intensity cycling intervals reduce performance and perceived fatigability at work-matched but not task failure. Med Sci Sports Exerc 55:690–699. https://doi.org/10.1249/mss.0000000000003097
McClean Z, Young A, Pohl AJ et al (2023) Blood flow restriction during high-intensity interval cycling exacerbates psychophysiological responses to a greater extent in females than males. J Appl Physiol 134:596–609. https://doi.org/10.1152/japplphysiol.00567.2022
McCormack HM, Horne DJ, Sheather S (1988) Clinical applications of visual analogue scales: a critical review. Psychol Med 18:1007–1019. https://doi.org/10.1017/s0033291700009934
Micklewright D, St Clair Gibson A, Gladwell V, Al Salman V (2017) Development and validity of the rating-of-fatigue scale. Sports Med 47:2375–2393. https://doi.org/10.1007/s40279-017-0711-5
Mira J, Aboodarda SJ, Floreani M et al (2018) Effects of endurance training on neuromuscular fatigue in healthy active men. part I: Strength loss and muscle fatigue. Eur J Appl Physiol 118:2281–2293. https://doi.org/10.1007/s00421-018-3950-8
Mitchell E, Martin N, Turner M et al (2019) The combined effect of sprint interval training and postexercise blood flow restriction on critical power, capillary growth, and mitochondrial proteins in trained cyclists. J Appl Physiol Bethesda Md 126:51–59. https://doi.org/10.1152/japplphysiol.01082.2017
Norbury R, Smith SA, Burnley M et al (2022) The effect of elevated muscle pain on neuromuscular fatigue during exercise. Eur J Appl Physiol 122:113–126. https://doi.org/10.1007/S00421-021-04814-1
Patterson SD, Hughes L, Warmington S et al (2019) Blood flow restriction exercise: considerations of methodology, application, and safety. Front Physiol 10:533. https://doi.org/10.3389/fphys.2020.599592
Pollak KA, Swenson JD, Vanhaitsma TA et al (2014) Exogenously applied muscle metabolites synergistically evoke sensations of muscle fatigue and pain in human subjects. Exp Physiol 99:368–380. https://doi.org/10.1113/expphysiol.2013.075812
Pope Z, Willardson J, Schoenfeld B (2013) Exercise and blood flow restriction. J Strength Cond 27:2914–2926
Scott BR, Loenneke JP, Slattery KM, Dascombe BJ (2015) Exercise with blood flow restriction: an updated evidence-based approach for enhanced muscular development. Sports Med 45:313–325. https://doi.org/10.1007/s40279-014-0288-1
Secher N, Niels H, Amann M (2012) Human investigations into the exercise pressor reflex. Exp Physiol 97:59–69. https://doi.org/10.1113/expphysiol.2011.057679
Smith SA, Micklewright D, Winter SL, Mauger AR (2020) Muscle pain induced by hypertonic saline in the knee extensors decreases single-limb isometric time to task failure. Eur J Appl Physiol 120:2047–2058. https://doi.org/10.1007/S00421-020-04425-2
Takarada Y, Nakamura Y, Aruga S et al (2000) Rapid increase in plasma growth hormone after low-intensity resistance exercise with vascular occlusion. J Appl Physiol 88:61–65. https://doi.org/10.1152/JAPPL.2000.88.1.61
van Melick N, Meddeler BM, Hoogeboom TJ et al (2017) How to determine leg dominance: The agreement between self-reported and observed performance in healthy adults. PLOS ONE 12:e0189876. https://doi.org/10.1371/journal.pone.0189876
Warburton DER, Jamnik VK, Bredin SSD, Gledhill N (2011) The physical activity readiness questionnaire for everyone (PAR-Q+): english north america version. Health Fit J Can 4:18–20
Zhang J, Iannetta D, Alzeeby M et al (2021) Exercising muscle mass influences neuromuscular, cardiorespiratory, and perceptual responses during and following ramp-incremental cycling to task failure. Am J Physiol-Regul Integr Comp Physiol 321:R238–R249. https://doi.org/10.1152/ajpregu.00286.2020
Acknowledgments
We would like to thank the participants of this study. This project was funded by the Natural Sciences and Engineering Research Council of Canada (RGPIN-439 2020-07075).
Author information
Authors and Affiliations
Contributions
ZM, JZ, and SJA conceived and designed the research. ZM, MY, and SJA conducted experiments. ZM, JZ, MY and NK analyzed the data. ZM, JZ, MY and NK wrote/edited the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by Guido Ferretti.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
McClean, Z.J., Zhang, J., Khaledi, N. et al. Intermittent blood flow occlusion modulates neuromuscular, perceptual, and cardiorespiratory determinants of exercise tolerance during cycling. Eur J Appl Physiol 123, 2295–2306 (2023). https://doi.org/10.1007/s00421-023-05242-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-023-05242-z