Abstract
Purpose
We hypothesized exercise vasodilation would be greater in women due to nitric oxide synthase (NOS) and cyclooxygenase (COX) signaling.
Methods
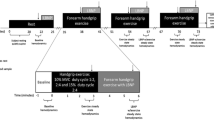
45 healthy adults (23 women, W, 22 men, M, 26 ± 1 years) completed two 10-min trials of dynamic forearm exercise at 15 % intensity. Forearm blood flow (FBF; Doppler ultrasound), arterial pressure (brachial catheter), and forearm lean mass were measured to calculate relative forearm vascular conductance (FVCrel) = FBF 100 mmHg−1 100 g−1 lean mass. Local intra-arterial infusion of L-NMMA or ketorolac acutely inhibited NOS and COX, respectively. In Trial 1, the first 5 min served as control exercise (CON), followed by 5 min of L-NMMA or ketorolac over the last 5 min of exercise. In Trial 2, the remaining drug was infused during 5–10 min, to achieve combined NOS–COX inhibition (double blockade, DB).
Results
Are mean ± SE. Women exhibited 29 % greater vasodilation in CON (ΔFVCrel, 19 ± 1 vs. 15 ± 1, p = 0.01). L-NMMA reduced ΔFVCrel (p < 0.001) (W: Δ −2.3 ± 1.3 vs. M: Δ −3.7 ± 0.8, p = 0.25); whereas, ketorolac modestly increased ΔFVCrel (p = 0.04) similarly between sexes (W: Δ 1.6 ± 1.1 vs. M: Δ 2.0 ± 1.6, p = 0.78). DB was also found to be similar between the sexes (p = 0.85).
Conclusion
These data clearly indicate women produce a greater exercise vasodilator response. Furthermore, contrary to experiments in animal models, these data are the first to demonstrate vascular control by NOS and COX is similar between sexes.






Similar content being viewed by others
Abbreviations
- ASAs:
-
Acetylsalicylic acid
- BMI:
-
Body mass index
- CON:
-
Control exercise
- COX:
-
Cyclooxygenase
- DEXA:
-
Dual-energy X-ray absorptiometry
- DB:
-
Double blockade, NOS–COX inhibition condition
- ECG:
-
Electrocardiogram
- EDHF:
-
Endothelium-derived hyperpolarizing factor
- FBF:
-
Forearm blood flow
- FVC:
-
Forearm vascular conductance
- KETO:
-
Ketorolac, COX inhibition condition
- L-NMMA:
-
L-NG-monomethyl arginine, NOS inhibition condition
- MAP:
-
Mean arterial pressure
- MBV:
-
Mean blood velocity
- MVC:
-
Maximal voluntary contraction
- NOS:
-
Nitric oxide synthase
- NSAIDs:
-
Non-steroidal anti-inflammatory drugs acetylsalicylic acid
References
Arain FA, Kuniyoshi FH, Abdalrhim AD, Miller VM (2009) Sex/gender medicine. The biological basis for personalized care in cardiovascular medicine. Circ J 73:1774–1782
Bagher P, Segal SS (2011) Regulation of blood flow in the microcirculation: role of conducted vasodilation. Acta Physiol 202:271–284. doi:10.1111/j.1748-1716.2010.02244.x
Blauwet LA, Redberg RF (2007) The role of sex-specific results reporting in cardiovascular disease. Cardiol Rev 15:275–278. doi:10.1097/CRD.0b013e318158b45b
Boushel R, Langberg H, Gemmer C et al (2002) Combined inhibition of nitric oxide and prostaglandins reduces human skeletal muscle blood flow during exercise. J Physiol 543:691–698. doi:10.1113/jphysiol.2002.021477
Casey DP, Joyner MJ (2009) NOS inhibition blunts and delays the compensatory dilation in hypoperfused contracting human muscles. J Appl Physiol 107:1685–1692. doi:10.1152/japplphysiol.00680.2009
Casey DP, Madery BD, Curry TB et al (2010) Nitric oxide contributes to the augmented vasodilatation during hypoxic exercise. J Physiol 588:373–385. doi:10.1113/jphysiol.2009.180489
Casey DP, Curry TB, Wilkins BW, Joyner MJ (2011) Nitric oxide-mediated vasodilation becomes independent of -adrenergic receptor activation with increased intensity of hypoxic exercise. J Appl Physiol 110:687–694. doi:10.1152/japplphysiol.00787.2010
Casey DP, Shepherd JRA, Joyner MJ (2013) Sex and vasodilator responses to hypoxia at rest and during exercise. J Appl Physiol 116:927–936. doi:10.1152/japplphysiol.00409.2013
Chen Y, Dangardt F, Osika W et al (2012) Atherosclerosis. Atherosclerosis 220:269–274. doi:10.1016/j.atherosclerosis.2011.10.030
Crecelius AR, Kirby BS, Voyles WF, Dinenno FA (2011) Augmented skeletal muscle hyperaemia during hypoxic exercise in humans is blunted by combined inhibition of nitric oxide and vasodilating prostaglandins. J Physiol 589:3671–3683. doi:10.1113/jphysiol.2011.209486
Crecelius AR, Kirby BS, Luckasen GJ et al (2013) Mechanisms of rapid vasodilation after a brief contraction in human skeletal muscle. AJP: Heart Circ Physiol 305:H29–H40. doi:10.1152/ajpheart.00298.2013
Crecelius AR, Luckasen GJ, Larson DG, Dinenno FA (2014) KIR channel activation contributes to onset and steady-state exercise hyperemia in humans. AJP: Heart Circ Physiol 307:H782–H791. doi:10.1152/ajpheart.00212.2014
Dinenno FA, Joyner MJ (2004) Combined NO and PG inhibition augments alpha-adrenergic vasoconstriction in contracting human skeletal muscle. AJP: Heart Circ Physiol 287:H2576–H2584. doi:10.1152/ajpheart.00621.2004
Duffy SJ, New G, Tran BT et al (1999) Relative contribution of vasodilator prostanoids and NO to metabolic vasodilation in the human forearm. Am J Physiol 276:H663–H670
Dyke CK, Proctor DN, Dietz NM, Joyner MJ (1995) Role of nitric oxide in exercise hyperaemia during prolonged rhythmic handgripping in humans. J Physiol 488(Pt 1):259–265
Fadel PJ, Zhao W, Thomas GD (2003) Impaired vasomodulation is associated with reduced neuronal nitric oxide synthase in skeletal muscle of ovariectomized rats. J Physiol 549:243–253. doi:10.1113/jphysiol.2003.038828
Garten RS, Groot HJ, Rossman MJ et al (2014) The role of muscle mass in exercise-induced hyperemia. J Appl Physiol 116:1204–1209. doi:10.1152/japplphysiol.00103.2014
Gavin KM, Seals DR, Silver AE, Moreau KL (2009) Vascular endothelial estrogen receptor α is modulated by estrogen status and related to endothelial function and endothelial nitric oxide synthase in healthy women. J Clin Endocrinol Metab 94:3513–3520. doi:10.1210/jc.2009-0278
Gonzales JU, Thompson BC, Thistlethwaite JR et al (2007) Forearm blood flow follows work rate during submaximal dynamic forearm exercise independent of sex. J Appl Physiol 103:1950–1957. doi:10.1152/japplphysiol.00452.2007
Hart EC, Charkoudian N, Wallin BG et al (2011) Sex and ageing differences in resting arterial pressure regulation: the role of the -adrenergic receptors. J Physiol 589:5285–5297. doi:10.1113/jphysiol.2011.212753
Heinonen I, Ilkka H, Saltin B et al (2011) Skeletal muscle blood flow and oxygen uptake at rest and during exercise in humans: a pet study with nitric oxide and cyclooxygenase inhibition. AJP: Heart Circ Physiol 300:H1510–H1517. doi:10.1152/ajpheart.00996.2010
Hellsten Y, Nyberg M, Jensen LG, Mortensen SP (2012) Vasodilator interactions in skeletal muscle blood flow regulation. J Physiol 590:6297–6305. doi:10.1113/jphysiol.2012.240762
Herr MD, Hogeman CS, Koch DW et al (2010) A real-time device for converting Doppler ultrasound audio signals into fluid flow velocity. AJP: Heart Circ Physiol 298:H1626–H1632. doi:10.1152/ajpheart.00713.2009
Hillig T, Krustrup P, Fleming I et al (2002) Cytochrome P450 2C9 plays an important role in the regulation of exercise-induced skeletal muscle blood flow and oxygen uptake in humans. J Physiol 546:307–314. doi:10.1113/jphysiol.2002.030833
Huang A, Sun D, Carroll MA et al (2001) EDHF mediates flow-induced dilation in skeletal muscle arterioles of female eNOS-KO mice. AJP: Heart Circ Physiol 280:H2462–H2469
Huxley VH (2007) Sex and the cardiovascular system: the intriguing tale of how women and men regulate cardiovascular function differently. AJP: Adv Physiol Edu 31:17–22. doi:10.1152/advan.00099.2006
Jackson WF (2005) Potassium channels in the peripheral microcirculation. Microcirculation 12:113–127. doi:10.1080/10739680590896072
Karamouzis M, Karamouzis I, Vamvakoudis E et al (2001a) The response of muscle interstitial prostaglandin E2(PGE2), prostacyclin I2(PGI2) and thromboxane A2(TXA2) levels during incremental dynamic exercise in humans determined by in vivo microdialysis. Prostaglandins Leukot Essent Fatty Acids 64:259–263. doi:10.1054/plef.2001.0269
Karamouzis M, Langberg H, Skovgaard D et al (2001b) In situ microdialysis of intramuscular prostaglandin and thromboxane in contracting skeletal muscle in humans. Acta Physiol Scand 171:71–76. doi:10.1046/j.1365-201X.2001.00775.x
Khan LH, Rosenfeld CR, Liu XT, Magness RR (2010) Regulation of the cGMP-cPKG pathway and large-conductance Ca2+-activated K+ channels in uterine arteries during the ovine ovarian cycle. AJP: Endocrinol Metab 298:E222–E228. doi:10.1152/ajpendo.00375.2009
Kneale BJ, Chowienczyk PJ, Brett SE et al (2000) Gender differences in sensitivity to adrenergic agonists of forearm resistance vasculature. J Am Coll Cardiol 36:1233–1238
Laughlin MH, Davis MJ, Secher NH et al (2012) Peripheral circulation. Compr Physiol 2:321–447. doi:10.1002/cphy.c100048
Limberg JK, Eldridge MW, Proctor LT et al (2010) Adrenergic control of blood flow during exercise: effect of sex and menstrual phase. J Appl Physiol 109:1360–1368. doi:10.1152/japplphysiol.00518.2010
Limberg JK, Harrell JW, Johansson RE et al (2013) Microvascular function in younger adults with obesity and metabolic syndrome: role of oxidative stress. AJP: Heart Circ Physiol 305:H1230–H1237. doi:10.1152/ajpheart.00291.2013
Limberg JK, Morgan BJ, Sebranek JJ et al (2014) Neural control of blood flow during exercise in human metabolic syndrome. Exp Physiol 99:1191–1202. doi:10.1113/expphysiol.2014.078048
Markwald RR, Kirby BS, Crecelius AR et al (2011) Combined inhibition of nitric oxide and vasodilating prostaglandins abolishes forearm vasodilatation to systemic hypoxia in healthy humans. J Physiol 589:1979–1990. doi:10.1113/jphysiol.2011.205013
Miller VM (2010) Sex-based differences in vascular function. Women’s Health 6:737–752. doi:10.2217/whe.10.53
Miller VM (2014) Why are sex and gender important to basic physiology and translational and individualized medicine? AJP: Heart Circ Physiol 306:H781–H788. doi:10.1152/ajpheart.00994.2013
Miner JA, Martini ER, Smith MM et al (2011) Short-term oral progesterone administration antagonizes the effect of transdermal estradiol on endothelium-dependent vasodilation in young healthy women. AJP: Heart Circ Physiol 301:H1716–H1722. doi:10.1152/ajpheart.00405.2011
Moore AW, Bearden SE, Segal SS (2010) Regional activation of rapid onset vasodilatation in mouse skeletal muscle: regulation through -adrenoreceptors. J Physiol 588:3321–3331. doi:10.1113/jphysiol.2010.193672
Moro P-J, Flavian A, Jacquier A et al (2011) Gender differences in response to cold pressor test assessed with velocity-encoded cardiovascular magnetic resonance of the coronary sinus. J Cardiovasc Magn Reson 13:54. doi:10.1186/1532-429X-13-54
Mortensen SP, Gonzalez-Alonso J, Damsgaard R et al (2007) Inhibition of nitric oxide and prostaglandins, but not endothelial-derived hyperpolarizing factors, reduces blood flow and aerobic energy turnover in the exercising human leg. J Physiol 581:853–861. doi:10.1113/jphysiol.2006.127423
Mortensen SP, Nyberg M, Thaning P et al (2009) Adenosine contributes to blood flow regulation in the exercising human leg by increasing prostaglandin and nitric oxide formation. Hypertension 53:993–999. doi:10.1161/HYPERTENSIONAHA.109.130880
Paffenbarger RS Jr, Blair SN, Lee I-M, Hyde RT (1993) Measurement of physical activity to assess health effects in free-living populations. Med Sci Sports Exerc 25:60–70
Pak KJ, Geary GG, Duckles SP, Krause DN (2002) Male–female differences in the relative contribution of endothelial vasodilators released by rat tail artery. Life Sci 71:1633–1642
Parker BA, Smithmyer SL, Pelberg JA et al (2007) Sex differences in leg vasodilation during graded knee extensor exercise in young adults. J Appl Physiol 103:1583–1591. doi:10.1152/japplphysiol.00662.2007
Parker BA, Smithmyer SL, Pelberg JA et al (2008) Sex-specific influence of aging on exercising leg blood flow. J Appl Physiol 104:655–664. doi:10.1152/japplphysiol.01150.2007
Rogers J, Sheriff DD (2004) Role of estrogen in nitric oxide- and prostaglandin-dependent modulation of vascular conductance during treadmill locomotion in rats. J Appl Physiol 97:756–763. doi:10.1152/japplphysiol.00115.2004
Saito Y, Iemitsu M, Otsuki T et al (2008) Gender differences in brachial blood flow during fatiguing intermittent handgrip. Med Sci Sports Exerc 40:684–690. doi:10.1249/MSS.0b013e3181614327
Schrage WG, Joyner MJ, Dinenno FA (2004) Local inhibition of nitric oxide and prostaglandins independently reduces forearm exercise hyperaemia in humans. J Physiol 557:599–611. doi:10.1113/jphysiol.2004.061283
Schrage WG, Wilkins BW, Johnson CP et al (2010) Roles of nitric oxide synthase and cyclooxygenase in leg vasodilation and oxygen consumption during prolonged low-intensity exercise in untrained humans. J Appl Physiol 109:768–777. doi:10.1152/japplphysiol.00326.2010
Scotland RS, Madhani M, Chauhan S et al (2005) Investigation of vascular responses in endothelial nitric oxide synthase/cyclooxygenase-1 double-knockout mice: key role for endothelium-derived hyperpolarizing factor in the regulation of blood pressure in vivo. Circulation 111:796–803. doi:10.1161/01.CIR.0000155238.70797.4E
Shaw LJ, Bairey Merz CN, Pepine CJ et al (2006) Insights from the NHLBI-sponsored Women’s Ischemia Syndrome Evaluation (WISE) Study part I: gender differences in traditional and novel risk factors, symptom evaluation, and gender-optimized diagnostic strategies. J Am Coll Cardiol 47:S4–S20. doi:10.1016/j.jacc.2005.01.072
Shoemaker JK, Naylor HL, Pozeg ZI, Hughson RL (1996) Failure of prostaglandins to modulate the time course of blood flow during dynamic forearm exercise in humans. J Appl Physiol 81:1516–1521
Shoemaker JK, Halliwill JR, Hughson RL, Joyner MJ (1997) Contributions of acetylcholine and nitric oxide to forearm blood flow at exercise onset and recovery. AJP: Heart Circ Physiol 273:H2388–H2395
Taylor MK, Larson GE, Hiller Lauby MD et al (2014) Sex differences in cardiovascular and subjective stress reactions: prospective evidence in a realistic military setting. Stress 17:70–78. doi:10.3109/10253890.2013.869208
Villar IC, Hobbs AJ, Ahluwalia A (2008) Sex differences in vascular function: implication of endothelium-derived hyperpolarizing factor. J Endocrinol 197:447–462. doi:10.1677/JOE-08-0070
Wilkins BW, Pike TL, Martin EA et al (2008) Exercise intensity-dependent contribution of beta-adrenergic receptor-mediated vasodilatation in hypoxic humans. J Physiol 586:1195–1205. doi:10.1113/jphysiol.2007.144113
Wray DW, Witman MAH, Ives SJ et al (2011) Progressive handgrip exercise: evidence of nitric oxide-dependent vasodilation and blood flow regulation in humans. AJP: Heart Circ Physiol 300:H1101–H1107. doi:10.1152/ajpheart.01115.2010
Acknowledgments
We would like to thank the subjects for their time and effort. We would also like to acknowledge Meghan Crain, Garrett Peltonen, Joshua Trierweiler, and Cameron Rousseau, for their help in data collection. The Clinical and Translational Science Award (CTSA) program, through the NIH National Center for Advancing Translational Sciences (NCATS), supported the described project grant UL1TR000427. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. Funding: NIH HL-105820.
Conflict of interest
The authors have no conflicts of interests and nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Massimo Pagani.
Rights and permissions
About this article
Cite this article
Kellawan, J.M., Johansson, R.E., Harrell, J.W. et al. Exercise vasodilation is greater in women: contributions of nitric oxide synthase and cyclooxygenase. Eur J Appl Physiol 115, 1735–1746 (2015). https://doi.org/10.1007/s00421-015-3160-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-015-3160-6