Abstract
Purpose
The aim of this study was to compare activation of cellular signaling pathways regulating protein synthesis and glucose uptake in skeletal muscle between resistance and endurance exercise. Moreover, the effect of resistance exercise volume was examined.
Methods
Three groups of male volunteers (26 ± 3 years) were examined: 5 × 10 repetition maximum (RM) resistance exercise (RE) with leg press device (5 × 10 RE; n = 8), 10 × 10 RE (n = 11), and endurance exercise (strenuous 50-min walking with extra load on a treadmill; EE; n = 8). Muscle biopsies were obtained from m.vastus lateralis 30 min pre- and post-exercise.
Results
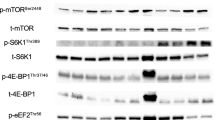
Downstream markers of mTORC1, p-p70S6KThr421/Ser424 and p-rpS6Ser240/244, increased more after 10 × 10 RE than after 5 × 10 RE (p < 0.05) and EE (p < 0.01–0.001). Exercise-induced changes in p-IRS-ISer636/639 that inhibit IRS-I signaling via negative feedback from hyperactivated mTORC1 signaling were greater (p < 0.05) after 10 × 10 RE compared with 5 × 10 RE and EE. The changes in energy sensor p-AMPKαThr172 were greater after 10 × 10 RE and EE (p < 0.05–0.01) than after 5 × 10 RE. A major regulator of glucose uptake in muscle, p-AS160Thr642, increased more after 10 × 10 RE than after 5 × 10 RE (p < 0.01) and EE (p < 0.05).
Conclusion
10 × 10 RE induced greater activation of important signaling proteins regulating glucose uptake (p-AS160) and protein synthesis (p-p70S6K, p-rpS6) than 5 × 10 RE and EE. The present findings further suggest that, especially after 10 × 10 RE, IRS-I signaling is downregulated and that AS160 is activated through AMPK signaling pathway.





Similar content being viewed by others
Abbreviations
- Akt:
-
AKT8 virus oncogene cellular homolog
- AMPK:
-
AMP-activated protein kinase
- AS160:
-
Akt substrate of 160 kDa
- CaMKII:
-
Ca2+/calmodulin-dependent protein kinase II
- EE:
-
Endurance exercise
- eEF:
-
Eukaryotic elongation factor
- Erk:
-
Extracellular signal-regulated kinase
- GLUT4:
-
Glucose transporter type 4
- HR:
-
Heart rate
- IRS-1:
-
Insulin receptor substrate 1
- LKB1:
-
Liver kinase B1
- MAPK:
-
Mitogen-activated protein kinase
- mTORC1:
-
Mechanistic target of rapamycin complex 1
- p70S6K:
-
p70 ribosomal S6 kinase
- PI3K:
-
Phosphoinositide 3-kinase
- RE:
-
Resistance exercise
- RM:
-
Repetition maximum
- rpS6:
-
Ribosomal protein S6
References
Camera DM, Edge J, Short MJ, Hawley JA, Coffey VG (2010) Early time course of Akt phosphorylation after endurance and resistance exercise. Med Sci Sports Exerc 42:1843–1852. doi:10.1249/MSS.0b013e3181d964e4
Coffey VG, Hawley JA (2007) The molecular bases of training adaptation. Sports Med 37:737–763
Colbran RJ, Fong YL, Schworer CM, Soderling TR (1988) Regulatory interactions of the calmodulin-binding, inhibitory, and autophosphorylation domains of Ca2 +/calmodulin-dependent protein kinase II. J Biol Chem 263:18145–18151
Cully M, Genevet A, Warne P et al (2010) A role for p38 stress-activated protein kinase in regulation of cell growth via TORC1. Mol Cell Biol 30:481–495. doi:10.1128/MCB.00688-09
Deshmukh A, Coffey VG, Zhong Z, Chibalin AV, Hawley JA, Zierath JR (2006) Exercise-induced phosphorylation of the novel Akt substrates AS160 and filamin A in human skeletal muscle. Diabetes 55:1776–1782. doi:10.2337/db05-1419
Dreyer HC, Drummond MJ, Glynn EL et al (2008) Resistance exercise increases human skeletal muscle AS160/TBC1D4 phosphorylation in association with enhanced leg glucose uptake during postexercise recovery. J Appl Physiol 105:1967–1974. doi:10.1152/japplphysiol.90562.2008
Hawley SA, Boudeau J, Reid JL et al (2003) Complexes between the LKB1 tumor suppressor, STRAD alpha/beta and MO25 alpha/beta are upstream kinases in the AMP-activated protein kinase cascade. J Biol 2:28. doi:10.1186/1475-4924-2-28
Howlett KF, Sakamoto K, Garnham A, Cameron-Smith D, Hargreaves M (2007) Resistance exercise and insulin regulate AS160 and interaction with 14-3-3 in human skeletal muscle. Diabetes 56:1608–1614. doi:10.2337/db06-1398
Hulmi JJ, Walker S, Ahtiainen JP, Nyman K, Kraemer WJ, Hakkinen K (2012) Molecular signaling in muscle is affected by the specificity of resistance exercise protocol. Scand J Med Sci Sports 22:240–248. doi:10.1111/j.1600-0838.2010.01198.x
Khamzina L, Veilleux A, Bergeron S, Marette A (2005) Increased activation of the mammalian target of rapamycin pathway in liver and skeletal muscle of obese rats: possible involvement in obesity-linked insulin resistance. Endocrinology 146:1473–1481. doi:10.1210/en.2004-0921
Knebel A, Haydon CE, Morrice N, Cohen P (2002) Stress-induced regulation of eukaryotic elongation factor 2 kinase by SB 203580-sensitive and -insensitive pathways. Biochem J 367:525–532. doi:10.1042/BJ20020916
Kramer HF, Witczak CA, Fujii N et al (2006) Distinct signals regulate AS160 phosphorylation in response to insulin, AICAR, and contraction in mouse skeletal muscle. Diabetes 55:2067–2076. doi:10.2337/db06-0150
Krieger JW (2010) Single vs. multiple sets of resistance exercise for muscle hypertrophy: a meta-analysis. J Strength Cond Res 24:1150–1159. doi:10.1519/JSC.0b013e3181d4d436
Kumar V, Atherton PJ, Selby A et al (2012) Muscle protein synthetic responses to exercise: effects of age, volume, and intensity. J Gerontol A Biol Sci Med Sci 67:1170–1177. doi:10.1093/gerona/gls141
Magnuson B, Ekim B, Fingar DC (2012) Regulation and function of ribosomal protein S6 kinase (S6 K) within mTOR signalling networks. Biochem J 441:1–21. doi:10.1042/BJ20110892
Ozes ON, Akca H, Mayo LD et al (2001) A phosphatidylinositol 3-kinase/Akt/mTOR pathway mediates and PTEN antagonizes tumor necrosis factor inhibition of insulin signaling through insulin receptor substrate-1. Proc Natl Acad Sci USA 98:4640–4645. doi:10.1073/pnas.051042298
Pende M, Um SH, Mieulet V et al (2004) S6K1(-/-)/S6K2(-/-) mice exhibit perinatal lethality and rapamycin-sensitive 5′-terminal oligopyrimidine mRNA translation and reveal a mitogen-activated protein kinase-dependent S6 kinase pathway. Mol Cell Biol 24:3112–3124
Peterson MD, Rhea MR, Alvar BA (2005) Applications of the dose-response for muscular strength development: a review of meta-analytic efficacy and reliability for designing training prescription. J Strength Cond Res 19:950–958. doi:10.1519/R-16874.1
Reed ME, Ben-Ezra V, Biggerstaff KD, Nichols DL (2012) The effects of two bouts of high- and low-volume resistance exercise on glucose tolerance in normoglycemic women. J Strength Cond Res 26:251–260. doi:10.1519/JSC.0b013e318218dea3
Richter EA, Ruderman NB (2009) AMPK and the biochemistry of exercise: implications for human health and disease. Biochem J 418:261–275. doi:10.1042/BJ20082055
Robbins DW, Marshall PW, McEwen M (2012) The effect of training volume on lower-body strength. J Strength Cond Res 26:34–39. doi:10.1519/JSC.0b013e31821d5cc4
Rockl KS, Witczak CA, Goodyear LJ (2008) Signaling mechanisms in skeletal muscle: acute responses and chronic adaptations to exercise. IUBMB Life 60:145–153. doi:10.1002/iub.21
Rose AJ, Richter EA (2009) Regulatory mechanisms of skeletal muscle protein turnover during exercise. J Appl Physiol 106:1702–1711. doi:10.1152/japplphysiol.91375.2008
Roux PP, Ballif BA, Anjum R, Gygi SP, Blenis J (2004) Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase. Proc Natl Acad Sci USA 101:13489–13494. doi:10.1073/pnas.0405659101
Roux PP, Shahbazian D, Vu H et al (2007) RAS/ERK signaling promotes site-specific ribosomal protein S6 phosphorylation via RSK and stimulates cap-dependent translation. J Biol Chem 282:14056–14064. doi:10.1074/jbc.M700906200
Ruvinsky I, Sharon N, Lerer T et al (2005) Ribosomal protein S6 phosphorylation is a determinant of cell size and glucose homeostasis. Genes Dev 19:2199–2211. doi:10.1101/gad.351605
Ryazanov AG, Shestakova EA, Natapov PG (1988) Phosphorylation of elongation factor 2 by EF-2 kinase affects rate of translation. Nature 334:170–173. doi:10.1038/334170a0
Sanchez AM, Candau RB, Csibi A, Pagano AF, Raibon A, Bernardi H (2012) The role of AMP-activated protein kinase in the coordination of skeletal muscle turnover and energy homeostasis. Am J Physiol Cell Physiol 303:C475–C485. doi:10.1152/ajpcell.00125.2012
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307:1098–1101. doi:10.1126/science.1106148
Schweitzer GG, Arias EB (2012) Cartee GD (2012) Sustained postexercise increases in AS160 Thr642 and Ser588 phosphorylation in skeletal muscle without sustained increases in kinase phosphorylation. J Appl Physiol 113:1852–1861. doi:10.1152/japplphysiol.00619.2012
Sriwijitkamol A, Coletta DK, Wajcberg E et al (2007) Effect of acute exercise on AMPK signaling in skeletal muscle of subjects with type 2 diabetes: a time-course and dose-response study. Diabetes 56:836–848. doi:10.2337/db06-1119
Terzis G, Spengos K, Mascher H, Georgiadis G, Manta P, Blomstrand E (2010) The degree of p70 S6 k and S6 phosphorylation in human skeletal muscle in response to resistance exercise depends on the training volume. Eur J Appl Physiol 110:835–843. doi:10.1007/s00421-010-1527-2
Walker DK, Dickinson JM, Timmerman KL et al (2011) Exercise, amino acids, and aging in the control of human muscle protein synthesis. Med Sci Sports Exerc 43:2249–2258. doi:10.1249/MSS.0b013e318223b037
Walker S, Hulmi JJ, Wernbom M et al (2013) Variable resistance training promotes greater fatigue resistance but not hypertrophy versus constant resistance training. Eur J Appl Physiol 113:2233–2244. doi:10.1007/s00421-013-2653-4
Woods A, Johnstone SR, Dickerson K et al (2003) LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr Biol 13:2004–2008
Acknowledgments
The authors thank Ms. Sheila Gagnon, MSc. Opri Jokelainen, Dr. Ritva Taipale, Mr. Risto Puurtinen, and Mrs. Aila Ollikainen for their work in data collection and analyses. This work was supported by a grant from the Scientific Advisory Board for Defence and David Sports Ltd.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Martin Flueck.
Rights and permissions
About this article
Cite this article
Ahtiainen, J.P., Walker, S., Silvennoinen, M. et al. Exercise type and volume alter signaling pathways regulating skeletal muscle glucose uptake and protein synthesis. Eur J Appl Physiol 115, 1835–1845 (2015). https://doi.org/10.1007/s00421-015-3155-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-015-3155-3