Abstract
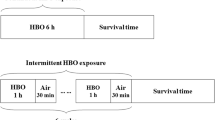
Intermittent exposure to air is used as a protective strategy against hyperbaric O2 (HBO2) toxicity. Little is known about optimal intermittent exposure schedules and the mechanism of protection. In this study, we examined the role of antioxidant enzymes, and inflammatory cytokines in the mechanism of HBO2 tolerance by intermittent air breaks. One group of rats was exposed continuously to 282 kPa O2 until death. Other groups were exposed to 30, 60, and 120 min intervals of HBO2 with different numbers of intermittent 30 min air breaks (1–12 breaks). After the final break, animals were exposed to HBO2 until death. In a separate experiment, animals were sacrificed before terminal exposure and lung tissues were collected for analysis of gene expression. Two intermittent schedules with 6 h cumulative O2 time (30/30 and 60/30 min schedules) were compared with continuous exposure to HBO2 for 6 h and with intermittent exposure of 8 h (120/30 min schedule) duration. Continuous exposure resulted in activation of inflammatory cytokine TNF-α and IL-1β mRNA expression, an increase in lung protein nitration and activation of inducible NOS (iNOS) mRNA. Inflammatory response was not observed at intermittent exposures of the same cumulative O2 time duration (30/30 and 60/30 min schedule). Expression of heme oxygenase-1 (HO-1) mRNA was significantly increased in all exposure groups while manganese superoxide dismutase (MnSOD) mRNA expression was increased only in continuous and 120/30 exposure groups. Results show that intermittent exposure to air protects against pulmonary HBO2 toxicity by inhibiting inflammation. The mechanism of inhibition may involve the antiinflammatory and antioxidative effect of HO-1 but some other mechanisms may also be involved in protection by intermittent air breaks.






Similar content being viewed by others
References
Arieli R, Gutterman A (1997) Recovery time constant in central nervous system O2 toxicity. Eur J Appl Physiol 75:182–187
Arieli R, Yalov A, Goldenshluger A (2002) Modeling pulmonary and CNS O(2) toxicity and estimation of parameters for humans. J Appl Physiol 92:248–256
Camhi S, Alam J, Otterbein L, Sylvester SL, Choi AMK (1995) Induction of heme oxygenase-1 gene expression by lipopolysaccharide is mediated by AP-1 activation. Am J Respir Cell Mol Biol 13:387–398
Chavko M, McCarron RM (2006) Extension of brain tolerance to hyperbaric O2 by intermittent air breaks is related to the time of CBF increase. Brain Res 1084:196–201
Choi AMK, Alam J (1996) Heme oxygenase-1: function, regulation and implication of a novel stress inducible protein in oxidant-induced lung injury. Am J Resp Cell Mol Biol 15:9–19
Clark JM (1993) Oxygen toxicity. In: Bennett PB, Elliot DH (eds) The physiology and medicine of diving, 4th edn. Saunders, London, pp 121–169
Clark JM, Lambertsen CJ (1971) Pulmonary oxygen toxicity. A Rev Pharm Rev 23:37–133
Clark JM, Lambertsen CJ, Gelfand R, Troxel AB (2006) Optimization of oxygen tolerance extension in rats by intermittent exposure. J Appl Physiol 100:869–879
Clerch LB (2000) Post-transcriptional regulation of lung antioxidant enzyme gene expression. Ann NY Acad Sci 899:103–111
Crapo JD (1986) Morphologic changes in pulmonary oxygen toxicity. Am Rev Physiol 48:721–731
Das KC, Lewis-Molock Y, White CW (1995) Thiol modulation of TNF-α and IL-1 induced MnSOD gene expression and activation of NF-kB. Mol Cell Biochem 148:45–57
Fink MP (2002) Role of reactive oxygen and nitrogen species in acute respiratory distress syndrome. Curr Opin Crit Care 8:6–11
Geiger M, Stone A, Mason SN, Oldham KT, Guice KS (1997) Differential nitric oxide production by microvascular and macrovascular endothelial cells. Am J Physiol 273:L275–L281
Haddad IY, Zhu S, Ischiropoulos H, Matalon S (1996) Nitration of surfactant protein A results in decreased ability to aggregate lipids. Am J Physiol Lung Cell Mol Physiol 270:L281–L288
Hall DA (1967) The influence of the systematic fluctuation of pO2 upon the nature and rate of development of oxygen toxicity in guinea pigs. Master’s thesis. University of Pensylvania
Harabin AL, Survanshi SS, Weathersby PK, Hays JR, Homer LD (1988) The modulation of oxygen toxicity by intermittent exposure. Toxicol Appl Pharmacol 93:298–311
Harabin AL, Braisted JC, Flynn ET (1990) Response of antioxidant enzymes to intermittent and continuous hyperbaric oxygen. J Appl Physiol 69:328–335
Hesse AK, Dorger M, Kupatt C, Krombach F (2004) Proinflammatory role of inducible nitric oxide synthase in acute hyperoxic lung injury. Respir Res 5:11
Hierholzer C, Menezes JM, Ungeheuer A, Billiar TR, Tweardy DJ, Harbrecht BG (2002) A nitric oxide scavenger protects against pulmonary inflammation following hemorrhagic shock. Shock 17:98–103
Ho YS, Dey MS, Crapo JD (1996) Antioxidant enzyme expression in rat lung during hyperoxia. Am J Physiol 270:L810–L818
Huang T-Y, Tsai P-S, Wang T-Y, Huang C-L, Huang C-J (2005) Hyperbaric oxygen attenuation of lipopolysaccharide-induced acute lung injury involves heme oxygenase-1. Acta Anesthesiol Scand 49:1293–1301
Kobayashi H, Hataishi R, Mitsufuji H (2001) Antiinflammatory properties of inducible nitric oxide synthase in acute hyperoxic lung injury. Am J Resp Cell Mol Biol 24:390–397
Lee PJ, Alam J, Sylvester SL, Inamdar N, Otterbein L, Choi AMK (1996) Regulation of heme oxygenase-1 expression in vivo and in vitro in hyperoxic lung injury. Am J Respir Cell Mol Biol 14:556–568
Lentsch AB, Czermak BJ, Bless NM, Ward PA (1998) NF-kappaB activation during IgG immune complex-induced lung injury: requirements for TNF-alpha and IL-1beta but not complement. Am J Pathol 152:1327–1336
Nagata K, Iwasaki Y, Yamada T, Yuba T, Kono K, Hosogi S, Ohsugi S, Kuwahara H, Marunaka Y (2007) Overexpression of manganese superoxide dismutase by N-acetylcysteine in hyperoxic lung injury. Respir Med 101:800–807
Otterbein LE, Choi AMK (2000) Heme oxygenase: colors of defense against cellular stress. Am J Physiol Lung Cell Mol Physiol 279:L1029–L1037
Peng HB, Rajavashisth TB, Libby P, Liao JK (1995) Nitric oxide inhibits macrophage-colony stimulating factor gene transcription in vascular endothelial cells. J Biol Chem 270:17050–17055
Rogers RJ, Monnier JM, Nick HS (2001) Tumor necrosis factor-α selectively induces MnSOD expression via mitochondria-to-nucleus signaling, whereas interleukin-1β utilizes an alternative pathway. J Biol Chem 276:20419–20427
Tsan M (1997) Superoxide dismutase and pulmonary oxygen toxicity. Proc Soc Exp Biol Med 214:107–113
Tsan M, White JE, Caska B, Epstein CJ, Lee CY (1998) Susceptibility of heterozygous MnSOD gene knockout mice to oxygen toxicity. Am J Respir Cell Mol Biol 19:114–120
Warner BB, Stuart L, Gebb S, Wispe JR (1996) Redox regulation of manganese superoxide dismutase. Am J Physiol 271:L150–L158
Zaher TE, Miller EJ, Morrow DMP, Javdan M, Mantell LL (2007) Hyperoxia-induced signal transduction pathways in pulmonary epithelial cells. Free Rad Biol Med 42:897–908
Acknowledgments
This work was supported by Office of Naval Research Work Unit #62236N.04122.1343.A0502. The opinions expressed herein are those of the authors and do not reflect the official policy of the Department of Navy, Department of Defense, of the US Government.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chavko, M., Mahon, R.T. & McCarron, R.M. Mechanisms of protection against pulmonary hyperbaric O2 toxicity by intermittent air breaks. Eur J Appl Physiol 102, 525–532 (2008). https://doi.org/10.1007/s00421-007-0611-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-007-0611-8