Abstract
Objective
The mechanisms of action of arsenic in the development of lung cancer are still not yet elucidated. Considering the relationship between arsenic and squamous cell carcinomas of the skin, we hypothesized that arsenic exposure may be more closely associated with squamous cell carcinoma of the lung.
Methods
A comprehensive histopathological database and a detailed job-exposure matrix developed for former German uranium miners with exposure to arsenic, radon, and quartz were analyzed to quantitatively assess the effect of arsenic regarding cell type of lung cancer. The distributions of major lung cancer cell types in 1,786 German uranium miners were associated with levels of arsenic exposure under control for the other lung carcinogens. To evaluate the arsenic effects in association with a frequent occupational lung disease in miners stratification by silicosis was performed.
Results
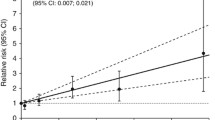
There was an arsenic-related increase of the proportion of squamous cell carcinoma of the lung but restricted to miners without silicosis. The increase was found at all levels of co-exposure to radon and quartz dust. In miners with silicosis, the proportion of adenocarcinoma increased with rising arsenic exposure. Arsenic exposure was associated with non-small cell lung cancer. Silicosis turned out as major determinant of the cell type related with arsenic.
Conclusion
These results indicate a cell type characteristic effect of arsenic in the development of lung cancer.

Similar content being viewed by others
References
Andrew AS, Warren AJ, Barchowsky A et al (2003) Genomic and proteomic profiling of responses to toxic metals in human lung cells. Environ Health Perspect 111:825–838
Axelson O, Dahlgren E, Jansson CD et al (1978) Arsenic exposure and mortality: a case-referent study from a Swedish copper smelter. Br J Ind Med 35:8–15
Bauer H-D (2000) Studie zur retrospektiven Analyse der Belastungssituation im Uranerzbergbau der ehemaligen SDAG Wismut mit Ausnahme der Strahlenbelastung für die Zeit von 1946 bis 1990. HVBG, Sankt Augustin
Borczuk AC, Gorenstein L, Walter KL et al (2003) Non-small-cell lung cancer molecular signatures recapitulate lung developmental pathways. Am J Pathol 163:1949–1960
Cavigelli M, Li WW, Lin AN et al (1996) The tumor promoter arsenite stimulates AP-1 activity by inhibiting a JNK phosphatase. EMBO J 15:6269–6279
Centeno JA, Mullick FG, Martinez L et al (2002) Pathology related to chronic arsenic exposure. Environ Health Perspect 110:883–886
Chen Y, Graziano JH, Parvez F et al (2006) Modification of risk of arsenic-induced skin lesions by sunlight exposure, smoking, and occupational exposures in Bangladesh. Epidemiology 17:459–467. doi:10.1097/01.ede.0000220554.50837.7f
Dahmann D, Bauer H-D, Stoyke G (2008) Retrospective exposure assessement for respirable and inhalable dust, crystalline silica and arsenic in the former German uranium mines of SAG/SDAG Wismut. Int Arch Occup Environ Health 81:949–958. doi:10.1007/s00420-007-0287-8
Fisseler-Eckhoff A, Erfkamp S, Muller KM (1996) Cytokeratin expression in preneoplastic lesions and early squamous cell carcinoma of the bronchi. Pathol Res Pract 192:552–559
Giangreco A, Groot KR, Janes SM (2007) Lung cancer and lung stem cells: strange bedfellows? Am J Respir Crit Care Med 175:547–553. doi:10.1164/rccm.200607-984PP
Guo HR, Wang NS, Hu H, Monson RR (2004) Cell type specificity of lung cancer associated with arsenic ingestion. Cancer Epidemiol Biomarkers Prev 13:638–643
Hosmer DW, Lemeshow S (2000) Applied logistic regression, 2nd edn. Wiley, New York
Huang RN, Ho IC, Yih LH et al (1995) Sodium arsenite induces chromosome endoreduplication and inhibits protein phosphatase-activity in human fibroblasts. Environ Mol Mutagen 25:188–196. doi:10.1002/em.2850250304
IARC (1973) Arsenic and inorganic arsenic compounds. In: IARC. Some inorganic and organometallic compounds, 2nd edn. IARC (International Agency for Research on Cancer), Lyon
IARC (1980) Arsenic and arsenic compounds. In: IARC. Some metals and metallic compounds, 23rd edn. IARC (International Agency for Research on Cancer), Lyon
IARC (1987) Overall evaluation of carcinogenicity: an updating of IARC monographs. Monogr Eval Carcinog Risks Hum 1–41:56–178
Ives JC, Buffler PA, Greenberg SD (1983) Environmental associations and histopathologic patterns of carcinoma of the lung: the challenge and dilemma in epidemiologic studies. Am Rev Respir Dis 128:195–209
Kahn P (1993) A grisly archive of key cancer data. Science 259:448–451. doi:10.1126/science.8424163
Kim CFB, Jackson EL, Woolfenden AE et al (2005) Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121:823–835. doi:10.1016/j.cell.2005.03.032
Kreuzer M, Müller K, Brachner A et al (2000) Histopathologic findings of lung carcinoma in German uranium miners. Cancer 89:2613–2621. doi:10.1002/1097-0142(20001215)89:12<2613::AID-CNCR14>3.0.CO;2-Y
Lau ATY, Chiu JF (2006) Proteomic and biochemical analyses of in vitro carcinogen-induced lung cell transformation: synergism between arsenic and benzo[a]pyrene. Proteomics 6:1619–1630. doi:10.1002/pmic.200500332
Lehmann F, Hambeck F, Linkert KH, et al. (1998) Belastung durch ionisierende Strahlung im Uranerzbergbau der ehemaligen DDR: Abschlussbericht zu einem Forschungsvorhaben. HVBG, Sankt Augustin
Liao TF (2000) Estimated precision for predictions from generalized linear models in sociological research. Qual Quant 34:137–152. doi:10.1023/A:1004798429785
Liu J, Xie YX, Ducharme DMK et al (2006) Global gene expression associated with hepatocarcinogenesis in adult male mice induced by in utero arsenic exposure. Environ Health Perspect 114:404–411
Mohner M, Lindtner M, Otten H et al (2006) Leukemia and exposure to ionizing radiation among German uranium miners. Am J Ind Med 49:238–248. doi:10.1002/ajim.20289
Newman JA, Archer VE, Saccomanno G et al (1976) Histologic types of bronchogenic carcinoma among members of copper-mining and smelting communities. Ann N Y Acad Sci 271:260–268. doi:10.1111/j.1749-6632.1976.tb23120.x
Pershagen G, Bergmann F, Klominek J et al (1987) Histological types of lung cancer among smelter workers exposed to arsenic. Br J Ind Med 44:454–458
Polyak K, Hahn WC (2006) Roots and stems: stem cells in cancer. Nat Med 12:296–300. doi:10.1038/nm1379
Ramirez P, Del Razo LM, Gutierrez-Ruiz MC et al (2000) Arsenite induces DNA-protein crosslinks and cytokeratin expression in the WRL-68 human hepatic cell line. Carcinogenesis 21:701–706. doi:10.1093/carcin/21.4.701
Schmidt P, Strauss RP (1975) The prediction of occupation using multiple logit models. Int Econ Rev 16:471–486. doi:10.2307/2525826
Simonato L, Agudo A, Ahrens W et al (2001) Lung cancer and cigarette smoking in Europe: an update of risk estimates and an assessment of inter-country heterogeneity. Int J Cancer 91:876–887. doi:10.1002/1097-0215(200102)9999:9999<::AID-IJC1139>3.0.CO;2-7
Stang A, Pohlabeln H, Muller KM et al (2006) Diagnostic agreement in the histopathological evaluation of lung cancer tissue in a population-based case–control study. Lung Cancer 52:29–36. doi:10.1016/j.lungcan.2005.11.012
Taeger D, Fritsch A, Wiethege T et al (2006a) Role of exposure to radon and silicosis on the cell type of lung carcinoma in German uranium miners. Cancer 106:881–889. doi:10.1002/cncr.21677
Taeger D, Pesch B, Johnen G et al (2006b) Silicosis as modifying factor on the distribution of major histopathological types of lung cancer under cumulative quartz exposure. Air Qual Control 66:450–454
Taeger D, Krahn U, Wiethege T et al (2008) A study on lung cancer mortality related to radon, quartz, and arsenic exposures in German uranium miners. J Toxicol Environ Health A 71:859–865. doi:10.1080/15287390801987972
Tapio S, Grosche B (2006) Arsenic in the aetiology of cancer. Mutat Res 612:215–246. doi:10.1016/j.mrrev.2006.02.001
Tapio S, Danescu-Mayer J, Asmuss M et al (2005) Combined effects of gamma radiation and arsenite on the proteome of human TK6 lymphoblastoid cells. Mutat Res 581:141–152
Tokudome S, Haratake J, Horie A et al (1988) Histologic type of lung cancer among male Japanese copper smelter workers. Am J Ind Med 14:137–143. doi:10.1002/ajim.4700140204
Travis WD, Colby TV, Corrin B et al (1999) Histological typing of lung and pleural tumors, 3rd edn. Springer, Berlin
Tsubokawa F, Nishisaka T, Takeshima Y et al (2002) Heterogeneity of expression of cytokeratin subtypes in squamous cell carcinoma of the lung: with special reference to CK14 overexpression in cancer of high-proliferative and lymphogenous metastatic potential. Pathol Int 52:286–293. doi:10.1046/j.1440-1827.2002.01353.x
Wesch H, Wiethege T, Spiethoff A et al (1999) German uranium miner study—historical background and available histopathological material. Radiat Res 152:48–51. doi:10.2307/3580113
WHO (1977) Manual of the international statistical classification of diseases, injuries and causes of death, 9th revision. WHO (World Health Organization), Geneva
Wicks HJ, Archer VE, Auerbach O et al (1981) Arsenic exposure in a copper smelter as related to histological type of lung cancer. Am J Ind Med 2:25–31. doi:10.1002/ajim.4700020106
Wistuba II, Mao L, Gazdar AF (2002) Smoking molecular damage in bronchial epithelium. Oncogene 21:7298–7306. doi:10.1038/sj.onc.1205806
Yu HS, Liao WT, Chai CY (2006) Arsenic carcinogenesis in the skin. J Biomed Sci 13:657–666. doi:10.1007/s11373-006-9092-8
Acknowledgments
The authors gratefully acknowledge the Bundesanstalt für Arbeitsschutz und Arbeitsmedizin (BAuA, German Federal Institute for Occupational Safety and Health) for access to the Health Data Archive of the WISMUT corporation, the Deutsche Gesetzliche Unfallversicherung (DGUV) for access to the job-exposure matrix for ionizing radiation, quartz dust, arsenic exposure, and job history of WISMUT miners and the Deutsches Krebsforschungszentrum (DKFZ) for the database developed for the Archive.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taeger, D., Johnen, G., Wiethege, T. et al. Major histopathological patterns of lung cancer related to arsenic exposure in German uranium miners. Int Arch Occup Environ Health 82, 867–875 (2009). https://doi.org/10.1007/s00420-008-0386-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00420-008-0386-1