Abstract
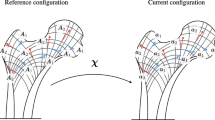
We construct in the present paper the effective growth response of trabecular bone, based on micromechanical analyses at the scale of a representative volume element consisting of individual trabeculae defining the representative unit cell (RUC). From a fundamental perspective, we adopt the physically and micromechanically motivated point of view that growth (resp. resorption) occurs as the surface remodeling of the individual trabeculae, the averaging of which leads to a net growth at the mesoscopic scale of the RUC. The effective model for the growing unit cell relies on an average kinematics which has been formulated in terms of the effective growth velocity gradient and effective elastic rate of deformation tensor, both functions of the rate of the effective density and stress applied over the RUC. The evaluation of the relation between the average rate of growth tensor and elastic growth rate versus the external stress applied to the RUC is obtained from a split of the average rate of growth tensor into its spherical and deviatoric parts, each of which being related to the corresponding applied stress quantities. The effective growth model is written for three different loading conditions as a polynomial relation between the components of the rate of growth tensor and similar components of the stress tensor under planar conditions, with a nonlinear function of the effective density as a weighting factor. The developed methodology is quite general and is applicable to any choice of the RUC morphology representative of the internal bone architecture.













Similar content being viewed by others
References
Beaupré, G.S., Orr, T.E., Carter, D.R.: An approach for time-dependent bone modeling and remodeling-application: a preliminary remodeling simulation. J. Orthop. Res. 8, 662–670 (1990)
Carter, D.R., Van der Meulen, M.C.H., Beaupré, G.S.: Mechanical factors in bone growth and development. Bone 18, S5–S10 (1996)
Chen, G., Pettet, G.J., Pearcy, M., McElwain, D.L.S.: Modelling external bone adaptation using evolutionary structural optimisation. Biomech. Model. Mechanobiol. 6, 275–285 (2007)
Cowin, S.C., Firoozbakhsh, K.: Bone remodeling of diaphysial surfaces under constant load: theoretical predictions. J. Biomech. 14, 471–484 (1981)
Cowin, S.C., Hegedus, D.H.: Bone remodeling I: theory of adaptive elasticity. J. Elast. 6, 313–325 (1976)
Doblaré, M., Garcia, J.M.: Anisotropic bone remodelling model based on a continuum damage-repair theory. J. Biomech. 35, 1–17 (2002)
Drozdov, A.D.: Volumetric growth of viscoelastic solids. Mech. Solids 25, 99–106 (1990)
Entov, V.M.: Mechanical model of scoliosis. Mech. Solids 18, 199–206 (1983)
Epstein, M., Maugin, G.A.: Thermomechanics of volumetric growth in uniform bodies. Int. J. Plast. 16, 951–978 (2000)
Fernandes, P., Rodrigues, H., Jacobs, C.: A model of bone adaptation using a global optimisation criterion based on the trajectorial theory of wolff. Comput. Methods Biomech. Biomed. Eng. 2, 125–138 (1999)
Fridez, P., Rakotomanana, L., Terrier, A., Leyvraz, P.F.: Three dimensional model of bone external adaptation. Comput. Methods Biomech. Biomed. Eng. 2, 189–196 (1998)
Ganghoffer, J.F., Plotnikov, P.I., Sokołowski, J.: Mathematical modeling of volumetric material growth. Arch. Appl. Mech. 84, 1357–1371 (2014)
Ganghoffer, J.F., Sokolowski, J.: A micromechanical approach to volumetric and surface growth in the framework of shape optimization. Int. J. Eng. Sci. 74, 207–226 (2014)
Ganghoffer, J.F.: A contribution to the mechanics and thermodynamics of surface growth, application to bone remodeling. Int. J. Eng. Sci. 50(1), 166–191 (2012)
Ganghoffer, J.F.: Mechanical modeling of growth considering domain variation—part II: volumetric and surface growth involving Eshelby tensors. J. Mech. Phys. Solids 58(9), 1434–1459 (2010)
Garijo, N., Fernàndez, J.R., Pérez, M.A., García-Aznar, J.M.: Numerical stability and convergence analysis of bone remodeling model. Comput. Methods Appl. Mech. Eng. 271, 253–268 (2014)
Goda, I., Assidi, M., Belouettar, S., Ganghoffer, J.F.: A micropolar anisotropic constitutive model of cancellous bone from discrete homogenization. J. Mech. Behav. Biomed. Mater. 16, 87–108 (2012)
Goda, I., Ganghoffer, J.F., Maurice, G.: Combined bone internal and external remodeling based on Eshelby stress. Int. J. Solids Struct. 94–95, 138–157 (2016)
Goda, I., Ganghoffer, J.F.: 3D plastic collapse and brittle fracture surface models of trabecular bone from asymptotic homogenization method. Int. J. Eng. Sci. 87, 58–82 (2015)
Hsu, F.H.: The influences of mechanical loads on the form of a growing elastic body. J. Biomech. 1, 303–311 (1968)
Huiskes, R., Weinans, H., Grootenboer, H.J., Dalstra, M., Fudala, B., Slooff, T.J.: Adaptive bone-remodeling theory applied to prosthetic-design analysis. J. Biomech. 20, 1135–1150 (1987)
Huiskes, R., Ruimerman, R., van Lenthe, G.H., Janssen, J.D.: Effects of mechanical forces on maintenance and adaptation of form in trabecular bone. Nature 405, 704–706 (2000)
Kaczmarczyk, L., Pearce, C.J.: Efficient numerical analysis of bone remodelling. J. Mech. Behav. Biomed. 4, 858–867 (2011)
Kuhl, E., Menzel, A., Steinmann, P.: Computational modeling of growth. Comput. Mech. 32, 71–88 (2003)
Lekszycki, T., dell’Isola, F.: A mixture model with evolving mass densities for describing synthesis and resorption phenomena in bones reconstructed with bio-resorbable materials. J. Appl. Math. Mech/Z. Angew. Math. Mech. 92(6), 426–444 (2012)
Levenston, M.E., Carter, D.R.: An energy dissipation- based model for damage stimulated bone adaptation. J. Biomech. 31, 579–586 (1998)
Lu, Y., Lekszycki, T.: Modelling of bone fracture healing: influence of gap size and angiogenesis into bioresorbable bone substitute. Math. Mech. Solids 1–14 (2016). doi:10.1177/1081286516653272
Martin, R.B.: Mathematical model for repair of fatigue damage and stress fracture in osteonal bone. J. Orthop. Res. 13(3), 309–316 (1995)
McNamara, B.P., Prendergast, P.J., Taylor, D.: Prediction of bone adaptation in the ulnar-osteotomized sheep’s forelimb using an anatomical finite element model. J. Biomed. Eng. 14, 209–216 (1992)
Menzel, A., Kuhl, E.: Frontiers in growth and remodeling. Mech. Res. Commun. 42, 1–14 (2012)
Negus, C., Impelluso, T.: Continuum remodeling revisited. Biomech. Model. Mechanobiol. 6(4), 211–226 (2007)
Prendergast, P.J., Taylor, D.: Prediction of bone adaptation using damage accumulation. J. Biomech. 27, 1067–1076 (1994)
Ramtani, S., Zidi, M.: A theoretical model of the effect of continuum damage on a bone adaptation model. J. Biomech. 34, 471–479 (2001)
Ramtani, S., Zidi, M.: Damaged-bone remodeling theory: thermodynamical approach. Mech. Res. Commun. 26(6), 701–708 (1999)
Rodriguez, E.K., Hoger, A., McCullogh, A.D.: Stress-dependent finite growth in soft elastic tissues. J. Biomech. 27(4), 455–467 (1994)
Sanz-Herrera, J.A., Garcia-Aznar, J.M., Doblare, M.: A mathematical model for bone tissue regeneration inside a specific type of scaffold. Biomech. Model. Mechanobiol. 7(5), 355–366 (2008)
Skalak, R., Farrow, D.A., Hoger, A.: Kinematics of surface growth. J. Math. Biol. 35, 869–907 (1997)
Skalak, R.: Growth as a finite displacement field. In: Carlsson, D.E., Shield, R.T. (eds.) Proceedings of the IUTAM Symposium on Finite Elasticity, Martinus Nijhoff, The Hague, pp. 347–355 (1981)
Stein, A.A.: The deformation of a rod of growing biological material under longitudinal compression. J. Appl. Math. Mech. 59, 139–146 (1995)
van der Meulen, M.C.H., Beaupré, G.S., Carter, D.R.: Mechanobiologic influences in long bone crosssectional growth. Bone 14, 635–642 (1993)
van Eijden, T.M.G.J., van Ruijven, L.J., Giesen, E.B.W.: Bone tissue stiffness in the mandibular condyle is dependent on the direction and density of the cancellous structure. Calcif. Tissue Int. 75, 502–508 (2004)
Weinans, H., Huiskes, R., Grootenboer, H.J.: The behavior of adaptive bone-remodeling simulation-models. J. Biomech. 25, 1425–1441 (1992)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Louna, Z., Goda, I., Ganghoffer, JF. et al. Formulation of an effective growth response of trabecular bone based on micromechanical analyses at the trabecular level. Arch Appl Mech 87, 457–477 (2017). https://doi.org/10.1007/s00419-016-1204-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00419-016-1204-y