Abstract
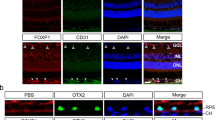
Epithelial-to-mesenchymal transition (EMT) of the retinal pigment epithelium (RPE) is commonly observed at sites of choroidal neovascularization in patients suffering from age-related macular degeneration. To learn in an experimental model how RPE EMT affects the biology of the choroidal vasculature, we studied transgenic mice (βB1-TGF-β1) with ocular overexpression of transforming growth factor-β1 (TGF-β1). RPE EMT was detectable at postnatal day (P)1 and included marked structural and functional alterations such as loss of the outer blood–retina barrier and reduced mRNA expression of the RPE-characteristic molecules Rlbp1, Rpe65, Rbp1 and Vegfa. Moreover, vascular endothelial growth factor (VEGF) was not detectable by immunohistochemistry at the RPE/choroid interface, while RPE cells stained intensely for α-smooth muscle actin. The choriocapillaris, the characteristic choroidal capillary network adjacent to the RPE, developed normally and was not obviously changed in embryonic transgenic eyes but was absent at P1 indicating its atrophy. At around the same time, photoreceptors stopped to differentiate and photoreceptor apoptosis was abundant in the second week of life. Structural changes were also seen in the retinal vasculature of transgenic animals, which did not form intraretinal vessels, and the hyaloid vasculature, which did not regress. In addition, the amounts of retinal HIF-1α and its mRNA were markedly reduced. We conclude that high amounts of active TGF-β1 in the mouse eye cause transdifferentiation of the RPE to a mesenchymal phenotype. The loss of epithelial differentiation leads to the diminished synthesis of RPE-characteristic molecules including that of VEGF. Lack of RPE-derived VEGF causes atrophy of the choriocapillaris, a scenario that disrupts photoreceptor differentiation and finally results in photoreceptor apoptosis. Lack of retinal vessel formation and of hyaloid vessel regression might be caused by the decrease in the metabolic requirements of the neuroretina leading to low amounts of retinal HIF-1α. In summary, our data indicate that failure of RPE differentiation may well precede and cause atrophy of the choriocapillaris. In contrast, RPE EMT is not sufficient to cause choroidal neovascularization.






Similar content being viewed by others
References
Ambati J, Fowler BJ (2012) Mechanisms of age-related macular degeneration. Neuron 75:26–39
Biesemeier A, Taubitz T, Julien S, Yoeruek E, Schraermeyer U (2014) Choriocapillaris breakdown precedes retinal degeneration in age-related macular degeneration. Neurobiol Aging 35:2562–2573
Bochaton-Piallat ML, Kapetanios AD, Donati G, Redard M, Gabbiani G, Pournaras CJ (2000) TGF-beta1, TGF-beta receptor II and ED-A fibronectin expression in myofibroblast of vitreoretinopathy. Invest Ophthalmol Vis Sci 41:2336–2342
Braunger BM, Ohlmann A, Koch M, Tanimoto N, Volz C, Yang Y, Bosl MR, Cvekl A, Jagle H, Seeliger MW, Tamm ER (2013a) Constitutive overexpression of Norrin activates Wnt/beta-catenin and endothelin-2 signaling to protect photoreceptors from light damage. Neurobiol Dis 50:1–12
Braunger BM, Pielmeier S, Demmer C, Landstorfer V, Kawall D, Abramov N, Leibinger M, Kleiter I, Fischer D, Jagle H, Tamm ER (2013b) TGF-beta signaling protects retinal neurons from programmed cell death during the development of the mammalian eye. J Neurosci 33:14246–14258
Braunger BM, Leimbeck SV, Schlecht A, Volz C, Jagle H, Tamm ER (2015) Deletion of ocular transforming growth factor beta signaling mimics essential characteristics of diabetic retinopathy. Am J Pathol 185:1749–1768
Chen X, Xiao W, Wang W, Luo L, Ye S, Liu Y (2014) The complex interplay between ERK1/2, TGFbeta/Smad, and Jagged/Notch signaling pathways in the regulation of epithelial–mesenchymal transition in retinal pigment epithelium cells. PLoS ONE 9:e96365
Connor TB, Roberts AB, Sporn MB, Danielpour D, Dart LL, Michels RG, de Bustros S, Enger C, Kato H, Lansing M, Hayashi H, Glaser BM (1989) Correlation of fibrosis and transforming growth factor-b type 2 levels in the eye. J Clin Invest 83:1661–1666
Cvetanovic M, Patel JM, Marti HH, Kini AR, Opal P (2011) Vascular endothelial growth factor ameliorates the ataxic phenotype in a mouse model of spinocerebellar ataxia type 1. Nat Med 17:1445–1447
D’Amore PA, Smith SR (1993) Growth factor effects on cells of the vascular wall: a survey. Growth Factors 8:61–75
Del Priore LV, Hornbeck R, Kaplan HJ, Jones Z, Valentino TL, Mosinger-Ogilvie J, Swinn M (1995) Debridement of the pig retinal pigment epithelium in vivo. Arch Ophthalmol 113:939–944
Duncan MK, Roth HJ, Thompson M, Kantorow M, Piatigorsky J (1995) Chicken βB1 crystallin: gene sequence and evidence for functional conservation of promoter activity between chicken and mouse. Biochim Biophys Acta 1261:68–76
Duncan MK, Li X, Ogino H, Yasuda K, Piatigorsky J (1996) Developmental regulation of the chicken βB1-crystallin promoter in transgenic mice. Mech Dev 57:79–89
Dvashi Z, Goldberg M, Adir O, Shapira M, Pollack A (2015) TGF-beta1 induced transdifferentiation of RPE cells is mediated by TAK1. PLoS ONE 10:e0122229
Finger RP, Fimmers R, Holz FG, Scholl HP (2011) Incidence of blindness and severe visual impairment in Germany: projections for 2030. Invest Ophthalmol Vis Sci 52:4381–4389
Flügel-Koch C, Ohlmann A, Piatigorsky J, Tamm ER (2002) Disruption of anterior segment development by TGF-beta1 overexpression in the eyes of transgenic mice. Dev Dyn 225:111–125
Ford KM, Saint-Geniez M, Walshe T, Zahr A, D’Amore PA (2011) Expression and role of VEGF in the adult retinal pigment epithelium. Invest Ophthalmol Vis Sci 52:9478–9487
Friedman DS, O’Colmain BJ, Munoz B, Tomany SC, McCarty C, de Jong PT, Nemesure B, Mitchell P, Kempen J, Eye Diseases Prevalence Research G (2004) Prevalence of age-related macular degeneration in the United States. Arch Ophthalmol 122:564–572
Fruttiger M (2002) Development of the mouse retinal vasculature: angiogenesis versus vasculogenesis. Invest Ophthalmol Vis Sci 43:522–527
Gamulescu MA, Chen Y, He S, Spee C, Jin M, Ryan SJ, Hinton DR (2006) Transforming growth factor beta2-induced myofibroblastic differentiation of human retinal pigment epithelial cells: regulation by extracellular matrix proteins and hepatocyte growth factor. Exp Eye Res 83:212–222
Gimenez E, Montoliu L (2001) A simple polymerase chain reaction assay for genotyping the retinal degeneration mutation (Pdeb(rd1)) in FVB/N-derived transgenic mice. Lab Anim 35:153–156
Guarino M, Tosoni A, Nebuloni M (2009) Direct contribution of epithelium to organ fibrosis: epithelial–mesenchymal transition. Hum Pathol 40:1365–1376
Guidry C, Medeiros NE, Curcio CA (2002) Phenotypic variation of retinal pigment epithelium in age-related macular degeneration. Invest Ophthalmol Vis Sci 43:267–273
Hales AM, Schulz MW, Chamberlain CG, McAvoy JW (1994) TGF-b1 induces lens cells to accumulate a-smooth muscle actin, a marker for subcapsular cataracts. Curr Eye Res 13:885–890
Herrnberger L, Ebner K, Junglas B, Tamm ER (2012a) The role of plasmalemma vesicle-associated protein (PLVAP) in endothelial cells of Schlemm’s canal and ocular capillaries. Exp Eye Res 105:27–33
Herrnberger L, Seitz R, Kuespert S, Bösl MR, Fuchshofer R, Tamm ER (2012b) Lack of endothelial diaphragms in fenestrae and caveolae of mutant Plvap-deficient mice. Histochem Cell Biol 138:709–724
Hirasawa M, Noda K, Noda S, Suzuki M, Ozawa Y, Shinoda K, Inoue M, Ogawa Y, Tsubota K, Ishida S (2011) Transcriptional factors associated with epithelial–mesenchymal transition in choroidal neovascularization. Mol Vis 17:1222–1230
Ito S, Karnovsky MJ (1968) Formaldehyde-glutaraldehyde fixatives containing trinitro compounds. J Cell Biol 39:168A–169A
Ito M, Yoshioka M (1999) Regression of the hyaloid vessels and pupillary membrane of the mouse. Anat Embryol (Berl) 200:403–411
Jun JH, Joo CK (2016) MicroRNA-124 controls transforming growth factor beta1-induced epithelial–mesenchymal transition in the retinal pigment epithelium by targeting RHOG. Invest Ophthalmol Vis Sci 57:12–22
Kalluri R, Neilson EG (2003) Epithelial–mesenchymal transition and its implications for fibrosis. J Clin Invest 112:1776–1784
Kilic U, Kilic E, Jarve A, Guo Z, Spudich A, Bieber K, Barzena U, Bassetti CL, Marti HH, Hermann DM (2006) Human vascular endothelial growth factor protects axotomized retinal ganglion cells in vivo by activating ERK-1/2 and Akt pathways. J Neurosci 26:12439–12446
Klaver CC, Wolfs RC, Vingerling JR, Hofman A, de Jong PT (1998) Age-specific prevalence and causes of blindness and visual impairment in an older population: the Rotterdam Study. Arch Ophthalmol 116:653–658
Korte GE, Reppucci V, Henkind P (1984) RPE destruction causes choriocapillary atrophy. Invest Ophthalmol Vis Sci 25:1135–1145
Kurihara T, Westenskow PD, Krohne TU, Aguilar E, Johnson RS, Friedlander M (2011) Astrocyte pVHL and HIF-alpha isoforms are required for embryonic-to-adult vascular transition in the eye. J Cell Biol 195:689–701
Kurihara T, Westenskow PD, Bravo S, Aguilar E, Friedlander M (2012) Targeted deletion of Vegfa in adult mice induces vision loss. J Clin Invest 122:4213–4217
Kurosaka D, Muraki Y, Inoue M, Katsura H (1996) TGF-beta 2 increases alpha-smooth muscle actin expression in bovine retinal pigment epithelial cells. Curr Eye Res 15:1144–1147
Le YZ, Bai Y, Zhu M, Zheng L (2010) Temporal requirement of RPE-derived VEGF in the development of choroidal vasculature. J Neurochem 112:1584–1592
Lee SC, Kwon OW, Seong GJ, Kim SH, Ahn JE, Kay ED (2001) Epitheliomesenchymal transdifferentiation of cultured RPE cells. Ophthalmic Res 33:80–86
Lim LS, Mitchell P, Seddon JM, Holz FG, Wong TY (2012) Age-related macular degeneration. Lancet 379:1728–1738
Lopez PF, Sippy BD, Lambert HM, Thach AB, Hinton DR (1996) Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically excised age-related macular degeneration-related choroidal neovascular membranes. Invest Ophthalmol Vis Sci 37:855–868
Mancel E, Hirsch M (1989) Development of tight junctions in the human ciliary epithelium. Exp Eye Res 48:87–97
Marneros AG, Fan J, Yokoyama Y, Gerber HP, Ferrara N, Crouch RK, Olsen BR (2005) Vascular endothelial growth factor expression in the retinal pigment epithelium is essential for choriocapillaris development and visual function. Am J Pathol 167:1451–1459
McLeod DS, Grebe R, Bhutto I, Merges C, Baba T, Lutty GA (2009) Relationship between RPE and choriocapillaris in age-related macular degeneration. Invest Ophthalmol Vis Sci 50:4982–4991
Miettinen PJ, Ebner R, Lopez AR, Derynck R (1994) TGF-beta induced transdifferentiation of mammary epithelial cells to mesenchymal cells: involvement of type I receptors. J Cell Biol 127:2021–2036
Nakamura-Ishizu A, Kurihara T, Okuno Y, Ozawa Y, Kishi K, Goda N, Tsubota K, Okano H, Suda T, Kubota Y (2012) The formation of an angiogenic astrocyte template is regulated by the neuroretina in a HIF-1-dependent manner. Dev Biol 363:106–114
Nishijima K, Ng YS, Zhong L, Bradley J, Schubert W, Jo N, Akita J, Samuelsson SJ, Robinson GS, Adamis AP, Shima DT (2007) Vascular endothelial growth factor-A is a survival factor for retinal neurons and a critical neuroprotectant during the adaptive response to ischemic injury. Am J Pathol 171:53–67
Pepper MS (1997) Transforming growth factor-beta: vasculogenesis, angiogenesis, and vessel wall integrity. Cytokine Growth Factor Rev 8:21–43
RayChaudhury A, D’Amore PA (1991) Endothelial cell regulation by transforming growth factor-beta. J Cell Biochem 47:224–229
Roberts AB, Tian F, Byfield SD, Stuelten C, Ooshima A, Saika S, Flanders KC (2006) Smad3 is key to TGF-beta-mediated epithelial-to-mesenchymal transition, fibrosis, tumor suppression and metastasis. Cytokine Growth Factor Rev 17:19–27
Robinson GS, Ju M, Shih SC, Xu X, McMahon G, Caldwell RB, Smith LE (2001) Nonvascular role for VEGF: VEGFR-1, 2 activity is critical for neural retinal development. Faseb J 15:1215–1217
Rousseau B, Larrieu-Lahargue F, Bikfalvi A, Javerzat S (2003) Involvement of fibroblast growth factors in choroidal angiogenesis and retinal vascularization. Exp Eye Res 77:147–156
Saari JC, Bredberg L, Garwin GG (1982) Identification of the endogenous retinoids associated with three cellular retinoid-binding proteins from bovine retina and retinal pigment epithelium. J Biol Chem 257:13329–13333
Saari JC, Nawrot M, Kennedy BN, Garwin GG, Hurley JB, Huang J, Possin DE, Crabb JW (2001) Visual cycle impairment in cellular retinaldehyde binding protein (CRALBP) knockout mice results in delayed dark adaptation. Neuron 29:739–748
Saika S, Kono-Saika S, Tanaka T, Yamanaka O, Ohnishi Y, Sato M, Muragaki Y, Ooshima A, Yoo J, Flanders KC, Roberts AB (2004) Smad3 is required for dedifferentiation of retinal pigment epithelium following retinal detachment in mice. Lab Invest 84:1245–1258
Saint-Geniez M, D’Amore PA (2004) Development and pathology of the hyaloid, choroidal and retinal vasculature. Int J Dev Biol 48:1045–1058
Saint-Geniez M, Maldonado AE, D’Amore PA (2006) VEGF expression and receptor activation in the choroid during development and in the adult. Invest Ophthalmol Vis Sci 47:3135–3142
Saint-Geniez M, Maharaj AS, Walshe TE, Tucker BA, Sekiyama E, Kurihara T, Darland DC, Young MJ, D’Amore PA (2008) Endogenous VEGF is required for visual function: evidence for a survival role on Muller cells and photoreceptors. PLoS ONE 3:e3554
Saint-Geniez M, Kurihara T, Sekiyama E, Maldonado AE, D’Amore PA (2009) An essential role for RPE-derived soluble VEGF in the maintenance of the choriocapillaris. Proc Natl Acad Sci USA 106:18751–18756
Serrano-Gomez SJ, Maziveyi M, Alahari SK (2016) Regulation of epithelial–mesenchymal transition through epigenetic and post-translational modifications. Mol Cancer 15:18
Strauss O (2005) The retinal pigment epithelium in visual function. Physiol Rev 85:845–881
Tamiya S, Kaplan HJ (2016) Role of epithelial–mesenchymal transition in proliferative vitreoretinopathy. Exp Eye Res 142:26–31
Thiery JP, Sleeman JP (2006) Complex networks orchestrate epithelial–mesenchymal transitions. Nat Rev Mol Cell Biol 7:131–142
Thiery JP, Acloque H, Huang RY, Nieto MA (2009) Epithelial–mesenchymal transitions in development and disease. Cell 139:871–890
Tornquist P, Alm A (1979) Retinal and choroidal contribution to retinal metabolism in vivo. A study in pigs. Acta Physiol Scand 106:351–357
Watanabe D, Takagi H, Suzuma K, Oh H, Ohashi H, Honda Y (2005) Expression of connective tissue growth factor and its potential role in choroidal neovascularization. Retina 25:911–918
Xu J, Lamouille S, Derynck R (2009) TGF-beta-induced epithelial to mesenchymal transition. Cell Res 19:156–172
Yang S, Yao H, Li M, Li H, Wang F (2016) Long non-coding RNA MALAT1 mediates transforming growth factor beta1-induced epithelial–mesenchymal transition of retinal pigment epithelial cells. PLoS ONE 11:e0152687
Zhao S, Overbeek PA (2001a) Elevated TGFbeta signaling inhibits ocular vascular development. Dev Biol 237:45–53
Zhao S, Overbeek PA (2001b) Regulation of choroid development by the retinal pigment epithelium. Mol Vis 7:277–282
Acknowledgments
The excellent technical help of Angelika Pach, Margit Schimmel and Elke Stauber is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors dedicate this article to Detlev Drenckhahn. Over many years he has shaped and advanced the fields of anatomy and cell biology in a tremendously positive way.
Andreas Ohlmann and Michael Scholz have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ohlmann, A., Scholz, M., Koch, M. et al. Epithelial–mesenchymal transition of the retinal pigment epithelium causes choriocapillaris atrophy. Histochem Cell Biol 146, 769–780 (2016). https://doi.org/10.1007/s00418-016-1461-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-016-1461-4