Abstract
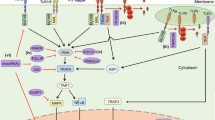
Wnt10b is a signaling protein regulating skin development and homeostasis, and the expression of Wnt10b is restricted to epidermal keratinocytes in embryonic and postnatal skin. Recent studies indicate an elevated expression of Wnt10b in skin tumors. However, how Wnt10b regulates skin tumorigenesis remains largely unknown. Here we report that continuous expression of Wnt10b mediates transformation of epidermal keratinocytes through activating genes involved in EGF/MAPK signaling pathways. We first established a prolonged Wnt10b overexpression system in JB6P− cells to represent the elevated Wnt10b expression level in skin keratinocytes. Through expression assays and observations under phase-contrast microscopy, prolonged expression of Wnt10b activated Wnt/β-catenin pathway and induced morphological changes of cells showing longer protrusions and multilayer growth, indicating early-stage cell transformation. Wnt10b also increased cellular proliferation and migration according to BrdU incorporation and cell mobility assays. Furthermore, multi-doses of AdWnt10b treatment to JB6P− cells induced colony formation, stronger invasive ability in transwell system, and anchorage-independent growth in agar gel. In molecular level, AdWnt10b treatment induced increased transcriptional expressions of Egf, downstream Mapk pathway factors, and MMPs. Administration of Wnt antagonist DKK1 blocked the tumor promotion process induced by Wnt10b. Taken together, these findings clearly demonstrate that Wnt10b promotes epidermal keratinocyte transformation through induced Egf pathway.







Similar content being viewed by others
Abbreviations
- AdGFP:
-
Control adenoviruses
- AdWnt10b:
-
Wnt10b adenoviruses
- AP-1:
-
Activator protein
- BCC:
-
Basal cell carcinoma
- DKK1:
-
Dickkopf 1
- ECM:
-
Extracellular matrix
- EGF:
-
Epidermal growth factor
- NF-KB:
-
Nuclear factor kappa B
- PAGE:
-
Polyacrylamide gel electrophoresis
- PBS:
-
Phosphate-buffered saline
- PVDF:
-
Polyvinylidene fluoride
- RT-PCR:
-
Reverse transcription-polymerase chain reaction
- SCC:
-
Squamous cell carcinomas
- SDS:
-
Sodium dodecyl sulfate
- TNF-a:
-
Tumor necrosis factor-a
- TPA:
-
12-O-tetradecanoylphorbol-13-acetate
- Wnt10b:
-
Wingless-type MMTV integration site family, member 10B
References
Andl T, Reddy ST, Gaddapara T, Millar SE (2002) WNT signals are required for the initiation of hair follicle development. Dev Cell 2(5):643–653
Bernstein LR, Colburn NH (1989) AP1/jun function is differentially induced in promotion-sensitive and resistant JB6 cells. Science 244(4904):566–569
Bhatia N, Spiegelman VS (2005) Activation of Wnt/beta-catenin/Tcf signaling in mouse skin carcinogenesis. Mol Carcinog 42(4):213–221. doi:10.1002/mc.20077
Brennan KR, Brown AM (2004) Wnt proteins in mammary development and cancer. J Mammary Gland Biol Neoplasia 9(2):119–131. doi:10.1023/B:JOMG.0000037157.94207.33
Colburn NH, Gindhart TD (1981) Specific binding of transforming growth factor correlates with promotion of anchorage independence in EGF receptorless mouse JB6 cells. Biochem Biophys Res Commun 102(2):799–807
Colburn NH, Koehler BA, Nelson KJ (1980) A cell culture assay for tumor-promoter-dependent progression toward neoplastic phenotype: detection of tumor promoters and promotion inhibitors. Teratog Carcinog Mutagen 1(1):87–96
Colburn NH, Wendel EJ, Abruzzo G (1981) Dissociation of mitogenesis and late-stage promotion of tumor cell phenotype by phorbol esters: mitogen-resistant variants are sensitive to promotion. Proc Natl Acad Sci USA 78(11):6912–6916
Coppe JP, Boysen M, Sun CH, Wong BJ, Kang MK, Park NH, Desprez PY, Campisi J, Krtolica A (2008) A role for fibroblasts in mediating the effects of tobacco-induced epithelial cell growth and invasion. Mol Cancer Res 6(7):1085–1098. doi:10.1158/1541-7786.MCR-08-0062
Dong Z, Birrer MJ, Watts RG, Matrisian LM, Colburn NH (1994) Blocking of tumor promoter-induced AP-1 activity inhibits induced transformation in JB6 mouse epidermal cells. Proc Natl Acad Sci USA 91(2):609–613
Han CY, Hien TT, Lim SC, Kang KW (2011) Role of Pin1 in UVA-induced cell proliferation and malignant transformation in epidermal cells. Biochem Biophys Res Commun 410(1):68–74. doi:10.1016/j.bbrc.2011.05.106
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B (1998) A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 95(5):2509–2514
Hennings H, Glick AB, Greenhalgh DA, Morgan DL, Strickland JE, Tennenbaum T, Yuspa SH (1993) Critical aspects of initiation, promotion, and progression in multistage epidermal carcinogenesis. Proc Soc Exp Biol Med 202(1):1–8
Heuberger J, Birchmeier W (2010) Interplay of cadherin-mediated cell adhesion and canonical Wnt signaling. Cold Spring Harb Perspect Biol 2(2):a002915. doi:10.1101/cshperspect.a002915
Hsu TC, Young MR, Cmarik J, Colburn NH (2000) Activator protein 1 (AP-1)- and nuclear factor kappaB (NF-kappaB)-dependent transcriptional events in carcinogenesis. Free Radic Biol Med 28(9):1338–1348
Hu J, Nakano H, Sakurai H, Colburn NH (2004) Insufficient p65 phosphorylation at S536 specifically contributes to the lack of NF-kappaB activation and transformation in resistant JB6 cells. Carcinogenesis 25(10):1991–2003. doi:10.1093/carcin/bgh198
Huang C, Ma WY, Young MR, Colburn N, Dong Z (1998) Shortage of mitogen-activated protein kinase is responsible for resistance to AP-1 transactivation and transformation in mouse JB6 cells. Proc Natl Acad Sci USA 95(1):156–161
Ingraham CA, Park GC, Makarenkova HP, Crossin KL (2011) Matrix metalloproteinase (MMP)-9 induced by Wnt signaling increases the proliferation and migration of embryonic neural stem cells at low O2 levels. J Biol Chem 286(20):17649–17657. doi:10.1074/jbc.M111.229427
Kamino M, Kishida M, Kibe T, Ikoma K, Iijima M, Hirano H, Tokudome M, Chen L, Koriyama C, Yamada K, Arita K, Kishida S (2011) Wnt-5a signaling is correlated with infiltrative activity in human glioma by inducing cellular migration and MMP-2. Cancer Sci 102(3):540–548. doi:10.1111/j.1349-7006.2010.01815.x
Kopper L, Hajdu M (2004) Tumor stem cells. Pathol Oncol Res 10(2):69–73
Lane TF, Leder P (1997) Wnt-10b directs hypermorphic development and transformation in mammary glands of male and female mice. Oncogene 15(18):2133–2144. doi:10.1038/sj.onc.1201593
Lei M, Gao X, Yang L, Yang T, Lian X (2011) Gsdma3 gene is needed for the induction of apoptosis-driven catagen during mouse hair follicle cycle. Histochem Cell Biol 136(3):335–343. doi:10.1007/s00418-011-0845-8
Lei M, Bai X, Yang T, Lai X, Qiu W, Yang L, Lian X (2012) Gsdma3 is a new factor needed for TNF-alpha-mediated apoptosis signal pathway in mouse skin keratinocytes. Histochem Cell Biol 138(3):385–396. doi:10.1007/s00418-012-0960-1
Lei MX, Chuong CM, Widelitz RB (2013) Tuning Wnt signals for more or fewer hairs. J Invest Dermatol 133(1):7–9. doi:10.1038/jid.2012.446
Lei M, Guo H, Qiu W, Lai X, Yang T, Widelitz RB, Chuong CM, Lian X, Yang L (2014) Modulating hair follicle size with Wnt10b/DKK1 during hair regeneration. Exp Dermatol 23(6):407–413. doi:10.1111/exd.12416
Li L, Xie T (2005) Stem cell niche: structure and function. Annu Rev Cell Dev Biol 21:605–631. doi:10.1146/annurev.cellbio.21.012704.131525
Li YH, Zhang K, Ye JX, Lian XH, Yang T (2011) Wnt10b promotes growth of hair follicles via a canonical Wnt signalling pathway. Clin Exp Dermatol 36(5):534–540. doi:10.1111/j.1365-2230.2011.04019.x
Li YH, Zhang K, Yang K, Ye JX, Xing YZ, Guo HY, Deng F, Lian XH, Yang T (2013) Adenovirus-mediated Wnt10b overexpression induces hair follicle regeneration. J Invest Dermatol 133(1):42–48. doi:10.1038/jid.2012.235
Li W, Liu J, Zhao Y (2014) PKM2 inhibitor shikonin suppresses TPA-induced mitochondrial malfunction and proliferation of skin epidermal JB6 cells. Mol Carcinog 53(5):403–412. doi:10.1002/mc.21988
Liang CC, Park AY, Guan JL (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2(2):329–333. doi:10.1038/nprot.2007.30
Lu Z, Liu M, Stribinskis V, Klinge CM, Ramos KS, Colburn NH, Li Y (2008) MicroRNA-21 promotes cell transformation by targeting the programmed cell death 4 gene. Oncogene 27(31):4373–4379. doi:10.1038/onc.2008.72
Ouji Y, Yoshikawa M, Moriya K, Nishiofuku M, Matsuda R, Ishizaka S (2008) Wnt-10b, uniquely among Wnts, promotes epithelial differentiation and shaft growth. Biochem Biophys Res Commun 367(2):299–304. doi:10.1016/j.bbrc.2007.12.091
Ouyang W, Li J, Ma Q, Huang C (2006) Essential roles of PI-3 K/Akt/IKKbeta/NFkappaB pathway in cyclin D1 induction by arsenite in JB6 Cl41 cells. Carcinogenesis 27(4):864–873. doi:10.1093/carcin/bgi321
Paredes-Gonzalez X, Fuentes F, Su ZY, Kong AN (2014) Apigenin reactivates Nrf2 anti-oxidative stress signaling in mouse skin epidermal JB6 P + cells through epigenetics modifications. AAPS J 16(4):727–735. doi:10.1208/s12248-014-9613-8
Pukrop T, Klemm F, Hagemann T, Gradl D, Schulz M, Siemes S, Trumper L, Binder C (2006) Wnt 5a signaling is critical for macrophage-induced invasion of breast cancer cell lines. Proc Natl Acad Sci USA 103(14):5454–5459. doi:10.1073/pnas.0509703103
Saikali M, Ghantous A, Halawi R, Talhouk SN, Saliba NA, Darwiche N (2012) Sesquiterpene lactones isolated from indigenous Middle Eastern plants inhibit tumor promoter-induced transformation of JB6 cells. BMC Complement Altern Med 12:89. doi:10.1186/1472-6882-12-89
Schmidt KN, Amstad P, Cerutti P, Baeuerle PA (1996) Identification of hydrogen peroxide as the relevant messenger in the activation pathway of transcription factor NF-kappaB. Adv Exp Med Biol 387:63–68
Sharov AA, Mardaryev AN, Sharova TY, Grachtchouk M, Atoyan R, Byers HR, Seykora JT, Overbeek P, Dlugosz A, Botchkarev VA (2009) Bone morphogenetic protein antagonist noggin promotes skin tumorigenesis via stimulation of the Wnt and Shh signaling pathways. Am J Pathol 175(3):1303–1314. doi:10.2353/ajpath.2009.090163
Sick S, Reinker S, Timmer J, Schlake T (2006) WNT and DKK determine hair follicle spacing through a reaction-diffusion mechanism. Science 314(5804):1447–1450. doi:10.1126/science.1130088
Singh N, Sun Y, Nakamura K, Smith MR, Colburn NH (1995) C-JUN/AP-1 as possible mediators of tumor necrosis factor-alpha-induced apoptotic response in mouse JB6 tumor cells. Oncol Res 7(7–8):353–362
Soto E, Yanagisawa M, Marlow LA, Copland JA, Perez EA, Anastasiadis PZ (2008) p120 catenin induces opposing effects on tumor cell growth depending on E-cadherin expression. J Cell Biol 183(4):737–749. doi:10.1083/jcb.200805113
Spyratos F, Delarue JC, Andrieu C, Lidereau R, Champeme MH, Hacene K, Brunet M (1990) Epidermal growth factor receptors and prognosis in primary breast cancer. Breast Cancer Res Treat 17(2):83–89
Su ZY, Zhang C, Lee JH, Shu L, Wu TY, Khor TO, Conney AH, Lu YP, Kong AN (2014) Requirement and epigenetics reprogramming of Nrf2 in suppression of tumor promoter TPA-induced mouse skin cell transformation by sulforaphane. Cancer Prev Res (Phila) 7(3):319–329. doi:10.1158/1940-6207.CAPR-13-0313-T
Suzukawa K, Weber TJ, Colburn NH (2002) AP-1, NF-kappa-B, and ERK activation thresholds for promotion of neoplastic transformation in the mouse epidermal JB6 model. Environ Health Perspect 110(9):865–870
Vaughan MB, Ramirez RD, Andrews CM, Wright WE, Shay JW (2009) H-ras expression in immortalized keratinocytes produces an invasive epithelium in cultured skin equivalents. PLoS ONE 4(11):e7908. doi:10.1371/journal.pone.0007908
Widelitz RB (2008) Wnt signaling in skin organogenesis. Organogenesis 4(2):123–133
Wilkins-Port CE, Higgins PJ (2007) Regulation of extracellular matrix remodeling following transforming growth factor-beta1/epidermal growth factor-stimulated epithelial-mesenchymal transition in human premalignant keratinocytes. Cells Tissues Organs 185(1–3):116–122. doi:10.1159/000101312
Wittwer JA, Robbins D, Wang F, Codarin S, Shen X, Kevil CG, Huang TT, Van Remmen H, Richardson A, Zhao Y (2011) Enhancing mitochondrial respiration suppresses tumor promoter TPA-induced PKM2 expression and cell transformation in skin epidermal JB6 cells. Cancer Prev Res (Phila) 4(9):1476–1484. doi:10.1158/1940-6207.CAPR-11-0028
Wong SY, Reiter JF (2011) Wounding mobilizes hair follicle stem cells to form tumors. Proc Natl Acad Sci USA 108(10):4093–4098. doi:10.1073/pnas.1013098108
Wu B, Crampton SP, Hughes CC (2007) Wnt signaling induces matrix metalloproteinase expression and regulates T cell transmigration. Immunity 26(2):227–239. doi:10.1016/j.immuni.2006.12.007
Yamaguchi Y, Passeron T, Hoashi T, Watabe H, Rouzaud F, Yasumoto K, Hara T, Tohyama C, Katayama I, Miki T, Hearing VJ (2008) Dickkopf 1 (DKK1) regulates skin pigmentation and thickness by affecting Wnt/beta-catenin signaling in keratinocytes. FASEB J 22(4):1009–1020. doi:10.1096/fj.07-9475com
Yang S, Misner BJ, Chiu RJ, Meyskens FL Jr (2007) Redox effector factor-1, combined with reactive oxygen species, plays an important role in the transformation of JB6 cells. Carcinogenesis 28(11):2382–2390. doi:10.1093/carcin/bgm128
Yang SH, Andl T, Grachtchouk V, Wang A, Liu J, Syu LJ, Ferris J, Wang TS, Glick AB, Millar SE, Dlugosz AA (2008) Pathological responses to oncogenic Hedgehog signaling in skin are dependent on canonical Wnt/beta3-catenin signaling. Nat Genet 40(9):1130–1135. doi:10.1038/ng.192
Ye J, Yang T, Guo H, Tang Y, Deng F, Li Y, Xing Y, Yang L, Yang K (2013) Wnt10b promotes differentiation of mouse hair follicle melanocytes. Int J Med Sci 10(6):691–698. doi:10.7150/ijms.6170
Zhang ZG, Bothe I, Hirche F, Zweers M, Gullberg D, Pfitzer G, Krieg T, Eckes B, Aumailley M (2006) Interactions of primary fibroblasts and keratinocytes with extracellular matrix proteins: contribution of alpha 2 beta 1 integrin. J Cell Sci 119(Pt 9):1886–1895. doi:10.1242/jcs.02921
Acknowledgments
This study was supported by Grants 81171515 from National Nature Science Foundation of China and CSTC; Innovation and Attracting Talents Program for College and University (‘111’ Project) (B06023), China; and the “BaYu” Scholars Project in Chongqing for Dr. Cheng-Ming Chuong. We thank Dr. Pin-Chi Tang (National Chung Hsing University) and Dr. Ya-Chen Liang (University of Southern California) for carefully revising the manuscript.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
M. Lei and X. Lai have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lei, M., Lai, X., Bai, X. et al. Prolonged overexpression of Wnt10b induces epidermal keratinocyte transformation through activating EGF pathway. Histochem Cell Biol 144, 209–221 (2015). https://doi.org/10.1007/s00418-015-1330-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-015-1330-6