Scientific inquiry starts with observation. The more we can see, the more we can investigate.
Martin Chalfie
2008 Nobel Lecture
Abstract
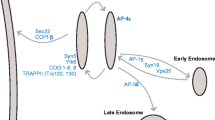
The Golgi apparatus contains multiple classes of cisternae that differ in structure, composition, and function, but there is no consensus about the number and definition of these classes. A useful way to classify Golgi cisternae is according to the trafficking pathways by which the cisternae import and export components. By this criterion, we propose that Golgi cisternae can be divided into three classes that correspond to functional stages of maturation. First, cisternae at the cisternal assembly stage receive COPII vesicles from the ER and recycle components to the ER in COPI vesicles. At this stage, new cisternae are generated. Second, cisternae at the carbohydrate synthesis stage exchange material with one another via COPI vesicles. At this stage, most of the glycosylation and polysaccharide synthesis reactions occur. Third, cisternae at the carrier formation stage produce clathrin-coated vesicles and exchange material with endosomes. At this stage, biosynthetic cargo proteins are packaged into various transport carriers, and the cisternae ultimately disassemble. Discrete transitions occur as a cisterna matures from one stage to the next. Within each stage, the structure and composition of a cisterna can evolve, but the trafficking pathways remain unchanged. This model offers a unified framework for understanding the properties of the Golgi in diverse organisms.


Similar content being viewed by others
References
Anitei M, Hoflack B (2011) Exit from the trans-Golgi network: from molecules to mechanisms. Curr Opin Cell Biol 23:443–451
Appenzeller-Herzog C, Hauri HP (2006) The ER-Golgi intermediate compartment (ERGIC): in search of its identity and function. J Cell Sci 119:2173–2183
Atmodjo MA, Hao Z, Mohnen D (2013) Evolving views of pectin biosynthesis. Annu Rev Plant Biol 64:747–779
Banfield DK (2011) Mechanisms of protein retention in the Golgi. Cold Spring Harb Perspect Biol 3:a005264
Bankaitis VA, Garcia-Mata R, Mousley CJ (2012) Golgi membrane dynamics and lipid metabolism. Curr Biol 22:R414–R424
Bannykh SI, Balch WE (1997) Membrane dynamics at the endoplasmic reticulum-Golgi interface. J Cell Biol 138:1–4
Bard F, Malhotra V (2006) The formation of TGN-to-plasma membrane transport carriers. Annu Rev Cell Dev Biol 22:439–455
Barlowe CK, Miller EA (2013) Secretory protein biogenesis and traffic in the early secretory pathway. Genetics 193:383–410
Becker B, Melkonian M (1996) The secretory pathway of protists: spatial and functional organization and evolution. Microbiol Rev 60:697–721
Behnia R, Barr FA, Flanagan JJ, Barlowe C, Munro S (2007) The yeast orthologue of GRASP65 forms a complex with a coiled-coil protein that contributes to ER to Golgi traffic. J Cell Biol 176:255–261
Bentley M, Liang Y, Mullen K, Xu D, Sztul E, Hay JC (2006) SNARE status regulates tether recruitment and function in homotypic COPII vesicle fusion. J Biol Chem 281:38825–38833
Bevis BJ, Hammond AT, Reinke CA, Glick BS (2002) De novo formation of transitional ER sites and Golgi structures in Pichia pastoris. Nat Cell Biol 4:750–756
Bonifacino JS, Glick BS (2004) The mechanisms of vesicle budding and fusion. Cell 116:153–166
Brigance WT, Barlowe C, Graham TR (2000) Organization of the yeast Golgi complex into at least four functionally distinct compartments. Mol Biol Cell 11:171–182
Chanat E, Huttner WB (1991) Milieu-induced, selective aggregation of regulated secretory proteins in the trans-Golgi network. J Cell Biol 115:1505–1519
Chantalat S, Park SK, Hua Z, Liu K, Gobin R, Peyroche A, Rambourg A, Graham TR, Jackson CL (2004) The Arf activator Gea2p and the P-type ATPase Drs2p interact at the Golgi in Saccharomyces cerevisiae. J Cell Sci 117:711–722
Cosson P, Amherdt M, Rothman JE, Orci L (2002) A resident Golgi protein is excluded from peri-Golgi vesicles in NRK cells. Proc Natl Acad Sci USA 99:12831–12834
Daboussi L, Costaguta G, Payne GS (2012) Phosphoinositide-mediated clathrin adaptor progression at the trans-Golgi network. Nat Cell Biol 14:239–248
De Matteis MA, Luini A (2008) Exiting the Golgi complex. Nat Rev Mol Cell Biol 9:273–284
Dettmer J, Hong-Hermesdorf A, Stierhof YD, Schumacher K (2006) Vacuolar H+-ATPase activity is required for endocytic and secretory trafficking in Arabidopsis. Plant Cell 18:715–730
Dick G, Akslen-Hoel LK, Grøndahl F, Kjos I, Prydz K (2012) Proteoglycan synthesis and Golgi organization in polarized epithelial cells. J Histochem Cytochem 60:926–935
Donohoe BS, Kang BH, Staehelin LA (2007) Identification and characterization of COPIa- and COPIb-type vesicle classes associated with plant and algal Golgi. Proc Natl Acad Sci USA 104:163–168
Donohoe BS, Kang BH, Gerl MJ, Gergely ZR, McMichael CM, Bednarek SY, Staehelin LA (2013) cis-Golgi cisternal assembly and biosynthetic activation occur sequentially in plants and algae. Traffic 14:551–567
Driouich A, Staehelin LA (1997) The plant Golgi apparatus: structural organization and functional properties. In: Berger EG, Roth J (eds) The Golgi apparatus. Birkhäuser, Basel, pp 275–301
Duden R, Schekman R (1997) Insights into Golgi function through mutants in yeast and animal cells. In: Berger EG, Roth J (eds) The Golgi apparatus. Birkhäuser Verlag, Basel, pp 219–246
Dunphy WG, Rothman JE (1985) Compartmental organization of the Golgi stack. Cell 42:13–21
Farquhar MG, Hauri H-P (1997) Protein sorting and vesicular traffic in the Golgi apparatus. In: Berger EG, Roth J (eds) The Golgi apparatus. Birkhäuser Verlag, Basel, pp 63–129
Farquhar MG, Palade GE (1981) The Golgi apparatus (complex)—(1954–1981)—from artifact to center stage. J Cell Biol 91:77s–103s
Faso C, Boulaflous A, Brandizzi F (2009) The plant Golgi apparatus: last 10 years of answered and open questions. FEBS Lett 583:3752–3757
Gilchrist A, Au CE, Hiding J, Bell AW, Fernandez-Rodriguez J, Lesimple S, Nagaya H, Roy L, Gosline SJ, Hallett M, Paiement J, Kearney RE, Nilsson T, Bergeron JJ (2006) Quantitative proteomics analysis of the secretory pathway. Cell 127:1265–1281
Glick BS, Luini A (2011) Models for Golgi traffic: a critical assessment. Cold Spring Harb Perspect Biol 3:a005215
Glick BS, Malhotra V (1998) The curious status of the Golgi apparatus. Cell 95:883–889
Glick BS, Nakano A (2009) Membrane traffic within the Golgi stack. Annu Rev Cell Dev Biol 25:113–132
Glick BS, Elston T, Oster G (1997) A cisternal maturation mechanism can explain the asymmetry of the Golgi stack. FEBS Lett 414:177–181
Goldberg DE, Kornfeld S (1983) Evidence for extensive subcellular organization of asparagine-linked oligosaccharide processing and lysosomal enzyme phosphorylation. J Biol Chem 258:3159–3165
Graham TR, Burd CG (2011) Coordination of Golgi functions by phosphatidylinositol 4-kinases. Trends Cell Biol 21:113–121
Griffiths G, Simons K (1986) The trans Golgi network: sorting at the exit site of the Golgi complex. Science 234:438–443
Hanada K, Kumagai K, Tomishige N, Yamaji T (2009) CERT-mediated trafficking of ceramide. Biochim Biophys Acta 1791:684–691
Hawes C (2005) Cell biology of the plant Golgi apparatus. New Phytol 165:29–44
Jedd G, Richardson CJ, Litt RJ, Segev N (1995) The Ypt1 GTPase is essential for the first two steps of the yeast secretory pathway. J Cell Biol 131:583–590
Jedd G, Mulholland J, Segev N (1997) Two new Ypt GTPases are required for exit from the yeast trans-Golgi compartment. J Cell Biol 137:563–580
Kang BH, Staehelin LA (2008) ER-to-Golgi transport by COPII vesicles in Arabidopsis involves a ribosome-excluding scaffold that is transferred with the vesicles to the Golgi matrix. Protoplasma 234:51–64
Kang BH, Nielsen E, Preuss ML, Mastronarde D, Staehelin LA (2011) Electron tomography of RabA4b- and PI-4 Kβ1-labeled trans Golgi network compartments in Arabidopsis. Traffic 12:313–329
Kinseth MA, Anjard C, Fuller D, Guizzunti G, Loomis WF, Malhotra V (2007) The Golgi associated protein GRASP is required for unconventional secretion during development. Cell 130:524–534
Klausner RD, Donaldson JG, Lippincott-Schwartz J (1992) Brefeldin A: insights into the control of membrane traffic and organelle structure. J Cell Biol 116:1071–1080
Klumperman J (2011) Architecture of the mammalian Golgi. Cold Spring Harb Perspect Biol 3:a005181
Kornfeld R, Kornfeld S (1985) Assembly of asparagine-linked oligosaccharides. Annu Rev Biochem 54:631–664
Kweon HS, Beznoussenko GV, Micaroni M, Polishchuk RS, Trucco A, Martella O, Di Giandomenico D, Marra P, Fusella A, Di Pentima A, Berger EG, Geerts WJ, Koster AJ, Burger KN, Luini A, Mironov AA (2004) Golgi enzymes are enriched in perforated zones of Golgi cisternae but are depleted in COPI vesicles. Mol Biol Cell 15:4710–4724
Ladinsky MS, Mastronarde DN, McIntosh JR, Howell KE, Staehelin LA (1999) Golgi structure in three dimensions: functional insights from the normal rat kidney cell. J Cell Biol 144:1135–1149
Lanoix J, Ouwendijk J, Stark A, Szafer E, Cassel D, Dejgaard K, Weiss M, Nilsson T (2001) Sorting of Golgi resident proteins into different subpopulations of COPI vesicles: a role for ArfGAP1. J Cell Biol 155:1199–1212
Lavieu G, Zheng H, Rothman JE (2013) Stapled Golgi cisternae remain in place as cargo passes through the stack. eLife 2:e00558
Lerich A, Hillmer S, Langhans M, Scheuring D, van Bentum P, Robinson DG (2012) ER import sites and their relationship to ER exit sites: a new model for bidirectional ER-Golgi transport in higher plants. Front Plant Sci 3:143
Levi SK, Bhattacharyya D, Strack RL, Austin JRI, Glick BS (2010) The yeast GRASP Grh1 colocalizes with COPII and is dispensable for organizing the secretory pathway. Traffic 11:1168–1179
Lippincott-Schwartz J, Roberts TH, Hirschberg K (2000) Secretory protein trafficking and organelle dynamics in living cells. Annu Rev Cell Dev Biol 16:557–589
Liu S, Storrie B (2012) Are Rab proteins the link between Golgi organization and membrane trafficking? Cell Mol Life Sci 69:4093–4106
Lord C, Ferro-Novick S, Miller EA (2013) The highly conserved COPII coat complex sorts cargo from the endoplasmic reticulum and targets it to the Golgi. Cold Spring Harb Perspect Biol 5:a013367
Losev E, Reinke CA, Jellen J, Strongin DE, Bevis BJ, Glick BS (2006) Golgi maturation visualized in living yeast. Nature 22:1002–1006
Lowery J, Szul T, Styers M, Holloway Z, Oorschot V, Klumperman J, Sztul E (2013) The Sec7 guanine nucleotide exchange factor GBF1 regulates membrane recruitment of BIG1 and BIG2 guanine nucleotide exchange factors to the trans-Golgi network (TGN). J Biol Chem 288:11532–11545
Machamer CE (1993) Targeting and retention of Golgi proteins. Curr Opin Cell Biol 5:606–612
Malsam J, Söllner TH (2011) Organization of SNAREs within the Golgi stack. Cold Spring Harb Perspect Biol 3:a005249
Malsam J, Satoh A, Pelletier L, Warren G (2005) Golgin tethers define subpopulations of COPI vesicles. Science 307:1095–1098
Marra P, Maffucci T, Daniele T, Di Tullio G, Ikehara Y, Chan EKL, Luini A, Beznoussenko G, Mironov A, De Matteis MA (2001) The GM130 and GRASP65 Golgi proteins cycle through and define a subdomain of the intermediate compartment. Nat Cell Biol 3:1101–1113
Marsh BJ, Mastronarde DN, Buttle KF, Howell KE, McIntosh JR (2001) Organellar relationships in the Golgi region of the pancreatic beta cell line, HIT-T15, visualized by high resolution electron tomography. Proc Natl Acad Sci USA 98:2399–2406
Marsh BJ, Volkmann N, McIntosh JR, Howell KE (2004) Direct continuities between cisternae at different levels of the Golgi complex in glucose-stimulated mouse islet beta cells. Proc Natl Acad Sci USA 101:5565–5570
Martínez-Menárguez JA, Prekeris R, Oorschot VMJ, Scheller R, Slot JW, Geuze HJ, Klumperman J (2001) Peri-Golgi vesicles contain retrograde but not anterograde proteins consistent with the cisternal progression model of intra-Golgi transport. J Cell Biol 155:1213–1224
Matsuura-Tokita K, Takeuchi M, Ichihara A, Mikuriya K, Nakano A (2006) Live imaging of yeast Golgi cisternal maturation. Nature 22:1007–1010
Mellman I, Simons K (1992) The Golgi complex: in vitro veritas? Cell 68:829–840
Mizuno-Yamasaki E, Rivera-Molina F, Novick P (2012) GTPase networks in membrane traffic. Annu Rev Biochem 81:637–659
Moelleken J, Malsam J, Betts MJ, Movafeghi A, Reckmann I, Meissner I, Hellwig A, Russell RB, Söllner T, Brügger B, Wieland FT (2007) Differential localization of coatomer complex isoforms within the Golgi apparatus. Proc Natl Acad Sci USA 104:4425–4430
Mogelsvang S, Gomez-Ospina N, Soderholm J, Glick BS, Staehelin LA (2003) Tomographic evidence for continuous turnover of Golgi cisternae in Pichia pastoris. Mol Biol Cell 14:2277–2291
Mogelsvang S, Marsh BJ, Ladinsky MS, Howell KE (2004) Predicting function from structure: 3D structure studies of the mammalian Golgi complex. Traffic 5:338–345
Mollenhauer HH, Morré DJ (1991) Perspectives on Golgi apparatus form and function. J Electron Microsc Tech 17:2–14
Mollenhauer HH, Whaley WG (1963) An observation on the functioning of the Golgi apparatus. J Cell Biol 17:222–225
Mowbrey K, Dacks JB (2009) Evolution and diversity of the Golgi body. FEBS Lett 583:3738–3745
Munro S (2011) The golgin coiled-coil proteins of the Golgi apparatus. Cold Spring Harb Perspect Biol 3:a005256
Myers MD, Payne GS (2013) Clathrin, adaptors and disease: insights from the yeast Saccharomyces cerevisiae. Front Biosci 18:862–891
Nebenführ A, Ritzenthaler C, Robinson DG (2002) Brefeldin A: deciphering an enigmatic inhibitor of secretion. Plant Physiol 130:1102–1108
Nilsson T, Pypaert M, Hoe MH, Slusarewicz P, Berger EG, Warren G (1993) Overlapping distribution of two glycosyltransferases in the Golgi apparatus of HeLa cells. J Cell Biol 120:5–13
Orci L, Stamnes M, Ravazzola M, Amherdt M, Perrelet A, Söllner TH, Rothman JE (1997) Bidirectional transport by distinct populations of COPI-coated vesicles. Cell 90:335–349
Orci L, Amherdt M, Ravazzola M, Perrelet A, Rothman JE (2000a) Exclusion of Golgi residents from transport vesicles budding from Golgi cisternae in intact cells. J Cell Biol 150:1263–1270
Orci L, Ravazzola M, Volchuk A, Engel T, Gmachl M, Amherdt M, Perrelet A, Söllner TH, Rothman JE (2000b) Anterograde flow of cargo across the Golgi stack potentially mediated via bidirectional “percolating” COPI vesicles. Proc Natl Acad Sci USA 97:10400–10405
Papanikou E, Glick BS (2009) The yeast Golgi apparatus: insights and mysteries. FEBS Lett 583:3746–3751
Parsons HT, Christiansen K, Knierim B, Carroll A, Ito J, Batth TS, Smith-Moritz AM, Morrison S, McInerney P, Hadi MZ, Auer M, Mukhopadhyay A, Petzold CJ, Scheller HV, Loqué D, Heazlewood JL (2012) Isolation and proteomic characterization of the Arabidopsis Golgi defines functional and novel components involved in plant cell wall biosynthesis. Plant Physiol 159:12–26
Patterson GH, Hirschberg K, Polishchuk RS, Gerlich D, Phair RD, Lippincott-Schwartz J (2008) Transport through the Golgi apparatus by rapid partitioning within a two-phase membrane system. Cell 133:1055–1067
Pelham HRB (1988) Evidence that luminal ER proteins are sorted from secreted proteins in a post-ER compartment. EMBO J 7:913–918
Pelham HRB (1998) Getting through the Golgi complex. Trends Cell Biol 8:45–49
Pfeffer SR (2010) How the Golgi works: a cisternal progenitor model. Proc Natl Acad Sci USA 107:19614–19618
Pfeffer SR (2011) Entry at the trans-face of the Golgi. Cold Spring Harb Perspect Biol 3:a005272
Polishchuk RS, Polishchuk EV, Marra P, Alberti S, Buccione R, Luini A, Mironov AA (2000) Correlative light-electron microscopy reveals the tubular-saccular ultrastructure of carriers operating between the Golgi apparatus and plasma membrane. J Cell Biol 148:45–58
Popoff V, Adolf F, Brügger B, Wieland F (2011) COPI budding within the Golgi stack. Cold Spring Harb Perspect Biol 3:a005231
Preuss D, Mulholland J, Franzusoff A, Segev N, Botstein D (1992) Characterization of the Saccharomyces Golgi complex through the cell cycle by immunoelectron microscopy. Mol Biol Cell 3:789–803
Rabouille C, Klumperman J (2005) Opinion: the maturing role of COPI vesicles in intra-Golgi transport. Nat Rev Mol Cell Biol 6:812–817
Rabouille C, Hui N, Hunte F, Kieckbusch R, Berger EG, Warren G, Nilsson T (1995) Mapping the distribution of Golgi enzymes involved in the construction of complex oligosaccharides. J Cell Sci 108:1617–1627
Rambourg A, Clermont Y (1997) Three-dimensional structure of the Golgi apparatus in mammalian cells. In: Berger EG, Roth J (eds) The Golgi apparatus. Birkhäuser Verlag, Basel, pp 37–61
Ren Y, Yip CK, Tripathi A, Huie D, Jeffrey PD, Walz T, Hughson FM (2009) A structure-based mechanism for vesicle capture by the multisubunit tethering complex Dsl1. Cell 139:1119–1129
Richardson BC, McDonold CM, Fromme JC (2012) The Sec7 Arf-GEF is recruited to the trans-Golgi network by positive feedback. Dev Cell 22:799–810
Rink J, Ghigo E, Kalaidzidis Y, Zerial M (2005) Rab conversion as a mechanism of progression from early to late endosomes. Cell 122:735–749
Rizzo R, Parashuraman S, Mirabelli P, Puri C, Lucocq J, Luini A (2013) The dynamics of engineered resident proteins in the mammalian Golgi complex relies on cisternal maturation. J Cell Biol 201:1027–1036
Rockwell NC, Krysan DJ, Komiyama T, Fuller RS (2002) Precursor processing by Kex2/furin proteases. Chem Rev 102:4525–4548
Rothman JE (1981) The Golgi apparatus: two organelles in tandem. Science 213:1212–1219
Rothman JE, Wieland FT (1996) Protein sorting by transport vesicles. Science 272:227–234
Ruiz-May E, Kim SJ, Brandizzi F, Rose JK (2012) The secreted plant N-glycoproteome and associated secretory pathways. Front Plant Sci 3:117
Schmidt WK, Moore HP (1995) Ionic milieu controls the compartment-specific activation of pro-opiomelanocortin processing in AtT-20 cells. Mol Biol Cell 6:1271–1285
Sharpe HJ, Stevens TJ, Munro S (2010) A comprehensive comparison of transmembrane domains reveals organelle-specific properties. Cell 142:158–169
Smith RD, Lupashin VV (2008) Role of the conserved oligomeric Golgi (COG) complex in protein glycosylation. Carbohydr Res 343:2024–2031
Sönnichsen B, Lowe M, Levine T, Jämsä E, Dirac-Svejstrup B, Warren G (1998) A role for giantin in docking COPI vesicles to Golgi membranes. J Cell Biol 140:1013–1021
Spang A (2012) The DSL1 complex: the smallest but not the least CATCHR. Traffic 13:908–913
Spang A, Herrmann JM, Hamamoto S, Schekman R (2001) The ADP ribosylation factor-nucleotide exchange factors Gea1p and Gea2p have overlapping, but not redundant functions in retrograde transport from the Golgi to the endoplasmic reticulum. Mol Biol Cell 12:1035–1045
Staehelin LA, Kang BH (2008) Nanoscale architecture of endoplasmic reticulum export sites and of Golgi membranes as determined by electron tomography. Plant Physiol 147:1454–1468
Stalder D, Antonny B (2013) Arf GTPase regulation through cascade mechanisms and positive feedback loops. FEBS Lett 587:2028–2035
Stanley P (2011) Golgi glycosylation. Cold Spring Harb Perspect Biol 3:a005199
Storrie B, Micaroni M, Morgan GP, Jones N, Kamykowski JA, Wilkins N, Pan TH, Marsh BJ (2012) Electron tomography reveals Rab6 is essential to the trafficking of trans-Golgi clathrin and COPI-coated vesicles and the maintenance of Golgi cisternal number. Traffic 13:727–744
Suvorova ES, Duden R, Lupashin VV (2002) The Sec34/Sec35p complex, a Ypt1p effector required for retrograde intra-Golgi trafficking, interacts with Golgi SNAREs and COPI vesicle coat proteins. J Cell Biol 157:631–643
Trucco A, Polishchuk RS, Martella O, Pentima AD, Fusella A, Giandomenico DD, Pietro ES, Beznoussenko GV, Polishchuk EV, Baldassarre M, Buccione R, Geerts WJ, Koster AJ, Burger KN, Mironov AA, Luini A (2004) Secretory traffic triggers the formation of tubular continuities across Golgi sub-compartments. Nat Cell Biol 6:1071–1081
Tsai PC, Hsu JW, Liu YW, Chen KY, Lee FJ (2013) Arl1p regulates spatial membrane organization at the trans-Golgi network through interaction with Arf-GEF Gea2p and flippase Drs2p. Proc Natl Acad Sci USA 110:E668–E677
Tu L, Tai WC, Chen L, Banfield DK (2008) Signal-mediated dynamic retention of glycosyltransferases in the Golgi. Science 321:404–407
Ungar D, Oka T, Krieger M, Hughson FM (2006) Retrograde transport on the COG railway. Trends Cell Biol 16:113–120
Valdivia RH, Baggott D, Chuang JS, Schekman R (2002) The yeast clathrin adaptor protein complex 1 is required for the efficient retention of a subset of late Golgi membrane proteins. Dev Cell 2:283–294
Velasco A, Hendricks L, Moremen KW, Tulsiani DRP, Touster O, Farquhar MG (1993) Cell type-dependent variations in the subcellular distribution of α-mannosidase I and II. J Cell Biol 122:39–51
Viotti C, Bubeck J, Stierhof YD, Krebs M, Langhans M, van den Berg W, van Dongen W, Richter S, Geldner N, Takano J, Jürgens G, de Vries SC, Robinson DG, Schumacher K (2010) Endocytic and secretory traffic in Arabidopsis merge in the trans-Golgi network/early endosome, an independent and highly dynamic organelle. Plant Cell 22:1344–1357
von Blume J, Alleaume AM, Kienzle C, Carreras-Sureda A, Valverde M, Malhotra V (2012) Cab45 is required for Ca2+-dependent secretory cargo sorting at the trans-Golgi network. J Cell Biol 199:1057–1066
Wakana Y, van Galen J, Meissner F, Scarpa M, Polishchuk RS, Mann M, Malhotra V (2012) A new class of carriers that transport selective cargo from the trans Golgi network to the cell surface. EMBO J 31:3976–3990
Woollard AA, Moore I (2008) The functions of Rab GTPases in plant membrane traffic. Curr Opin Plant Biol 11:610–619
Zeuschner D, Geerts WJ, van Donselaar E, Humbel BM, Slot JW, Koster AJ, Klumperman J (2006) Immuno-electron tomography of ER exit sites reveals the existence of free COPII-coated transport carriers. Nat Cell Biol 8:377–383
Zhang GF, Staehelin LA (1992) Functional compartmentation of the Golgi apparatus of plant cells: immunocytochemical analysis of high-pressure frozen and freeze-substituted sycamore maple suspension culture cells. Plant Physiol 99:1070–1083
Acknowledgments
Thanks to Vivek Malhotra and members of the Glick lab for helpful discussion. This work was supported by U.S. National Institutes of Health grants T32 GM007183 to K.J.D. and R01 GM061156 to B.S.G.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Day, K.J., Staehelin, L.A. & Glick, B.S. A three-stage model of Golgi structure and function. Histochem Cell Biol 140, 239–249 (2013). https://doi.org/10.1007/s00418-013-1128-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-013-1128-3