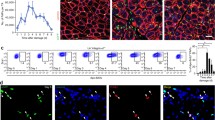
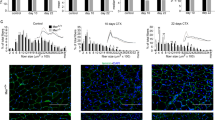
Abstract
Skeletal muscle regeneration in pathology and following injury requires the coordinated actions of inflammatory cells and myogenic cells to remove damaged tissue and rebuild syncytial muscle cells, respectively. Following contusion injury to muscle, the cytokine leukemia inhibitor factor (LIF) is up-regulated and knockout of Lif negatively impacts on morphometric parameters of muscle regeneration. Although it was speculated that LIF regulates muscle regeneration through direct effects on myogenic cells, the inflammatory effects of LIF have not been examined in regenerating skeletal muscle. Therefore, the expression and function of LIF was examined using the antagonist MH35-BD during specific inflammatory and myogenic stages of notexin-induced muscle regeneration in mice. LIF protein and mRNA were up-regulated in two distinct phases following intramuscular injection of notexin into tibialis anterior muscles. The first phase of LIF up-regulation coincided with the increased expression of pro-inflammatory cytokines; the second phase coincided with myogenic differentiation and formation of new myotubes. Administration of the LIF receptor antagonist MH35-BD during the second phase of LIF up-regulation had no significant effects on transcript expression of genes required for myogenic differentiation or associated with inflammation; there were no significant differences in morphometric parameters of the regenerating muscle. Conversely, when MH35-BD was administered during the acute inflammatory phase, increased gene transcripts for the pro-inflammatory cytokines Tnf (Tumor necrosis factor), Il1b (Interleukin-1β) and Il6 (Interleukin-6) alongside an increase in the number of Ly6G positive neutrophils infiltrating the muscle were observed. This was followed by a reduction in Myog (Myogenin) mRNA, which is required for myogenic differentiation, and the subsequent number of myotubes formed was significantly decreased in MH35-BD-treated groups compared to sham. Thus, antagonism of the LIF receptor during the inflammatory phase of skeletal muscle regeneration appeared to induce an inflammatory response that inhibited subsequent myotube formation. We propose that the predominant role of LIF in skeletal muscle regeneration appears to be in regulating the inflammatory response rather than directly effecting myogenic cells.










Similar content being viewed by others
References
Akita S, Malkin J, Melmed S (1996) Disrupted murine leukemia inhibitory factor (LIF) gene attenuates adrenocorticotropic hormone (ACTH) secretion. Endocrinology 137:3140
Alter J, Rozentzweig D, Bengal E (2008) Inhibition of myoblast differentiation by tumor necrosis factor is mediated by c-Jun N-terminal Kinase 1 and leukemia inhibitory factor. J Biol Chem 283:23224
Alvarez B, Quinn LBS, Busquets S, Quiles MT, López-Soriano FJ, Argilés JM (2002) Tumor necrosis factor-[alpha] exerts interleukin-6-dependent and-independent effects on cultured skeletal muscle cells. Biochimica et Biophysica Acta (BBA)-Mol Cell Res 1542:66–72
Austin L, Burgess A (1991) Stimulation of myoblast proliferation in culture by leukaemia inhibitory factor and other cytokines. J Neurol Sci 101:193–197
Austin L, Bower J, Kurek J, Vakakis N (1992) Effects of leukaemia inhibitory factor and other cytokines on murine and human myoblast proliferation. J Neurol Sci 112:185–191
Austin L, Bower J, Bennett T, Lynch G, Kapsa R, White J, Barnard W, Gregorevic P, Byrne E (2000) Leukemia inhibitory factor ameliorates muscle fiber degeneration in the mdx mouse. Muscle Nerve 23:1700–1705
Bamberger AM, Schulte HM, Wullbrand A, Jung R, Beil FU, Bamberger CM (2000) Expression of leukemia inhibitory factor (LIF) and LIF receptor (LIF-R) in the human adrenal cortex: implications for steroidogenesis. Mol Cell Endocrinol 162:145–149
Banner LR, Patterson PH, Allchorne A, Poole S, Woolf CJ (1998) Leukemia inhibitory factor is an anti-inflammatory and analgesic cytokine. J Neurosci 18:5456
Barnard W, Bower J, Brown MA, Murphy M, Austin L (1994) Leukemia inhibitory factor (LIF) infusion stimulates skeletal muscle regeneration after injury: injured muscle expresses lif mRNA. J Neurol Sci 123:108–113
Bower J, Vakakis N, Nicola N, Austin L (1995) Specific binding of leukemia inhibitory factor to murine myoblasts in culture. J Cell Physiol 164:93–98
Briguet A, Courdier-Fruh I, Foster M, Meier T, Magyar JP (2004) Histological parameters for the quantitative assessment of muscular dystrophy in the mdx-mouse. Neuromuscul Disord 14:675–682
Broussard SR, McCusker RH, Novakofski JE, Strle K, Shen WH, Johnson RW, Dantzer R, Kelley KW (2004) IL-1 impairs insulin-like growth factor I-induced differentiation and downstream activation signals of the insulin-like growth factor I receptor in myoblasts. J Immunol 172:7713
Contreras-Shannon V, Ochoa O, Reyes-Reyna SM, Sun D, Michalek JE, Kuziel WA, McManus LM, Shireman PK (2007) Fat accumulation with altered inflammation and regeneration in skeletal muscle of CCR2–/–mice following ischemic injury. Am J Physiol-Cell Physiol 292:C953
Duluc D, Delneste Y, Tan F, Moles MP, Grimaud L, Lenoir J, Preisser L, Anegon I, Catala L, Ifrah N (2007) Tumor-associated leukemia inhibitory factor and IL-6 skew monocyte differentiation into tumor-associated macrophage-like cells. Blood 110:4319–4330
Fairlie WD, Uboldi AD, McCoubrie JE, Wang CC, Lee EF, Yao S, De Souza DP, Mifsud S, Metcalf D, Nicola NA (2004) Affinity maturation of leukemia inhibitory factor and conversion to potent antagonists of signaling. J Biol Chem 279:2125
Fielding R, Manfredi T, Ding W, Fiatarone M, Evans W, Cannon J (1993) Acute phase response in exercise. III. Neutrophil and IL-1 beta accumulation in skeletal muscle. Am J Physiol-Regul, Integr Comp Physiol 265:R166
Gadient RA, Patterson PH (1999) Leukemia inhibitory factor, interleukin 6, and other cytokines using the GP130 transducing receptor: roles in inflammation and injury. Stem Cells 17:127–137
Galli SJ, Zsebo KM, Geissler EN (1993) The kit ligand, stem cell factor. Adv Immunol 55:1–96
Gearing DP, Gough N, King J, Hilton D, Nicola N, Simpson R, Nice E, Kelso A, Metcalf D (1987) Molecular cloning and expression of cDNA encoding a murine myeloid leukaemia inhibitory factor (LIF). EMBO J 6:3995
Gearing DP, Comeau MR, Friend DJ, Gimpel SD, Thut CJ, McGourty J, Brasher KK, King JA, Gillis S, Mosley B (1992) The IL-6 signal transducer, gp130: an oncostatin M receptor and affinity converter for the LIF receptor. Science 255:1434
Gordon S (2003) Alternative activation of macrophages. Nat Rev Immunol 3:23–35
Gorospe JRM, Tharp M, Demitsu T, Hoffman EP (1994) Dystrophin-deficient myofibers are vulnerable to mast cell granule-induced necrosis. Neuromuscul Disord 4:325–333
Gregorevic P, Hayes A, Lynch GS, Williams DA (2000) Functional properties of regenerating skeletal muscle following LIF administration. Muscle Nerve 23:1586–1588
Helliwell T, Gunhan O, Edwards R (1990) Mast cells in neuromuscular diseases. J Neurol Sci 98:267–276
Hendriks JJA, Slaets H, Carmans S, de Vries HE, Dijkstra CD, Stinissen P, Hellings N (2008) Leukemia inhibitory factor modulates production of inflammatory mediators and myelin phagocytosis by macrophages. J Neuroimmunol 204:52–57
Hilton D, Nicola N, Metcalf D (1988) Specific binding of murine leukemia inhibitory factor to normal and leukemic monocytic cells. Proc Nat Acad Sci 85:5971
Hilton DJ, Nicola NA, Waring PM, Metcalf D (1991) Clearance and fate of leukemia inhibitory factor (LIF) after injection into mice. J Cell Physiol 148:430–439
Hodgetts S, Radley H, Davies M, Grounds MD (2006) Reduced necrosis of dystrophic muscle by depletion of host neutrophils, or blocking TNFα function with Etanercept in mdx mice. Neuromuscul Disord : NMD 16:591–602
Holtmann B, Wiese S, Samsam M, Grohmann K, Pennica D, Martini R, Sendtner M (2005) Triple knock-out of CNTF, LIF, and CT-1 defines cooperative and distinct roles of these neurotrophic factors for motoneuron maintenance and function. J Neurosci 25:1778
Hunt L, Tudor E, White J (2010) Leukemia inhibitory factor-dependent increase in myoblast cell number is associated with phosphotidylinositol 3-kinase-mediated inhibition of apoptosis and not mitosis. Exp Cell Res 316:1002–1009
Hunt LC, Upadhyay A, Jazayeri JA, Tudor EM, White JD (2011) Caspase-3, myogenic transcription factors and cell cycle inhibitors are regulated by leukemia inhibitory factor to mediate inhibition of myogenic differentiation. Skeletal Muscle 1:17
Iida N, Grotendorst GR (1990) Cloning and sequencing of a new gro transcript from activated human monocytes: expression in leukocytes and wound tissue. Mol Cell Biol 10:5596
Jazayeri JA, De Weerd N, Raye W, Kivivuori S, Zabihi E, Carroll GJ (2007) In vitro evaluation of leukemia inhibitory factor receptor antagonists as candidate therapeutics for inflammatory arthritis. J Interferon Cytokine Res 27:281–290
Jo C, Kim H, Jo I, Choi I, Jung SC, Kim J, Kim SS, Jo SA (2005) Leukemia inhibitory factor blocks early differentiation of skeletal muscle cells by activating ERK. Biochimica et Biophysica Acta (BBA)-Molecul Cell Res 1743:187–197
Kovanen PT, Kaartinen M, Paavonen T (1995) Infiltrates of activated mast cells at the site of coronary atheromatous erosion or rupture in myocardial infarction. Circulation 92:1084–1088
Kurek JB, Nouri S, Kannourakis G, Murphy M, Austin L (1996) Leukemia inhibitory factor and interleukin-6 are produced by diseased and regenerating skeletal muscle. Muscle Nerve 19:1291–1301
Kurek JB, Bower JJ, Romanella M, Koentgen F, Murphy M, Austin L (1997) The role of leukemia inhibitory factor in skeletal muscle regeneration. Muscle Nerve 20:815–822
Kwon YW, Abbondanzo SJ, Stewart CL, Gurney ME (1995) Leukemia inhibitory factor influences the timing of programmed synapse withdrawal from neonatal muscles. J Neurobiol 28:35–50
Layton MJ, Owczarek CM, Metcalf D, Clark RL, Smith DK, Treutlein HR, Nicola NA (1994) Conversion of the biological specificity of murine to human leukemia inhibitory factor by replacing 6 amino acid residues. J Biol Chem 269:29891–29896
Lefaucheur JP, Sebille A (1995) Muscle regeneration following injury can be modified in vivo by immune neutralization of basic fibroblast growth factor, transforming growth factor [beta] 1 or insulin-like growth factor I. J Neuroimmunol 57:85–91
Li YP (2003) TNF- is a mitogen in skeletal muscle. Am J Physiol-Cell Physiol 285:C370
Metcalf D, Hilton D, Nicola N (1988) Clonal analysis of the actions of the murine leukemia inhibitory factor on leukemic and normal murine hemopoietic cells. Leuk Off J Leuk Soc Am, Leuk Res Fund, UK 2:216
Miyake T, Alli NS, Aziz A, Knudson J, Fernando P, Megeney LA, McDermott JC (2009) Cardiotrophin-1 maintains the undifferentiated state in skeletal myoblasts. J Biol Chem 284:19679
Moritz DR, Rodewald H-R, Gheyselinck J, Klemenz R (1998) The IL-1 receptor-related T1 antigen is expressed on immature and mature mast cells and on fetal blood mast cell progenitors. J Immunol 161:4866–4874
Nishikawa J, Sakuma K, Sorimachi Y, Yoshimoto K, Yasuhara M (2005) Increase of cardiotrophin 1 immunoreactivity in regenerating and overloaded but not denervated muscles of rats. Neuropathology 25:54–65
Ochoa O, Sun D, Reyes-Reyna SM, Waite LL, Michalek JE, McManus LM, Shireman PK (2007) Delayed angiogenesis and VEGF production in CCR2–/–mice during impaired skeletal muscle regeneration. Am J Physiol Regul Integrat Comp Physiol 293:R651
Pelus LM, Fukuda S (2006) Peripheral blood stem cell mobilization: the CXCR2 ligand GRO [beta] rapidly mobilizes hematopoietic stem cells with enhanced engraftment properties. Exp Hematol 34:1010–1020
Peterson JM, Feeback KD, Baas JH, Pizza FX (2006) Tumor necrosis factor-α promotes the accumulation of neutrophils and macrophages in skeletal muscle. J Appl Physiol 101:1394
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Radley HG, Grounds MD (2006) Cromolyn administration (to block mast cell degranulation) reduces necrosis of dystrophic muscle in mdx mice. Neurobiol Dis 23:387–397
Ray DW, Ren SG, Melmed S (1996) Leukemia inhibitory factor (LIF) stimulates proopiomelanocortin (POMC) expression in a corticotroph cell line. Role of STAT pathway. J Clin Investig 97:1852
Schofield G, Kimber SJ (2005) Leukocyte subpopulations in the uteri of leukemia inhibitory factor knockout mice during early pregnancy. Biol Reprod 72:872–878
Shireman PK, Contreras-Shannon V, Ochoa O, Karia BP, Michalek JE, McManus LM (2007) MCP-1 deficiency causes altered inflammation with impaired skeletal muscle regeneration. J Leukoc Biol 81:775
Spencer MJ, Montecino-Rodriguez E, Dorshkind K, Tidball JG (2001) Helper (CD4+) and cytotoxic (CD8+) T cells promote the pathology of dystrophin-deficient muscle. Clin Immunol 98:235–243
Srikuea R, Esser KA, Pholpramool C (2011) Leukaemia inhibitory factor is expressed in rat gastrocnemius muscle after contusion and increases proliferation of rat L6 myoblasts via c-Myc signalling. Clin Exp Pharmacol Physiol 38:501–509
Tham S, Dowsing B, Finkelstein D, Donato R, Cheema SS, Bartlett PF, Morrison WA (1997) Leukemia inhibitory factor enhances the regeneration of transected rat sciatic nerve and the function of reinnervated muscle. J Neurosci Res 47:208–215
Tidball JG, Villalta SA (2010) Regulatory interactions between muscle and the immune system during muscle regeneration. Am J Physiol Regul Integrat Comp Physiol 298:R1173
Upadhyay A, Sharma G, Kivivuori S, Raye WS, Zabihi E, Carroll GJ, Jazayeri JA (2009) Role of a LIF antagonist in LIF and OSM induced MMP-1, MMP-3, and TIMP-1 expression by primary articular chondrocytes. Cytokine 46:332–338
Villalta SA, Rinaldi C, Deng B, Liu G, Fedor B, Tidball JG (2011) Interleukin-10 reduces the pathology of mdx muscular dystrophy by deactivating M1 macrophages and modulating macrophage phenotype. Hum Mol Genet 20:790–805
Wang X, Wu H, Zhang Z, Liu S, Yang J, Chen X, Fan M (2008) Effects of interleukin-6, leukemia inhibitory factor, and ciliary neurotrophic factor on the proliferation and differentiation of adult human myoblasts. Cell Mol Neurobiol 28:113–124
Wehling M, Spencer MJ, Tidball JG (2001) A nitric oxide synthase transgene ameliorates muscular dystrophy in mdx mice. J Cell Biol 155:123–132
White JD, Davies M, Grounds MD (2001) Leukaemia inhibitory factor increases myoblast replication and survival and affects extracellular matrix production: combined in vivo and in vitro studies in post-natal skeletal muscle. Cell Tissue Res 306:129–141
White JD, Davies M, McGeachie J, Grounds MD (2002) An evaluation of leukaemia inhibitory factor as a potential therapeutic agent in the treatment of muscle disease. Neuromuscul Disord 12:909–916
Wolpe SD, Sherry B, Juers D, Davatelis G, Yurt RW, Cerami A (1989) Identification and characterization of macrophage inflammatory protein 2. Proc Nat Acad Sci 86:612
Woods AM, McIlmoil CJ, Rankin EN, Packer AA, Stevens JC, Macievic JA, Brown AB, Porter JP, Judd AM (2008) Leukemia inhibitory factor protein and receptors are expressed in the bovine adrenal cortex and increase cortisol and decrease adrenal androgen release. Domest Anim Endocrinol 35:217–230
Zentella A, Massague J (1992) Transforming growth factor beta induces myoblast differentiation in the presence of mitogens. Proc Nat Acad Sci 89:5176
Acknowledgments
The authors acknowledge the contributions of Kitipong Uaesoontrachoon and Charles Pagel for providing oligonucleotide primer sequences for the genes Myod1 and Myog. Su Toulson should also be acknowledged for her contributions toward the sampling of mice and subsequent processing of those samples. This work was funded by the Muscular Dystrophy Association of Australia (MDA) and supported by the Victorian Government’s Operational Infrastructure Support Program.
Conflict of interest
The authors declare that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.

418_2012_1018_MOESM1_ESM.jpg
Fig. S1 Quantitation of LIF in serum following intramuscular notexin injection by LIF ELISA. Data were analysed by Student’s t test comparing individual time points to uninjured (day 0), * represents P < 0.05, n = 5 (JPEG 502 kb)

418_2012_1018_MOESM2_ESM.jpg
Fig. S2 Relative mRNA levels for lymphocyte associated markers with notexin-induced regeneration following (a) MH35-BD administration at day 3 and (b) MH35-BD administration at day 0. Data were analysed by REST comparing MH35-BD to sham, * represents P < 0.05, n = 5 (JPEG 170 kb)

418_2012_1018_MOESM3_ESM.jpg
Fig. S3 Relative mRNA levels of Pcna with MH35-BD administration at day 0. Data were analysed by REST comparing MH35-BD to sham, * represents P < 0.05, n = 5 (JPEG 424 kb)

418_2012_1018_MOESM4_ESM.jpg
Fig. S4 A cross section of a tibialis anterior muscle 7 days after notexin injection immunofluorescently labeled for LIF without blocking peptide (−BP). A cross section of a tibialis anterior muscle 7 days after notein injection immunofluorescently labeled for LIF with the inclusion of the anti-LIF blocking peptide during incubation of the primary antibody (+BP) (JPEG 1476 kb)

418_2012_1018_MOESM5_ESM.jpg
Fig. S5 Relative mRNA levels of Lif with MH35-BD administration at a day 0 and b day 3. The same trend was observed for Lif expression as with Fig. 1B (JPEG 382 kb)
Rights and permissions
About this article
Cite this article
Hunt, L.C., Upadhyay, A., Jazayeri, J.A. et al. An anti-inflammatory role for leukemia inhibitory factor receptor signaling in regenerating skeletal muscle. Histochem Cell Biol 139, 13–34 (2013). https://doi.org/10.1007/s00418-012-1018-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-012-1018-0