Abstract
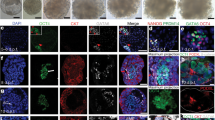
Human embryonic stem cell (hESC) differentiation in embryoid bodies (EBs) provides a valuable tool to study the interplay of different germ layers and their influence on cell differentiation. The gene expression of the developing EBs has been shown in many studies, but the protein expression and the spatial composition of different germ layers in human EBs have not been systematically studied. The aim of the present work was to study the temporal and spatial organisation of germ layers based on the expression of mesoderm (Brachyury T), endoderm (AFP) and ectoderm (SOX1) markers during the early stages of differentiation in eight hESC lines. Tissue multi-array technology was applied to study the protein expression of a large number of EBs. According to our results, EB formation and the organisation of germ layers occurred in a similar manner in all the lines. During 12 days of differentiation, all the germ layer markers were present, but no obvious distinct trajectories were formed. However, older EBs were highly organised in structure. Pluripotency marker OCT3/4 expression persisted unexpectedly long in the differentiating EBs. Cavity formation was observed in the immunocytological sections, and caspase-3 expression was high, suggesting a role of apoptosis in hESC differentiation and/or EB formation. The expression of Brachyury T was notably low in all the lines, also those with the best cardiac differentiation capacity, while the expression of SOX1 was higher in some lines, suggesting that the neural differentiation propensity may be detectable already in the early stages of EB differentiation.





Similar content being viewed by others
References
Adewumi O, Aflatoonian B, Ahrlund-Richter L, Amit M, Andrews PW, Beighton G, Bello PA, Benvenisty N, Berry LS, Bevan S, Blum B, Brooking J, Chen KG, Choo AB, Churchill GA, Corbel M, Damjanov I, Draper JS, Dvorak P, Emanuelsson K, Fleck RA, Ford A, Gertow K, Gertsenstein M, Gokhale PJ, Hamilton RS, Hampl A, Healy LE, Hovatta O, Hyllner J, Imreh MP, Itskovitz-Eldor J, Jackson J, Johnson JL, Jones M, Kee K, King BL, Knowles BB, Lako M, Lebrin F, Mallon BS, Manning D, Mayshar Y, McKay RD, Michalska AE, Mikkola M, Mileikovsky M, Minger SL, Moore HD, Mummery CL, Nagy A, Nakatsuji N, O’Brien CM, Oh SK, Olsson C, Otonkoski T, Park KY, Passier R, Patel H, Patel M, Pedersen R, Pera MF, Piekarczyk MS, Pera RA, Reubinoff BE, Robins AJ, Rossant J, Rugg-Gunn P, Schulz TC, Semb H, Sherrer ES, Siemen H, Stacey GN, Stojkovic M, Suemori H, Szatkiewicz J, Turetsky T, Tuuri T, van den Brink S, Vintersten K, Vuoristo S, Ward D, Weaver TA, Young LA, Zhang W (2007) Characterization of human embryonic stem cell lines by the International Stem Cell Initiative. Nat Biotechnol 25:803–816
Amit M, Carpenter MK, Inokuma MS, Chiu CP, Harris CP, Waknitz MA, Itskovitz-Eldor J, Thomson JA (2000) Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev Biol 227:271–278
Bauwens CL, Peerani R, Niebruegge S, Woodhouse KA, Kumacheva E, Husain M, Zandstra PW (2008) Control of human embryonic stem cell colony and aggregate size heterogeneity influences differentiation trajectories. Stem Cells 26:2300–2310
Beqqali A, Kloots J, Ward-van Oostwaard D, Mummery C, Passier R (2006) Genome-wide transcriptional profiling of human embryonic stem cells differentiating to cardiomyocytes. Stem Cells 24(8):1956–1967
Bettiol E, Sartiani L, Chicha L, Krause KH, Cerbai E, Jaconi ME (2007) Fetal bovine serum enables cardiac differentiation of human embryonic stem cells. Differentiation 75:669–681
Burridge PW, Anderson D, Priddle H, Barbadillo Munoz MD, Chamberlain S, Allegrucci C, Young LE, Denning C (2007) Improved human embryonic stem cell embryoid body homogeneity and cardiomyocyte differentiation from a novel V-96 plate aggregation system highlights interline variability. Stem Cells 25:929–938
Cao F, Wagner RA, Wilson KD, Xie X, Fu JD, Drukker M, Lee A, Li RA, Gambhir SS, Weissman IL, Robbins RC, Wu JC (2008) Transcriptional and functional profiling of human embryonic stem cell-derived cardiomyocytes. PLoS One 3:e3474
Conley BJ, Trounson AO, Mollard R (2004) Human embryonic stem cells form embryoid bodies containing visceral endoderm-like derivatives. Fetal Diagn Ther 19:218–223
Dvash T, Mayshar Y, Darr H, McElhaney M, Barker D, Yanuka O, Kotkow KJ, Rubin LL, Benvenisty N, Eiges R (2004) Temporal gene expression during differentiation of human embryonic stem cells and embryoid bodies. Hum Reprod 19:2875–2883
Fujita J, Crane AM, Souza MK, Dejosez M, Kyba M, Flavell RA, Thomson JA, Zwaka TP (2008) Caspase activity mediates the differentiation of embryonic stem cells. Cell Stem Cell 2:595–601
Graichen R, Xu X, Braam SR, Balakrishnan T, Norfiza S, Sieh S, Soo SY, Tham SC, Mummery C, Colman A, Zweigerdt R, Davidson BP (2008) Enhanced cardiomyogenesis of human embryonic stem cells by a small molecular inhibitor of p38 MAPK. Differentiation 76:357–370
Hoffman LM, Carpenter MK (2005) Characterization and culture of human embryonic stem cells. Nat Biotechnol 23:699–708
Hsiao EC, Yoshinaga Y, Nguyen TD, Musone SL, Kim JE, Swinton P, Espineda I, Manalac C, deJong PJ, Conklin BR (2008) Marking embryonic stem cells with a 2A self-cleaving peptide: a NKX2-5 emerald GFP BAC reporter. PLoS One 3:e2532
Itskovitz-Eldor J, Schuldiner M, Karsenti D, Eden A, Yanuka O, Amit M, Soreq H, Benvenisty N (2000) Differentiation of human embryonic stem cells into embryoid bodies compromising the three embryonic germ layers. Mol Med 6:88–95
Jacobson MD, Weil M, Raff MC (1997) Programmed cell death in animal development. Cell 88:347–354
Joza N, Susin SA, Daugas E, Stanford WL, Cho SK, Li CY, Sasaki T, Elia AJ, Cheng HY, Ravagnan L, Ferri KF, Zamzami N, Wakeham A, Hakem R, Yoshida H, Kong YY, Mak TW, Zuniga-Pflucker JC, Kroemer G, Penninger JM (2001) Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 410:549–554
Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A, Livne E, Binah O, Itskovitz-Eldor J, Gepstein L (2001) Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest 108:407–414
Keller GM (1995) In vitro differentiation of embryonic stem cells. Curr Opin Cell Biol 7:862–869
Kim SE, Kim BK, Gil JE, Kim SK, Kim JH (2007) Comparative analysis of the developmental competence of three human embryonic stem cell lines in vitro. Mol Cells 23:49–56
Laflamme MA, Chen KY, Naumova AV, Muskheli V, Fugate JA, Dupras SK, Reinecke H, Xu C, Hassanipour M, Police S, O’Sullivan C, Collins L, Chen Y, Minami E, Gill EA, Ueno S, Yuan C, Gold J, Murry CE (2007) Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol 25:1015–1024
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Lough J, Sugi Y (2000) Endoderm and heart development. Dev Dyn 217:327–342
Messina E, De Angelis L, Frati G, Morrone S, Chimenti S, Fiordaliso F, Salio M, Battaglia M, Latronico MVG, Coletta M, Vivarelli E, Frati L, Cossu G, Giacomello A (2004) Isolation and expansion of adult cardiac stem cells from human and murine heart. Circ Res 95:911–921
Mohr JC, Zhang J, Azarin SM, Soerens AG, de Pablo JJ, Thomson JA, Lyons GE, Palecek SP, Kamp TJ (2010) The microwell control of embryoid body size in order to regulate cardiac differentiation of human embryonic stem cells. Biomaterials 31:1885–1893
Mummery C, Ward-van Oostwaard D, Doevendans P, Spijker R, van den Brink S, Hassink R, van der Heyden M, Opthof T, Pera M, de la Riviere AB, Passier R, Tertoolen L (2003) Differentiation of human embryonic stem cells to cardiomyocytes: role of coculture with visceral endoderm-like cells. Circulation 107:2733–2740
Niebruegge S, Bauwens CL, Peerani R, Thavandiran N, Masse S, Sevaptisidis E, Nanthakumar K, Woodhouse K, Husain M, Kumacheva E, Zandstra PW (2009) Generation of human embryonic stem cell-derived mesoderm and cardiac cells using size-specified aggregates in an oxygen-controlled bioreactor. Biotechnol Bioeng 102:493–507
Niwa H, Miyazaki J, Smith AG (2000) Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat Genet 24:372–376
Osafune K, Caron L, Borowiak M, Martinez RJ, Fitz-Gerald CS, Sato Y, Cowan CA, Chien KR, Melton DA (2008) Marked differences in differentiation propensity among human embryonic stem cell lines. Nat Biotechnol 26:313–315
Pal R, Totey SS, Mamidi MK, Bhat V, Totey SM (2009) Propensity of human embryonic stem cell lines during early stage of lineage specification control their terminal differentiation into mature cell types. Exp Biol Med 234(10):1230–1243
Palmieri SL, Peter W, Hess H, Scholer HR (1994) Oct-4 transcription factor is differentially expressed in the mouse embryo during establishment of the first two extraembryonic cell lineages involved in implantation. Dev Biol 166:259–267
Parvinen M, Pelto-Huikko M, Soder O, Schultz R, Kaipia A, Mali P, Toppari J, Hakovirta H, Lonnerberg P, Ritzen EM (1992) Expression of beta-nerve growth factor and its receptor in rat seminiferous epithelium: specific function at the onset of meiosis. J Cell Biol 117:629–641
Passier R, Oostwaard DW, Snapper J, Kloots J, Hassink RJ, Kuijk E, Roelen B, de la Riviere AB, Mummery C (2005) Increased cardiomyocyte differentiation from human embryonic stem cells in serum-free cultures. Stem Cells 23:772–780
Pekkanen-Mattila M, Kerkela E, Tanskanen JM, Pietila M, Pelto-Huikko M, Hyttinen J, Skottman H, Suuronen R, Aalto-Setala K (2009) Substantial variation in the cardiac differentiation of human embryonic stem cell lines derived and propagated under the same conditions—a comparison of multiple cell lines. Ann Med 41:360–370
Pera MF, Trounson AO (2004) Human embryonic stem cells: prospects for development. Development 131:5515–5525
Sachlos E, Auguste DT (2008) Embryoid body morphology influences diffusive transport of inductive biochemicals: a strategy for stem cell differentiation. Biomaterials 29:4471–4480
Synnergren J, Adak S, Englund MCO, Giesler TL, Noaksson K, Lindahl A, Nilsson P, Nelson D, Abbot S, Olsson B, Sartipy P (2008) Cardiomyogenic gene expression profiling of differentiating human embryonic stem cells. J Biotechnol 134:162–170
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282:1145–1147
Xu C, Police S, Rao N, Carpenter MK (2002) Characterization and enrichment of cardiomyocytes derived from human embryonic stem cells. Circ Res 91:501–508
Xu XQ, Graichen R, Soo SY, Balakrishnan T, Rahmat SN, Sieh S, Tham SC, Freund C, Moore J, Mummery C, Colman A, Zweigerdt R, Davidson BP (2008) Chemically defined medium supporting cardiomyocyte differentiation of human embryonic stem cells. Differentiation 76:958–970
Yang L, Soonpaa MH, Adler ED, Roepke TK, Kattman SJ, Kennedy M, Henckaerts E, Bonham K, Abbott GW, Linden RM, Field LJ, Keller GM (2008) Human cardiovascular progenitor cells develop from a KDR+ embryonic-stem-cell-derived population. Nature 453:524–528
Yirme G, Amit M, Laevsky I, Osenberg S, Itskovitz-Eldor J (2008) Establishing a dynamic process for the formation, propagation, and differentiation of human embryoid bodies. Stem Cells Dev 17:1227–1241
Zandstra PW, Bauwens C, Yin T, Liu Q, Schiller H, Zweigerdt R, Pasumarthi KB, Field LJ (2003) Scalable production of embryonic stem cell-derived cardiomyocytes. Tissue Eng 9:767–778
Zeineddine D, Papadimou E, Chebli K, Gineste M, Liu J, Grey C, Thurig S, Behfar A, Wallace VA, Skerjanc IS, Puceat M (2006) Oct-3/4 dose dependently regulates specification of embryonic stem cells toward a cardiac lineage and early heart development. Dev Cell 11:535–546
Zhang J, Wilson GF, Soerens AG, Koonce CH, Yu J, Palecek SP, Thomson JA, Kamp TJ (2009) Functional cardiomyocytes derived from human induced pluripotent stem cells. Circ Res 104:e30–e41
Acknowledgments
We thank the heart team and the hESC maintenance laboratory of Regea. We are grateful for the technical assistance of Ulla Jukarainen in immunocytochemistry. We thank Prof. Outi Hovatta for the HS lines. The study was funded by the Academy of Finland, the Finnish Heart Research Foundation, the Finnish Cultural Foundation, the Competitive Research Funding of Pirkanmaa Hospital District, the Kalle Kaihari Foundation and the Ida Montin Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
418_2010_689_MOESM1_ESM.pdf
Supplementary Fig. 1. A tissue multi-array was prepared by punching 1 mm holes in 10% agarose. EBs were individually transferred to the wells filled with OCT compound, i.e. each hole contains replicates for individual samples from different hESC lines at different time points (PDF 238 kb)
418_2010_689_MOESM2_ESM.pdf
Supplementary Fig. 2. Average morphological changes and growth of EBs at the age of 1–20 days from four hESC lines. Scale bar 200 μm (PDF 623 kb)
418_2010_689_MOESM3_ESM.pdf
Supplementary Fig. 3. OCT3/4 shown to be strongly expressed at the protein level in undifferentiated hESC colonies (A). Infrequently single Brachyury T positive cells were seen (B), but the expression of SOX1 or AFP was never detected (C, D). Corresponding 4′,6-diamidino-2-phenylindole (DAPI) stainings (E–H). Scale bar 200 μm (PDF 301 kb)
418_2010_689_MOESM4_ESM.pdf
Supplementary Fig. 4. OCT3/4 protein expression in EBs from hESC lines HS181 and HS346. The expression of OCT3/4 is still quite high on day 4 and can be detected both in the middle of EBs and on the edges (A, B). A strongly OCT3/4 positive area in one 6-day-old EB from line HS181 (C) and more typical expression pattern in EB from HS346 (D). In general, the expression of OCT3/4 started to decrease from day 8 (E, G), although was still quite abundant in some EBs on day 8 (F) and day 10 (H). The site of inset is marked with black box showing the stained cells except in G, where no immunoreactivity was seen. Cavity formation is marked with asterisks in D. Scale bar 50 μm (PDF 289 kb)
418_2010_689_MOESM5_ESM.pdf
Supplementary Fig. 5. Beating aggregate consisting mainly of cardiomyocytes as almost all the cells stained positively with cardiac troponin T (A). Clear striated patterns in the cells can be seen in the inset (B) (arrows). Scale bar 50 μm (PDF 62.3 kb)
Rights and permissions
About this article
Cite this article
Pekkanen-Mattila, M., Pelto-Huikko, M., Kujala, V. et al. Spatial and temporal expression pattern of germ layer markers during human embryonic stem cell differentiation in embryoid bodies. Histochem Cell Biol 133, 595–606 (2010). https://doi.org/10.1007/s00418-010-0689-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-010-0689-7