Abstract
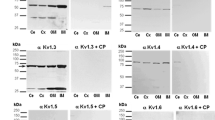
Several vectorial transport routes in mammalian cells involve clathrin and associated proteins. In kidney epithelia urine production requires numerous transport processes. However, only little is known about the distribution of clathrin and its associated proteins in this organ in situ. We now report on the presence and distribution of clathrin and its accessory proteins AP1, AP2, Eps15, Epsin, CALM and Clint/EpsinR in the epithelia of the rat kidney cortex using immunoblotting, immunofluorescence and immuno-electron microscopy. Our data show that all investigated proteins are ubiquitously present in rat kidney cortex epithelia, however, with distinct distribution patterns. In the renal corpuscle, podocytes showed the most conspicuous labelling. Clathrin, AP2 and CALM were highly expressed in foot processes, while AP1 was primarily localized in the cell body. In the proximal tubule all proteins were present in dots along the plasma membrane and most conspicuous below the brush border. However, clathrin and AP2 co-localized in vesicle subtypes distinct from those containing clathrin and AP1. In the distal tubule and in the cortical collecting duct all proteins were found in the apex of the cells; however, AP1 and Clint/EpsinR showed additional staining in perinuclear dots. The occurrence and distribution of the investigated proteins in kidney epithelia are discussed with respect to their possible involvement in the functions of the specific nephron segment.







Similar content being viewed by others
References
Ahle S, Mann A, Eichelsbacher U, Ungewickell E (1988) Structural relationships between clathrin assembly proteins from the Golgi and the plasma membrane. EMBO J 7:919–929
Bouley R, Sun T-X, Chenard M, McLaughlin M, McKee M, Lin HY, Brown D, Ausiello DA (2003) Functional role of the NpxxY motif in internalization of the type 2 vasopressin receptor in LLC-PK1 cells. Am J Physiol Cell Physiol 285:C750–C762
Carnegie JA, McCully ME, Robertson HA (1980) Embedment in glycol methacrylate at low temperature allows immunofluorescent localization of a labile tissue protein. J Histochem Cytochem 28:308–310
Cattoretti G, Becker MHG, Key G, Duchrow M, Schlüter C, Galle J, Gerdes J (1992) Monoclonal antibodies against recombinant parts of the Ki-67 antigen (MIB-1 and MIB-3) detect proliferating cells in microwave-processed formalin-fixed paraffin sections. J Pathol 168:357–363
Christensen EI, Birn H (2001) Megalin and cubilin: synergistic endocytic receptors in renal proximal tubule. Am J Physiol Renal Physiol 280:F562–F573
Fölsch H, Pypaert M, Maday S, Pelletier L, Mellman I (2003) The AP-1A and AP-1B clathrin adaptor complexes define biochemically and functionally distinct membrane domains. J Cell Biol 163:351–362
Ford MGJ, Mills IG, Peter BJ, Vallis Y, Praefcke GJK, Evans PR, McMahon HT (2002) Curvature of clathrin-coated pits driven by epsins. Nature 419:361–366
Geller DS (2005) Mineralocorticoid resistance. Clin Endocrinol 62:513–520
Gibson A, Futter CE, Maxwell S, Allchin EH, Shipman M, Kraehenbuhl J-P, Domingo D, Odorizzi G, Trowbridge IS, Hopkins CR (1998) Sorting mechanisms regulating protein traffic in the apical transcytotic pathway of polarized MDCK cells. J Cell Biol 143:81–94
Groos S, Reale E, Luciano L (2001) Re-evaluation of epoxy resin sections for light and electron microscopy immunostaining. J Histochem Cytochem 49:397–406
Hinrichsen L, Harborth J, Andrees L, Weber K, Ungewickell EJ (2003) Effects of clathrin heavy chain- and alpha-adaptin-specific small inhibitory RNAs on endocytic accessory proteins and receptor trafficking in HeLa cells. J Biol Chem 278:45160–45170
Hirst J, Motley A, Harasaki K, Peak Chew SY, Robinson MS (2003) EpsinR: an ENTH domain-containing protein that interacts with AP-1. Mol Biol Cell 14:625–641
Hunziker W, Simmen T, Honing S (1996) Trafficking of lysosomal membrane proteins in polarized kidney cells. Nephrologie 17:347–350
Kalthoff C, Alves J, Urbanke C, Knorr R, Ungewickell E (2002a) Unusual structural organization of the endocytic proteins AP180 and Epsin 1. J Biol Chem 277:8209–8216
Kalthoff C, Groos S, Kohl R, Mahrhold S, Ungewickell EJ (2002b) Clint: a novel clathrin-binding ENTH-domain protein at the Golgi. Mol Cell Biol 13:4060–4073
Karnovsky MJ (1965) A formaldehyde–glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137A–138A
Katsura T, Verbavatz J-M, Farinas J, Ma T, Ausiello DA, Verkman AS, Brown D (1995) Constitutive and regulated membrane expression of aquaporin 1 and aquaporin 2 water channels stably transfected LLC-PK1 epithelial cells. Proc Natl Acad Sci USA 92:7212–7216
Katzmann DJ, Odorizzi G, Emr SD (2002) Receptor downregulation and multivesicular-body sorting. Nat Rev Mol Cell Biol 3:893–905
Kim JM, Wu H, Green G, Winkler CA, Kopp JB, Miner JH, Unanue ER, Shaw AS (2003) CD2-Associated protein haploinsufficiency is linked to glomerular disease susceptibility. Science 300:1298–1300
King SL, Kozono D, Agre P (2004) From structure to disease: the evolving tale of aquaporin biology. Nat Rev Mol Cell Biol 5:687–698
Le Roy C, Wrana JL (2005) Clathrin- and non-clathrin mediated endocytic regulation of cell signalling. Nat Rev Mol Cell Biol 6:112–126
Madrid R, Le Maout S, Barrault M-B, Janvier K, Benichou S, Mérot J (2001) Polarized trafficking and surface expression of the AQP4 water channel are coordinated by serial and regulated interactions with different clathrin–adaptor complexes. EMBO J 20:7008–7021
Maunsbach AB (1976) Cellular mechanisms of tubular protein transport. In: Thurau K (ed) International review of physiology. Kidney and urinary tract physiology II, vol 11. University Park Press, Baltimore, pp 145–167
Merighi A (1992) Post-embedding electron microscopic immunocytochemistry. In: Polak JM, Priestley JV (eds) Electron microscopic immunocytochemistry—principles and practice. Oxford University Press, Oxford, pp 51–78
Meyerholz A, Hinrichsen L, Groos S, Esk P-C, Brandes G, Ungewickell EJ (2005) Effect of CALM-depletion on clathrin coat formation. Traffic 6:1225–1234
Mundel P, Kriz W (1995) Structure and function of podocytes: an update. Anat Embryol 192:385–397
Obermüller N, Kränzlin B, Blum WF, Gretz N, Witzgall R (2001) An endocytosis defect as a possible cause of proteinuria in polycystic kidney disease. Am J Physiol Renal Physiol 280:F244–F253
Owen DJ, Collins BM, Evans PR (2004) Adaptors for clathrin coats: structure and function. Annu Rev Cell Dev Biol 20:153–191
Rodman JS, Kerjaschki D, Merisko E, Farquhar MG (1984) Presence of an extensive clathrin coat on the apical plasmalemma of the rat kidney proximal tubule cell. J Cell Biol 98:1630–1636
Saint-Pol A, Yélamos B, Amessou M, Mills IG, Dugast M, Tenza D, Schu P, Antony C, McMahon HT, Lamaze C, Johannes L (2004) Clathrin adaptor epsinR is required for retrograde sorting on early endosomal membranes. Dev Cell 6:525–538
Shimkets RA, Lifton RP, Canessa CM (1997) The activity of the epithelial sodium channel is regulated by clathrin-mediated endocytosis. J Biol Chem 41:25537–25541
Traub LM (2005) Common principles in clathrin-mediated sorting at the Golgi and the plasma membrane. Biochim Biophys Acta 1744:415–437
Ungewickell A, Ward ME, Ungewickell E, Majerus PW (2004) The inositol polyphosphate 5-phosphatase OCRL associates with endosomes that are partially coated with clathrin. Proc Natl Acad Sci USA 101:13501–13506
Acknowledgements
We are grateful to Prof. M.S. Robinson who generously supplied the anti-Clint/EpsinR antibody. We would like to thank Ms. S. Fryk for linguistic correction of the manuscript and Ms. A. Hundt for photographic work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Preliminary results were presented at the 28th Annual Meeting of the German Society for Cell Biology, March 16th–19th 2005, Heidelberg, Germany.
Rights and permissions
About this article
Cite this article
Hasse, S., Ungewickell, E.J. & Groos, S. Clathrin and clathrin-accessory proteins in rat kidney cortex epithelia. Histochem Cell Biol 126, 219–229 (2006). https://doi.org/10.1007/s00418-006-0169-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-006-0169-2