Abstract
Purpose
To report the clinical and fluorescein angiographic (FA) features of demyelinating plaque-associated uveitis (DPU), a subset of uveitis in which patients have demyelinating plaques on the brain/cervical magnetic resonance image (MRI) but do not meet the criteria for multiple sclerosis (MS).
Methods
In this retrospective observational study, Persian Patients were diagnosed with DPU and included if (1) they never satisfied the MS criteria, (2) all other possible etiologies were excluded, and (3) they were followed for at least 2 years.
Results
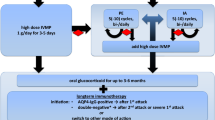
After a median follow-up of 3 years (interquartile range, 2.0–5.3), 8 out of 40 (20%) patients diagnosed with DPU were excluded as they subsequently met the MS criteria. Of remaining 32 patients studied, the mean age was 36.3±9.9 (range 20–56 years), and 30 (93.8%) were female. Twenty-four (75.0%) showed bilateral involvement and 27 (84.4%) had insidious-chronic course. Uveitis was classified as intermediate (with or without anterior uveitis) in 29 (90.6%) and isolated anterior in 3 (9.4%) patients. Nine (28.1%) patients had at least one systemic neurological complaint. Ocular findings were: granulomatous keratic precipitates in 43/44 (97.7%) eyes; snowballs in 25/52 (48.1%) eyes; snowbanks in 4/52 (7.7%) eyes; cystoid macular edema in 20/56 (35.7%) eyes; and optic neuritis in 5/56 (8.9%) eyes. Visual acuity was ≥ 20/40 in 39 eyes (69.6%) at presentation which improved to 46 eyes (81.2%) at 2-year follow up. The two most frequent findings in FA were optic disc leakage/staining in 44/52 (81.5%) eyes, and peripheral retinal perivascular leakage in 39/52 (76.9%) eyes, which in 14/52 (26.9%) eyes extended beyond the equator.
Conclusion
DPU usually presents as a bilateral chronic granulomatous intermediate and, less often, isolated anterior uveitis, especially in females. Most are neurologically asymptomatic. Visual outcome is generally favorable. In FA, peripheral retinal perivascular leakage is common. DPU patients have an increased tendency to develop MS and should be prohibited from anti-TNF treatment.



Similar content being viewed by others
References
Casselman P, Cassiman C, Casteels I et al (2021) Insights into multiple sclerosis-associated uveitis: a scoping review. Acta ophthalmologica 99(6):592–603. https://doi.org/10.1111/aos.14697
Messenger W, Hildebrandt L, Mackensen F et al (2015) Characterisation of uveitis in association with multiple sclerosis. Br J Ophthalmol 99(2):205–209. https://doi.org/10.1136/bjophthalmol-2014-305518
Bosch X, Saiz A, Ramos-Casals M, BIOGEAS Study Group (2011) Monoclonal antibody therapy-associated neurological disorders. Nature reviews. Neurology 7(3):165–172. https://doi.org/10.1038/nrneurol.2011.1
Mohan N, Edwards ET, Cupps TR et al (2001) Demyelination occurring during anti-tumor necrosis factor alpha therapy for inflammatory arthritides. Arthritis Rheum 44(12):2862–2869. https://doi.org/10.1002/1529-0131(200112)44:12%3c2862::aid-art474%3e3.0.co;2-w
Kemanetzoglou E, Andreadou E (2017) CNS Demyelination with TNF-α blockers. Curr Neurol Neurosc Rep 17(4):36. https://doi.org/10.1007/s11910-017-0742-1
Papadia M, Herbort CP (2010) Infliximab-induced demyelination causes visual disturbance mistaken for recurrence of HLA-B27-related uveitis. Ocul Immunol Inflamm 18(6):482–484. https://doi.org/10.3109/09273948.2010.496914
AlBloushi AF, Dheyab AM, Al-Swaina NF et al (2021) Clinical findings and outcomes of uveitis associated with multiple sclerosis. Europ J Ophthalmol 31(2):482–490. https://doi.org/10.1177/1120672120904667
Petrushkin H, Kidd D, Pavesio C (2015) Intermediate uveitis and multiple sclerosis: to scan or not to scan. Br J Ophthalmol 99(12):1591–1593. https://doi.org/10.1136/bjophthalmol-2015-307269
Polman CH, Reingold SC, Banwell B et al (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69(2):292–302. https://doi.org/10.1002/ana.22366
Thompson AJ, Banwell BL, Barkhof F et al (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17(2):162–173. https://doi.org/10.1016/S1474-4422(17)30470-2
Fazekas F, Offenbacher H, Fuchs S et al (1988) Criteria for an increased specificity of MRI interpretation in elderly subjects with suspected multiple sclerosis. Neurology 38(12):1822–1825. https://doi.org/10.1212/wnl.38.12.1822
Barkhof F, Filippi M, Miller DH et al (1997) Comparison of MRI criteria at first presentation to predict conversion to clinically definite multiple sclerosis. Brain: J Neurol 120(Pt 11):2059–2069. https://doi.org/10.1093/brain/120.11.2059
DA, Jabs, Nussenblatt RB, Rosenbaum JT, Standardization of Uveitis Nomenclature (SUN) Working Group (2005) Standardization of uveitis nomenclature for reporting clinical data. Results of the First International Workshop. Amer J Ophthalmol 140(3):509–516. https://doi.org/10.1016/j.ajo.2005.03.057
Abu El-Asrar AM, Herbort CP, Tabbara KF (2005) Retinal vasculitis. Ocul Immunol Inflamm 13(6):415–433. https://doi.org/10.1080/09273940591003828
Kerrison JB, Flynn T, Green WR (1994) Retinal pathologic changes in multiple sclerosis. Retina 14(5):445–451. https://doi.org/10.1097/00006982-199414050-00010
Prineas JW, Wright RG (1978) Macrophages, lymphocytes, and plasma cells in the perivascular compartment in chronic multiple sclerosis. Lab Investig J Tech Methods Pathol 38(4):409–421
Ciccarelli O, Barkhof F, Bodini B et al (2014) Pathogenesis of multiple sclerosis: insights from molecular and metabolic imaging. Lancet Neurol 13(8):807–822. https://doi.org/10.1016/S1474-4422(14)70101-2
Przeździecka-Dołyk J, Węgrzyn A, Turno-Kręcicka A, Misiuk-Hojło M (2016) Immunopathogenic Background of Pars Planitis. Arch Immunol Ther Exp 64(2):127–137. https://doi.org/10.1007/s00005-015-0361-y
Hochmeister S, Grundtner R, Bauer J et al (2006) Dysferlin is a new marker for leaky brain blood vessels in multiple sclerosis. J Neuropathol Exp Neurol 65(9):855–865. https://doi.org/10.1097/01.jnen.0000235119.52311.16
Kirk J, Plumb J, Mirakhur M, McQuaid S (2003) Tight junctional abnormality in multiple sclerosis white matter affects all calibres of vessel and is associated with blood-brain barrier leakage and active demyelination. J Pathol 201(2):319–327. https://doi.org/10.1002/path.1434
Malinowski SM, Pulido JS, Goeken NE, Brown CK, Folk JC (1993) The association of HLA-B8, B51, DR2, and multiple sclerosis in pars planitis. Ophthalmology 100(8):1199–1205. https://doi.org/10.1016/s0161-6420(93)31505-8
Olsson T, Barcellos LF, Alfredsson L (2017) Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nat Rev Neurol 13(1):25–36. https://doi.org/10.1038/nrneurol.2016.187
Malinowski SM, Pulido JS, Folk JC (1993) Long-term visual outcome and complications associated with pars planitis. Ophthalmology 100(6):818–825. https://doi.org/10.1016/s0161-6420(93)31567-8
Jakob E, Reuland MS, Mackensen F et al (2009) Uveitis subtypes in a german interdisciplinary uveitis center–analysis of 1916 patients. J Rheumatol 36(1):127–136. https://doi.org/10.3899/jrheum.080102
Jamshidi A, Gharibdoost F, Vojdanian M et al (2017) A phase III, randomized, two-armed, double-blind, parallel, active controlled, and non-inferiority clinical trial to compare efficacy and safety of biosimilar adalimumab (CinnoRA®) to the reference product (Humira®) in patients with active rheumatoid arthritis. Arthritis Res Ther 19(1):168. https://doi.org/10.1186/s13075-017-1371-4
Soheilian M, Ebrahimiadib N, Hedayatfar A et al (2022) Efficacy of biosimilar adalimumab in the treatment of Behçet’s uveitis. Ocul Immunol Inflamm 30(6):1495–1500. https://doi.org/10.1080/09273948.2021.1900276
Zein G, Berta A, Foster CS (2004) Multiple sclerosis-associated uveitis. Ocul Immunol Inflamm 12(2):137–142. https://doi.org/10.1080/09273940490895344
Chirpaz NM, Kerever SMP, Gavoille AM et al (2022) Relevance of brain MRI in patients with uveitis: retrospective cohort on 402 patients. Ocul Immunol Inflamm 30(5):1109–1115. https://doi.org/10.1080/09273948.2020.1870145
Azami M, YektaKooshali MH, Shohani M et al (2019) Epidemiology of multiple sclerosis in Iran: a systematic review and meta-analysis. PloS One 14(4):e0214738. https://doi.org/10.1371/journal.pone.0214738
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Ethics approval for this study was obtained from the Research Ethics Committees of Iran University of Medical Sciences (Research Ethics Committees Certificate: IR.IUMS.REC.1402.032).
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Research Ethics Committees of Iran University of Medical Sciences and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hedayatfar, A., Anvari, P., Herbort, C.P. et al. Demyelinating plaque-associated uveitis. Graefes Arch Clin Exp Ophthalmol 262, 575–582 (2024). https://doi.org/10.1007/s00417-023-06270-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-023-06270-3