Abstract
Purpose
This study aimed to determine the preferred protocol of corneal collagen cross-linking (CXL) in the treatment of progressive keratoconus.
Methods
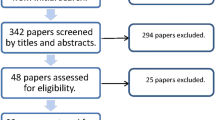
Relevant studies were retrieved in PubMed, EMBASE and Cochrane Central Register of Controlled Trials (CENTRAL). Maximum keratometry value (Kmax), best spectacle-corrected visual acuity (BSCVA), manifest refraction spherical equivalent (MRSE), and endothelial cell density (ECD) were evaluated in network meta-analysis.
Results
Eight randomized controlled trials (RCTs) were included. Low-level evidence suggested that aCXL with 30mW/cm2 for 3 min (aCXL-3) might be the best protocol for reducing BSCVA (65.22%) but worst protocol for reducing MRSE (51.53%). aCXL with 18mW/cm2 for 5 min (aCXL-5) might be the best protocol for reducing Kmax (39.58%) and MRSE (77.85%) but might be the worst for preserving ECD (50.98%). aCXL with 9mW/cm2 for 10 min (aCXL-10) might be the best protocol for preserving ECD (31.53%).
Conclusion
Overall, three protocols of aCXL are comparable in therapeutic efficacy and safety for treating progressive keratoconus. Despite no direct data comparing the efficacy of each technique according to different patients' profiles, it is reasonable to state that aCXL-5 may be the best for patients at early-stage to reduce Kmax and MRSE, aCXL-3 may be the best for patients at mid-stage to improve BSCVA, and aCXL-10 may be the best for patients at late-stage to preserve DEC.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article/as supplementary information files.
References
Rabinowitz YS (1998) Keratoconus. Surv Ophthalmol 42:297–319
Hashemi H, Mohebbi M, Asgari S (2020) Standard and accelerated corneal cross-linking long-term results: A randomized clinical trial. Eur J Ophthalmol 30:650–657. https://doi.org/10.1177/1120672119839927
Wollensak G, Spoerl E, Seiler T (2003) Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol 135:620–627. https://doi.org/10.1016/s0002-9394(02)02220-1
Cantemir A, Alexa AI, Galan BG et al (2017) Iontophoretic collagen cross-linking versus epithelium-off collagen cross-linking for early stage of progressive keratoconus – 3years follow-up study. Acta Ophthalmol 95:e649–e655
Wittig-Silva C, Franzco EC, Islam FMA et al (2014) A Randomized, Controlled Trial of Corneal Collagen Cross-Linking in Progressive Keratoconus. Ophthalmology 121:812–821
Hashemi H, Seyedian MA, Miraftab M et al (2013) Corneal collagen cross-linking with riboflavin and ultraviolet a irradiation for keratoconus: long-term results. Ophthalmology 120:1515–1520
O’Brart D, Kwong T, Patel P et al (2013) Long-term follow-up of riboflavin/ultraviolet A (370 nm) corneal collagen cross-linking to halt the progression of keratoconus. Br J Ophthalmol 97:433–437. https://doi.org/10.1136/bjophthalmol-2012-302556
Wen D, Li Q, Song B et al (2018) Comparison of Standard Versus Accelerated Corneal Collagen Cross-Linking for Keratoconus: A Meta-Analysis. Invest Ophthalmol Vis Sci 59:3920–3931. https://doi.org/10.1167/iovs.18-24656
Waszczykowska A, Jurowski P (2015) Two-year accelerated corneal cross-linking outcome in patients with progressive keratoconus. BioMed Res Int 2015:325157. https://doi.org/10.1155/2015/325157
Kymionis GD, Tsoulnaras KI, Liakopoulos DA et al (2016) Corneal Stromal Demarcation Line Depth Following Standard and a Modified High Intensity Corneal Cross-linking Protocol. J Refract Surg 32:218–222
Razmjoo H, Peyman A, Rahimi A et al (2017) Cornea Collagen Cross-linking for Keratoconus: A Comparison between Accelerated and Conventional Methods. Adv Biomed Res 6:10. https://doi.org/10.4103/2277-9175.200785
Kobashi H, Tsubota K (2020) Accelerated Versus Standard Corneal Cross-Linking for Progressive Keratoconus: A Meta-Analysis of Randomized Controlled Trials. Cornea 39:172–180. https://doi.org/10.1097/ico.0000000000002092
Shajari M, Kolb CM, Agha B et al (2019) Comparison of standard and accelerated corneal cross-linking for the treatment of keratoconus: a meta-analysis. Acta Ophthalmol 97:e22–e35. https://doi.org/10.1111/aos.13814
Schindl A, Rosado-Schlosser B, Trautinger F (2001) Reciprocity regulation in photobiology. An overview. Hautarzt 52:779–785. https://doi.org/10.1007/s001050170065
Lang PZ, Hafezi NL, Khandelwal SS et al (2019) Comparative Functional Outcomes After Corneal Crosslinking Using Standard, Accelerated, and Accelerated With Higher Total Fluence Protocols. Cornea 38:433–441. https://doi.org/10.1097/ico.0000000000001878
Shetty R, Pahuja NK, Nuijts RM et al (2015) Current Protocols of Corneal Collagen Cross-Linking: Visual, Refractive, and Tomographic Outcomes. Am J Ophthalmol 160:243–249. https://doi.org/10.1016/j.ajo.2015.05.019
Hoaglin DC, Hawkins N, Jansen JP et al (2011) Conducting indirect-treatment-comparison and network-meta-analysis studies: report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: part 2. Value Health J Int Soc Pharmacoecon Outcomes Res 14:429–437
Page MJ, Moher D, Bossuyt PM et al (2021) PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ (Clin Res Ed) 372:n160. https://doi.org/10.1136/bmj.n160
Hutton B, Salanti G, Caldwell DM et al (2015) The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 162:777–784. https://doi.org/10.7326/m14-2385
Jansen JP, Fleurence R, Devine B et al (2011) Interpreting indirect treatment comparisons and network meta-analysis for health-care decision making: report of the ISPOR Task Force on Indirect Treatment Comparisons Good Research Practices: part 1. Value in Health 14:417–428
Higgins JPT, Altman DG, Gøtzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ (Clin Res Ed) 343:d5928–d5928. https://doi.org/10.1136/bmj.d5928
Cipriani A, Higgins JPT, Geddes JR et al (2013) Conceptual and technical challenges in network meta-analysis. Ann Intern Med 159:130–137. https://doi.org/10.7326/0003-4819-159-2-201307160-00008
Brooks S, Gelman A (1998) General Methods for Monitoring Convergence of Iterative Simulations. J Comput Graphi Stat 7:434–455. https://doi.org/10.1080/10618600.1998.10474787
Burger DA, Schall R (2015) A Bayesian Nonlinear Mixed-Effects Regression Model for the Characterization of Early Bactericidal Activity of Tuberculosis Drugs. J Biopharm Stat 25:1247–1271. https://doi.org/10.1080/10543406.2014.971170
Singh S, Murad MH, Chandar AK et al (2015) Comparative Effectiveness of Pharmacological Interventions for Severe Alcoholic Hepatitis: A Systematic Review and Network Meta-analysis. Gastroenterology 149:958-970.e912. https://doi.org/10.1053/j.gastro.2015.06.006
Bowden J, Tierney JF, Copas AJ et al (2011) Quantifying, displaying and accounting for heterogeneity in the meta-analysis of RCTs using standard and generalised Q statistics. BMC Med Res Methodol 11:41. https://doi.org/10.1186/1471-2288-11-41
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Dias S, Welton NJ, Caldwell DM et al (2010) Checking consistency in mixed treatment comparison meta-analysis. Stat Med 29:932–944
Palma Perez S, Delgado Rodriguez M (2006) Practical considerations on detection of publication bias. Gac Sanit 20(Suppl 3):10–16. https://doi.org/10.1157/13101085
Hagem AM, Thorsrud A, Sandvik GF et al (2019) Randomized Study of Collagen Cross-Linking With Conventional Versus Accelerated UVA Irradiation Using Riboflavin With Hydroxypropyl Methylcellulose: Two-Year Results. Cornea 38:203–209. https://doi.org/10.1097/ico.0000000000001791
Hashemi H, Miraftab M, Seyedian MA et al (2015) Long-term Results of an Accelerated Corneal Cross-linking Protocol (18 mW/cm2) for the Treatment of Progressive Keratoconus. Am J Ophthalmol 160:1164-1170.e1161. https://doi.org/10.1016/j.ajo.2015.08.027
Hashemian H, Jabbarvand M, Khodaparast M et al (2014) Evaluation of corneal changes after conventional versus accelerated corneal cross-linking: a randomized controlled trial. J Refract Surg 30:837–842. https://doi.org/10.3928/1081597x-20141117-02
Sadoughi MM, Einollahi B, Baradaran-Rafii A et al (2018) Accelerated versus conventional corneal collagen cross-linking in patients with keratoconus: an intrapatient comparative study. Int Ophthalmol 38:67–74. https://doi.org/10.1007/s10792-016-0423-0
Sherif AM (2014) Accelerated versus conventional corneal collagen cross-linking in the treatment of mild keratoconus: a comparative study. Clin Ophthalmol 8:1435–1440. https://doi.org/10.2147/opth.S59840
Uçakhan Ö, Yeşiltaş YS (2020) Comparative 2-year outcomes of conventional and accelerated corneal collagen crosslinking in progressive keratoconus. Int J Ophthalmol 13:1223–1230. https://doi.org/10.18240/ijo.2020.08.07
Medeiros C, Giacomin N, Bueno R et al (2016) Accelerated corneal collagen crosslinking: Technique, efficacy, safety, and applications. J Cataract Refract Surg 42:1826–1835. https://doi.org/10.1016/j.jcrs.2016.11.028
Wang Z, Carter RE (2018) Ranking of the most effective treatments for cardiovascular disease using SUCRA: Is it as sweet as it appears? Eur J Prev Cardiol 25:842–843. https://doi.org/10.1177/2047487318767199
Spoerl E, Mrochen M, Sliney D et al (2007) Safety of UVA-riboflavin cross-linking of the cornea. Cornea 26:385–389. https://doi.org/10.1097/ICO.0b013e3180334f78
Acknowledgements
We would like to deeply appreciate all authors who performed all eligible studies which have been included in the present network meta-analysis.
Funding
This study was funded by the National Natural Science Foundation of China (Grant No. 81770955); Project of Shanghai Science and Technology (Grant No.17411950200). The sponsors and funding organizations had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Contributions
Substantially contributed to conception or design: Ling Sun, Xingtao Zhou. Contributed to acquisition, analysis, or interpretation of data: Lan Ding. Drafted the manuscript for important content: Ling Sun. Critically revised the manuscript for important intellectual content: Xingtao Zhou. Gave final approval: All authors.
Corresponding author
Ethics declarations
Consent to participate
NA.
Consent to publish
NA.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors. This study is a meta-analysis and does not involve ethical approval.
Conflicts of interest
No Conflicts of Interest:
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Informed consent
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary file 1: Table S1.
Search strategy of database.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ding, L., Sun, L. & Zhou, X. Network meta-analysis comparing efficacy and safety of different protocols of corneal cross-linking for the treatment of progressive keratoconus. Graefes Arch Clin Exp Ophthalmol 261, 2743–2753 (2023). https://doi.org/10.1007/s00417-023-06026-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-023-06026-z