Abstract
Purpose
To quantitatively analyze human corneal stromal dehydration and estimate proper corneal stromal exposure time during corneal refractive surgery.
Methods
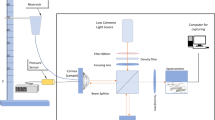
The central thickness changes over time in 34 pieces of human corneal tissue were measured using a white light interferometer. The corneal stromal tissue was obtained by femtosecond laser small incision lenticule extraction. The thickness-time dehydration fitting curves were drawn, and the determination coefficient R2 was calculated. The differences in the fitting curve equation coefficients were compared between the thin and thick lenticule groups. The optimal stromal exposure time was calculated under various conditions, including different optical zones and allowable refractive errors.
Results
A water loss variation model was successfully established. Linear and quadratic fitting curves were drawn, and the determination coefficient R2 values were significantly close to 1. The average values of R2 for quadratic curves and linear phases 1, 2, and 3 were 0.998 ± 0.002, 0.995 ± 0.007, 0.996 ± 0.003, and 0.984 ± 0.035, respectively. The optimal stromal exposure time varied under different optical zones and allowable diopter error conditions. Taking the allowable error of 0.50 D and the optical zone size of 6.5 mm as an example, the optimal time was approximately 24 s.
Conclusions
The dehydration rate of the human corneal stroma is nonlinear, and the quadratic stromal thickness–time dehydration fitting curve is more in line with the actual water loss trend. The length of the stroma exposure time may affect the postoperative refractive accuracy after corneal refractive surgery.








Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Marino GK, Santhiago MR, Wilson SE (2017) Femtosecond lasers and corneal surgical procedures. Asia Pac J Ophthalmol (Phila) 6:456–464. https://doi.org/10.22608/APO.2017163
Seider MI, Mcleod SD, Porco TC et al (2013) The effect of procedure room temperature and humidity on LASIK outcomes. Ophthalmology 120:2204–2208. https://doi.org/10.1016/j.ophtha.2013.04.015
Feltham MH (2002) The effect of water content on the 193 nm excimer laser ablation. Clin Exp Ophthalmol 30:99–103. https://doi.org/10.1046/j.1442-6404.2002.00496.x
Taylor ZD, Garritano J, Sung S et al (2015) THz and mm-wave sensing of corneal tissue water content: electromagnetic modeling and analysis. IEEE Trans Terahertz Sci Technol 5:170–183. https://doi.org/10.1109/TTHZ.2015.2392619
Rosa AM, Murta JN, Quadrado MJ et al (2009) Femtosecond laser versus mechanical microkeratomes for flap creation in laser in situ keratomileusis and effect of postoperative measurement interval on estimated femtosecond flap thickness. J Cataract Refract Surg 35:833–838. https://doi.org/10.1016/j.jcrs.2008.12.038
Aurich H, Wirbelauer C, Jaroszewski J et al (2006) Continuous measurement of corneal dehydration with online optical coherence pachymetry. Cornea 25:182–184. https://doi.org/10.1097/01.ico.0000176610.50461.77
Wirbelauer C, Pham DT (2004) Continuous monitoring of corneal thickness changes during LASIK with online optical coherence pachymetry. J Cataract Refract Surg 30:2559–2568. https://doi.org/10.1016/j.jcrs.2004.04.068
Wirbelauer C, Aurich H, Pham DT (2007) Online optical coherence pachymetry to evaluate intraoperative ablation parameters in LASIK. Graefes Arch Clin Exp Ophthalmol 245:775–781. https://doi.org/10.1007/s00417-006-0447-7
Qi P, Sun L, Ma J et al (2019) Ex vivo quantitative analysis of human corneal stroma dehydration by near-infrared absorption spectroscopy. J Biophotonics 12:e201800472. https://doi.org/10.1002/jbio.201800472
Liu YC, Williams GP, George BL et al (2017) Corneal lenticule storage before reimplantation. Mol Vis 23:753–764
Wolffsohn JS, Flitcroft DI, Gifford KL et al (2019) IMI - myopia control reports overview and introduction. Invest Ophthalmol Vis Sci 60:M1–M19. https://doi.org/10.1167/iovs.18-25980
Wen D, Mcalinden C, Flitcroft I et al (2017) Postoperative efficacy, predictability, safety, and visual quality of laser corneal refractive surgery: a network meta-analysis. Am J Ophthalmol 178:65–78. https://doi.org/10.1016/j.ajo.2017.03.013
Biscevic A, Ahmedbegovic-Pjano M, Pandurevic B et al (2020) Vector analysis of visual acuity and refractive outcomes of astigmatic corrections after T-PRK and Fs-LASIK. Acta Inform Med 28:180–184. https://doi.org/10.5455/aim.2020.28.180-184
Durairaj VD, Balentine J, Kouyoumdjian G et al (2000) The predictability of corneal flap thickness and tissue laser ablation in laser in situ keratomileusis. Ophthalmology 107:2140–2143. https://doi.org/10.1016/s0161-6420(00)00407-3
Kim WS, Jo JM (2001) Corneal hydration affects ablation during laser in situ keratomileusis surgery. Cornea 20:394–397. https://doi.org/10.1097/00003226-200105000-00011
Böhnke M, Chavanne P, Gianotti R et al (1998) Continuous non-contact corneal pachymetry with a high speed reflectometer. J Refract Surg 14:140–146
Subasinghe SK, Ogbuehi KC, Mitchell L et al (2021) Animal model with structural similarity to human corneal collagen fibrillar arrangement. Anat Sci Int 96:286–293. https://doi.org/10.1007/s12565-020-00590-8
Bourassa S, Benjamin WJ, Boltz RL (1991) Effect of humidity on the deswelling function of the human cornea. Curr Eye Res 10:493–500. https://doi.org/10.3109/02713689109001757
Wu Y, Wang Y, Zhang Z et al (2021) Quantitative analysis of human corneal lenticule surface microstructure irregularity with 3D optical profiler using white light interferometry. Curr Eye Res 46:461–469. https://doi.org/10.1080/02713683.2020.1809000
Xu L, Wang Y, Li J et al (2015) Comparison of forward light scatter changes between SMILE, femtosecond laser-assisted LASIK, and epipolis LASIK: results of a 1-year prospective study. J Refract Surg 31:752–758. https://doi.org/10.3928/1081597X-20151021-04
Gritz DC (2011) LASIK interface keratitis: epidemiology, diagnosis and care. Curr Opin Ophthalmol 22:251–255. https://doi.org/10.1097/ICU.0b013e3283477b52
Uysal BS, Duru N, Ozen U et al (2018) Impact of dehydration and fasting on intraocular pressure and corneal biomechanics measured by the ocular response analyzer. Int Ophthalmol 38:451–457. https://doi.org/10.1007/s10792-017-0479-5
Funding
This study was supported by the National Natural Science Foundation of China (No.81873684), Tianjin Health science and Technology Project (No. QN20007), and Science and Technology Foundation of Tianjin Eye Hospital (No. YKQN1919). The funding organization had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee (the Ethics Committee of Tianjin Eye Hospital (No. 201922)) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Patients signed informed consent regarding publishing their data and photographs.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, Y., Wang, Y., Zhang, Z. et al. Corneal stromal dehydration and optimal stromal exposure time during corneal refractive surgery measured using a three-dimensional optical profiler. Graefes Arch Clin Exp Ophthalmol 260, 4005–4013 (2022). https://doi.org/10.1007/s00417-022-05764-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-022-05764-w