Abstract
Purpose
To identify factors associated with diabetic macular edema (DME) and to characterize the types of DME present in eyes with proliferative diabetic retinopathy (PDR).
Methods
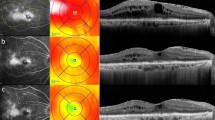
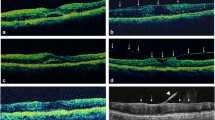
Observational, retrospective case series of PDR patients reviewed for demographic information, general medical history, ophthalmologic history, optical coherence tomography (OCT), and fluorescein angiogram image characteristics. DME and vitreomacular interface (VMI) status were determined using OCT images. DME was defined as center-involving DME (CI-DME) and noncenter-involving DME (NCI-DME). VMI was defined as vitreomacular adhesion (VMA), vitreomacular traction (VMT), or macular posterior vitreous detachment (PVD).
Results
A total of 293 eyes of 210 screened patients with PDR were included. Of the eyes, 194/293 (66.2%) had DME, and 99/293 (33.8%) had no DME; in univariable analysis, there were no significant differences in VMI status (p = 0.4) or epiretinal membrane (ERM, p = 0.1) between them. Of 194 eyes with DME, 90/194 (46.4%) had CI-DME, and 104/194 (53.6%) had NCI-DME. In univariable analysis, CI-DME eyes were significantly more likely than NCI-DME eyes to have a PVD (p = 0.029) and ERM (p < 0.001). In multivariable analysis, the presence of younger age (p = 0.028) and presence of ERM (p = 0.001) were significantly more likely to be observed in eyes with CI-DME.
Conclusion
In this exploratory study focused on diabetic patients with PDR, we determined that VMI status did not have a significant association with DME in general, but VMI status, younger age, and presence of ERM may be associated with CI-DME.


Similar content being viewed by others
Data availability
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
The code during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Authors No (1991) Early treatment diabetic retinopathy study design and baseline patient characteristics. ETDRS report number 7. Ophthalmology 98:741–756. https://doi.org/10.1016/s0161-6420(13)38009-9
Baharivand N, Zarghami N, Panahi F, Dokht Ghafari MY, Mahdavi Fard A, Mohajeri A (2012) Relationship between vitreous and serum vascular endothelial growth factor levels, control of diabetes and microalbuminuria in proliferative diabetic retinopathy. Clin Ophthalmol 6:185–191. https://doi.org/10.2147/OPTH.S27423
Funatsu H, Yamashita H, Nakamura S, Mimura T, Eguchi S, Noma H, Hori S (2006) Vitreous levels of pigment epithelium-derived factor and vascular endothelial growth factor are related to diabetic macular edema. Ophthalmology 113:294–301. https://doi.org/10.1016/j.ophtha.2005.10.030
Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, Pasquale LR, Thieme H, Iwamoto MA, Park JE (1994) Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med 331:1480–1487. https://doi.org/10.1056/NEJM199412013312203
Funatsu H, Noma H, Mimura T, Eguchi S, Hori S (2009) Association of vitreous inflammatory factors with diabetic macular edema. Ophthalmology 116:73–79. https://doi.org/10.1016/j.ophtha.2008.09.037
Nguyen QD, Brown DM, Marcus DM, Boyer DS, Patel S, Feiner L, Gibson A, Sy J, Rundle AC, Hopkins JJ, Rubio RG, Ehrlich JS, Group RaRR (2012) Ranibizumab for diabetic macular edema: results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology 119:789–801. https://doi.org/10.1016/j.ophtha.2011.12.039
Wells JA, Glassman AR, Ayala AR, Jampol LM, Bressler NM, Bressler SB, Brucker AJ, Ferris FL, Hampton GR, Jhaveri C, Melia M, Beck RW, Network DRCR (2016) Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema: two-year results from a comparative effectiveness randomized clinical trial. Ophthalmology 123:1351–1359. https://doi.org/10.1016/j.ophtha.2016.02.022
Heier JS, Korobelnik JF, Brown DM, Schmidt-Erfurth U, Do DV, Midena E, Boyer DS, Terasaki H, Kaiser PK, Marcus DM, Nguyen QD, Jaffe GJ, Slakter JS, Simader C, Soo Y, Schmelter T, Vitti R, Berliner AJ, Zeitz O, Metzig C, Holz FG (2016) Intravitreal aflibercept for diabetic macular edema: 148-week results from the VISTA and VIVID studies. Ophthalmology 123:2376–2385. https://doi.org/10.1016/j.ophtha.2016.07.032
Jorge R, Costa RA, Calucci D, Cintra LP, Scott IU (2006) Intravitreal bevacizumab (Avastin) for persistent new vessels in diabetic retinopathy (IBEPE study). Retina 26:1006–1013. https://doi.org/10.1097/01.iae.0000246884.76018.63
Acan D, Calan M, Er D, Arkan T, Kocak N, Bayraktar F, Kaynak S (2018) The prevalence and systemic risk factors of diabetic macular edema: a cross-sectional study from Turkey. BMC Ophthalmol 18:91. https://doi.org/10.1186/s12886-018-0753-y
Ophir A, Martinez MR, Mosqueda P, Trevino A (2010) Vitreous traction and epiretinal membranes in diabetic macular oedema using spectral-domain optical coherence tomography. Eye (Lond) 24:1545–1553. https://doi.org/10.1038/eye.2010.80
Gaucher D, Tadayoni R, Erginay A, Haouchine B, Gaudric A, Massin P (2005) Optical coherence tomography assessment of the vitreoretinal relationship in diabetic macular edema. Am J Ophthalmol 139:807–813. https://doi.org/10.1016/j.ajo.2004.12.084
Kim BY, Smith SD, Kaiser PK (2006) Optical coherence tomographic patterns of diabetic macular edema. Am J Ophthalmol 142:405–412. https://doi.org/10.1016/j.ajo.2006.04.023
Panozzo G, Parolini B, Gusson E, Mercanti A, Pinackatt S, Bertoldo G, Pignatto S (2004) Diabetic macular edema: an OCT-based classification. Semin Ophthalmol 19:13–20. https://doi.org/10.1080/08820530490519934
Shah SP, Patel M, Thomas D, Aldington S, Laidlaw DA (2006) Factors predicting outcome of vitrectomy for diabetic macular oedema: results of a prospective study. Br J Ophthalmol 90:33–36. https://doi.org/10.1136/bjo.2005.072934
Haller JA, Qin H, Apte RS, Beck RR, Bressler NM, Browning DJ, Danis RP, Glassman AR, Googe JM, Kollman C, Lauer AK, Peters MA, Stockman ME, Committee DRCRNW (2010) Vitrectomy outcomes in eyes with diabetic macular edema and vitreomacular traction. Ophthalmology 117:1087-1093.e1083. https://doi.org/10.1016/j.ophtha.2009.10.040
Nasrallah FP, Jalkh AE, Van Coppenolle F, Kado M, Trempe CL, McMeel JW, Schepens CL (1988) The role of the vitreous in diabetic macular edema. Ophthalmology 95:1335–1339. https://doi.org/10.1016/s0161-6420(88)33004-6
Hikichi T, Fujio N, Akiba J, Azuma Y, Takahashi M, Yoshida A (1997) Association between the short-term natural history of diabetic macular edema and the vitreomacular relationship in type II diabetes mellitus. Ophthalmology 104:473–478. https://doi.org/10.1016/s0161-6420(97)30289-9
Ying GS, Maguire MG, Glynn R, Rosner B (2018) Tutorial on biostatistics: statistical analysis for correlated binary eye data. Ophthalmic Epidemiol 25:1–12. https://doi.org/10.1080/09286586.2017.1320413
Armstrong RA (2014) When to use the Bonferroni correction. Ophthalmic Physiol Opt 34:502–508. https://doi.org/10.1111/opo.12131
Mangiafico SS (2015) An R Companion for the Handbook of Biological Statistics, version 1.3.2. https://rcompanion.org/rcompanion/. Accessed 19 July 2021
Varma R, Bressler NM, Doan QV, Gleeson M, Danese M, Bower JK, Selvin E, Dolan C, Fine J, Colman S, Turpcu A (2014) Prevalence of and risk factors for diabetic macular edema in the United States. JAMA Ophthalmol 132:1334–1340. https://doi.org/10.1001/jamaophthalmol.2014.2854
Cheung N, Tan SP, Lee SY, Cheung GCM, Tan G, Kumar N, Cheng CY, Wong TY (2017) Prevalence and risk factors for epiretinal membrane: the Singapore Epidemiology of Eye Disease study. Br J Ophthalmol 101:371–376. https://doi.org/10.1136/bjophthalmol-2016-308563
Hagenau F, Vogt D, Ziada J, Guenther SR, Haritoglou C, Wolf A, Priglinger SG, Schumann RG (2019) Vitrectomy for diabetic macular edema: optical coherence tomography criteria and pathology of the vitreomacular interface. Am J Ophthalmol 200:34–46. https://doi.org/10.1016/j.ajo.2018.12.004
Chen YS, Hackett SF, Schoenfeld CL, Vinores MA, Vinores SA, Campochiaro PA (1997) Localisation of vascular endothelial growth factor and its receptors to cells of vascular and avascular epiretinal membranes. Br J Ophthalmol 81:919–926. https://doi.org/10.1136/bjo.81.10.919
Yamamoto T, Akabane N, Takeuchi S (2001) Vitrectomy for diabetic macular edema: the role of posterior vitreous detachment and epimacular membrane. Am J Ophthalmol 132:369–377. https://doi.org/10.1016/s0002-9394(01)01050-9
Ishida S, Shinoda K, Kawashima S, Oguchi Y, Okada Y, Ikeda E (2000) Coexpression of VEGF receptors VEGF-R2 and neuropilin-1 in proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci 41:1649–1656
Ercalik NY, Imamoglu S, Kumral ET, Yenerel NM, Bardak H, Bardak Y (2016) Influence of the epiretinal membrane on ranibizumab therapy outcomes in patients with diabetic macular edema. Arq Bras Oftalmol 79:373–375. https://doi.org/10.5935/0004-2749.20160106
Kulikov AN, Sosnovskii SV, Berezin RD, Maltsev DS, Oskanov DH, Gribanov NA (2017) Vitreoretinal interface abnormalities in diabetic macular edema and effectiveness of anti-VEGF therapy: an optical coherence tomography study. Clin Ophthalmol 11:1995–2002. https://doi.org/10.2147/OPTH.S146019
Yoon D, Rusu I, Barbazetto I (2014) Reduced effect of anti-vascular endothelial growth factor agents on diabetics with vitreomacular interface abnormalities. Int Ophthalmol 34:817–823. https://doi.org/10.1007/s10792-013-9884-6
Flaxel CJ, Edwards AR, Aiello LP, Arrigg PG, Beck RW, Bressler NM, Bressler SB, Ferris FL, Gupta SK, Haller JA, Lazarus HS, Qin H (2010) Factors associated with visual acuity outcomes after vitrectomy for diabetic macular edema: diabetic retinopathy clinical research network. Retina 30:1488–1495. https://doi.org/10.1097/IAE.0b013e3181e7974f
Anderson W, Piggott K, Bao YK, Pham H, Kavali S, Rajagopal R (2019) Complete posterior vitreous detachment reduces the need for treatment of diabetic macular edema. Ophthalmic Surg Lasers Imaging Retina 50:e266–e273. https://doi.org/10.3928/23258160-20191031-13
Ghazi NG, Ciralsky JB, Shah SM, Campochiaro PA, Haller JA (2007) Optical coherence tomography findings in persistent diabetic macular edema: the vitreomacular interface. Am J Ophthalmol 144:747–754. https://doi.org/10.1016/j.ajo.2007.07.012
Funding
Funded in party by a Dean’s Research Scholarship (John R. O’Fee) from the Keck School of Medicine of the University of Southern California (Los Angeles, CA) and an unrestricted grant to the Department of Ophthalmology at USC Keck School of Medicine from Research to Prevent Blindness (New York, NY).
Author information
Authors and Affiliations
Contributions
John O’Fee: conception and design, literature search, data collection, data interpretation, writing, and figures. Joseph Juliano: conception and design, literature search, data analysis, data interpretation, and writing. Andrew A. Moshfeghi: conception and design, literature search, data interpretation, and writing.
Corresponding author
Ethics declarations
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. The study involved human participants and was determined to be exempt from review by the University of Southern California Institutional Review Board (08/20/2020, HS-20–00538).
Consent to participate
The study was retrospective in nature so it would not have been feasible to obtain informed consent from each participant.
Human and animal rights
The research involved human participants and was determined to be exempt from review by the University of Southern California Institutional Review Board; the study was retrospective, so it would not have been feasible to obtain informed consent from each participant.
Conflict of interest
The authors declare no competing interests.
Financial disclosures
John O’Fee and Dr. Juliano do not have financial disclosures to report.
Dr. Moshfeghi’s financial disclosures are as follows (for the last 3 years):
Company | Consultant | Research grant | Equity ownership | Speaker |
|---|---|---|---|---|
Allegro | x (inactive) | |||
Alcon Surgical | x (inactive) | |||
Alimera | x (inactive) | |||
Allergan | x | x (inactive) | ||
Bausch | x (inactive) | |||
Clearside | x (inactive) | |||
Eyepoint | x (inactive) | |||
Genentech | x | x (paid to my institution) | ||
Graybug | x (inactive) | |||
Novartis | x | x (paid to my institution) | ||
Ocular Therapeutix | x | x | ||
OptiSTENT | x | |||
Placid0 | x | x | ||
Pr3vent | x | x | ||
Regeneron | x | x (paid to my institution) | ||
Regenxbio | x | |||
Zeiss | x (inactive) | |||
Note: All are active unless otherwise specified as “inactive” | ||||
Note: All are paid to me unless otherwise specified |
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Meeting presentations
Presented, in part, at the Association of Research in Vision and Ophthalmology Annual Meeting on May 1, 2021, in virtual format. An abstract related to this work was presented at the 54th Annual Scientific Meeting of the Retina Society on September 30, 2021, in Chicago, IL.
Rights and permissions
About this article
Cite this article
O’Fee, J.R., Juliano, J. & Moshfeghi, A.A. Factors associated with diabetic macular edema in patients with proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 260, 2191–2200 (2022). https://doi.org/10.1007/s00417-022-05595-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-022-05595-9