Abstract
Purpose
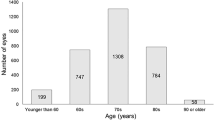
To compare the characteristics and response to treatment between patients with NVAMD presenting at the extremities of the AMD age spectrum.
Methods
Fifty-four eyes of 47 patients were included in this retrospective study, divided by age at NVAMD presentation under 65 (n = 15) or over 85 (n = 39) years. All patients were initially treated with 3 monthly bevacizumab injections, followed by a PRN protocol. Clinical parameters and OCT characteristics were recorded and analyzed at presentation, after the initial 3 monthly injections and at 1 year.
Results
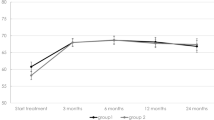
At presentation, patients in the young group had significantly higher rates of subretinal fluid (p = 0.005), a polypoidal choroidal vasculopathy–like pattern (p < 0.01) and a history of smoking (p = 0.004). Submacular hemorrhage and pigment epithelial detachments were more common in young patients, and intraretinal fluid was more common in elderly patients (all with borderline statistical significance). VA improved significantly more in the younger patients at 3 months and 1 year (p = 0.001 and 0.002, respectively), despite similar treatment protocols and mean number of injections. Bilateral involvement at baseline was more common in elderly patients (p = 0.008). The differences in OCT characteristics between groups remained throughout the study period.
Conclusion
There are considerable differences in the clinical manifestations and response to treatment between NVAMD patients at the extremities of the AMD age spectrum. Different pathophysiological, systemic, and genetic factors may play a role in such patients.




Similar content being viewed by others
References
Bressler NM (2004) Age-related macular degeneration is the leading cause of blindness. JAMA 291:1900–1901
Kahn HA, Leibowitz HM, Ganley JP, Kini MM, Colton T, Nickerson RS, Dawber TR The Framingham eye study (1977): I. Outline and major prevalence findings. Am J Epidemiol 106:17e32
Ferris FL, Fine SL, Hyman L (1984) Age-related macular degeneration and blindness due to neovascular maculopathy. Arch Ophthalmol 102:1640e2
Klein R, Klein BE, Linton KL (1992) Prevalence of age-related maculopathy. The Beaver Dam Eye Study. Ophthalmology 99:933–943
Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, Wong TY (2014) Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health 2(2):e106–e116
Li JQ, Welchowski T, Schmid M, Mauschitz MM, Holz FG, Finger RP (2019) Prevalence and incidence of age-related macular degeneration in Europe: a systematic review and meta-analysis. Br J Ophthalmol pii: bjophthalmol-2019-314422
Finger RP, Wickremasinghe SS, Baird PN, Guymer RH (2014) Predictors of anti-VEGF treatment response in neovascular age-related macular degeneration. Surv Ophthalmol 59(1):1–18
Subhi Y, Sørensen TL (2017) Neovascular age-related macular degeneration in the very old (≥90 years): epidemiology, adherence to treatment, and comparison of efficacy. J Ophthalmol 2017:7194927
Kim JH, Chang YS, Kim JW, Kim CG, Lee DW (2019) Age-related differences in the prevalence of subtypes of neovascular age-related macular degeneration in the first diagnosed eye. Graefes Arch Clin Exp Ophthalmol 257(5):891–898
Lechanteur YT, van de Camp PL, Smailhodzic D, van de Ven JP, Buitendijk GH, Klaver CC, Groenewoud JM, den Hollander AI, Hoyng CB, Klevering BJ (2015) Association of smoking and CFH and ARMS2 risk variants with younger age at onset of neovascular age-related macular degeneration. JAMA Ophthalmol 133(5):533–541
Grassmann F, International AMD Genomics Consortium (IAMDGC), Weber HF, Veitia RA (2020) Insights into the loss of the Y chromosome with age in control individuals and in patients with age-related macular degeneration using genotyping microarraydata. Hum Genet 139(3):401–407
Holladay JT (1997) Proper method for calculating average visual acuity. J Refract Cat Surg 13:388–391
Schulze-Bonsel K, Feltgen N, Burau H, Hansen L, Bach M (2006) Visual acuities “hand motion” and “counting fingers” can be quantified with the Freiburger visual acuity test. Invest Ophthalmol Vis Sci 47:1236–1240
Boyer DS, Antoszyk AN, Awh CC, Bhisitkul RB, Shapiro H, Acharya NR, MARINA Study Group (2007) Subgroup analysis of the MARINA study of ranibizumab in neovascular age-related macular degeneration. Ophthalmology 114:246–252
Yamashiro K, Tomita K, Tsujikawa A, Nakata I, Akagi-Kurashige Y, Miyake M, Ooto S, Tamura H, Yoshimura N (2012) Factors associated with the response of age-related macular degeneration to intravitreal ranibizumab treatment. Am J Ophthalmol 154:125–136
Ying GS, Maguire MG, Daniel E, Ferris FL, Jaffe GJ, Grunwald JE, Toth CA, Huang J, Martin DF, Comparison of Age-Related Macular Degeneration Treatments Trials (CATT) Research Group (2015) Association of baseline characteristics and early vision response with 2-year vision outcomes in the Comparison of AMD Treatments Trials (CATT). Ophthalmology 122:2523–2531
Fasler K, Moraes G, Wagner S, Kortuem KU, Chopra R, Faes L, Preston G, Pontikos N, Fu DJ, Patel P, Tufail A, Lee AY, Balaskas K, Keane PA (2019) One- and two-year visual outcomes from the Moorfields age-related macular degeneration database: a retrospective cohort study and an open science resource. BMJ Open 9(6):e027441
Guber J, Josifova T, Henrich PB, Guber I (2014) Clinical risk factors for poor anatomic response to ranibizumab in neovascular age-related macular degeneration. Open Ophthalmol J 8:3–6
Kang S, Roh YJ (2009) One-year results of intravitreal ranibizumab for neovascular age-related macular degeneration and clinical responses of various subgroups. Jpn J Ophthalmol 53:389–395
Smailhodzic D, Muether PS, Chen J, Kwestro A, Zhang AY, Omar A, Van de Ven JP, Keunen JE, Kirchhof B, Hoyng CB, Klevering BJ, Koenekoop RK, Fauser S, den Hollander AI (2012) Cumulative effect of risk alleles in CFH, ARMS2, and VEGFA on the response to ranibizumab treatment in age-related macular degeneration. Ophthalmology 119:2304–2311
Habibi I, Kort F, Sfar I, Chebil A, Bouraoui R, Ben Abdallah T, Gorgi Y, El Matri L (2016) Effect of risk alleles in CFH, C3, and VEGFA on the response to intravitreal bevacizumab in tunisian patients with neovascular age-related macular degeneration. Klin Monbl Augenheilkd 233:465–470
Haga A, Kawaji T, Ideta R, Inomata Y, Tanihara H (2018) Treat-and-extend versus every-other-month regimens with aflibercept in age-related macular degeneration. Acta Ophthalmol 96:e393–e398
Wykoff CC, Croft DE, Brown DM, Wang R, Payne JF, Clark L, Abdelfattah NS, Sadda SR, TREX-AMD Study Group (2015) Prospective trial of treat-and-extend versus monthly dosing for neovascular age-related macular degeneration: TREX-AMD 1-year results. Ophthalmology 122:2514–2522
Okada M, Kandasamy R, Chong EW, McGuiness M, Guymer RH (2018) The treat-and-extend injection regimen versus alternate dosing strategies in age-related macular degeneration: a systematic review and meta-analysis. Am J Ophthalmol 192:184–197
Gupta B, Adewoyin T, Patel SK, Sivaprasad S (2011) Comparison of two intravitreal ranibizumab treatment schedules for neovascular age-related macular degeneration. Br J Ophthalmol 95:386–390
Mekjavic PJ, Kraut A, Urbancic M, Lenassi E, Hawlina M (2011) Efficacy of 12-month treatment of neovascular age-related macular degeneration with intravitreal bevacizumab based on individually determined injection strategies after threeconsecutive monthly injections. Acta Ophthalmol 89:647–653
Arias L, Caminal JM, Casas L, Masuet C, Badia MB, Rubio M, Pujol O, Arruga J (2008) A study comparing two protocols of treatment with intravitreal bevacizumab (Avastin) for neovascular age-related macular degeneration. Br J Ophthalmol 92:1636–1641
Melamud A, Stinnett S, Fekrat S (2008) Treatment of neovascular age-related macular degeneration with intravitreal bevacizumab: efficacy of three consecutive monthly injections. Am J Ophthalmol 146:91–95
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Meir Medical Center and Clalit Healthcare Organization and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
This is a retrospective study that did not require informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rubowitz, A., Esa, S., Fradkin, M. et al. Neovascular age-related macular degeneration presenting at extremities of age: a comparative study. Graefes Arch Clin Exp Ophthalmol 258, 2399–2405 (2020). https://doi.org/10.1007/s00417-020-04893-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04893-4