Abstract
Purpose
To investigate the effect of image averaging on qualitative and quantitative assessments of optical coherence tomography angiography (OCTA) images from eyes of patients with branch retinal vein occlusion (BRVO).
Methods
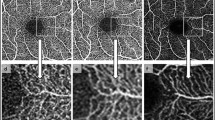
Macular OCTA images of 33 eyes of 33 patients with BRVO were obtained using the HS100 HR-SD-OCT system (Canon, Inc.). For each eye, five OCTA cube scans were obtained with a 3 × 3 mm scan protocol, and the data were averaged and compounded into a single high image quality cube data using built-in software. Pre- and post-averaging images were compared qualitatively and quantitatively in superficial capillary plexus (SCP) and deep capillary plexus (DCP) OCTA image slabs.
Results
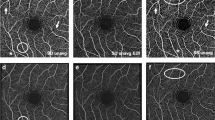
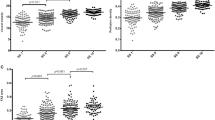
After averaging, all OCTA images showed marked improvement in image quality with less background noise and better vessel continuity. The number of microaneurysms in both the SCP and DCP was larger in single images than in averaged images. A significant increase in the detection rate of capillary telangiectasia in the DCP was noted after image averaging. The number of eyes with disrupted foveal avascular zone (FAZ) decreased significantly after averaging (P = .0253). Five eyes (15.2%) with a disrupted FAZ on the single image showed an intact FAZ after averaging. Vessel length density (VLD) and fractal dimension (FD) significantly decreased and vessel diameter index (VDI) increased after averaging, while significant changes were not observed in vessel density (VD) in both the SCP and DCP. In the SCP, lower VD, VLD, and fractal dimension were significantly correlated with worse visual acuity.
Conclusions
OCTA averaging has a significant effect on qualitative and quantitative assessments in eyes with BRVO.




Similar content being viewed by others
References
Hayreh SS, Zimmerman MB (2014) Branch retinal vein occlusion: natural history of visual outcome. JAMA Ophthalmol 132(1):13–22. https://doi.org/10.1001/jamaophthalmol.2013.5515
Heier JS, Campochiaro PA, Yau L, Li Z, Saroj N, Rubio RG, Lai P (2012) Ranibizumab for macular edema due to retinal vein occlusions: long-term follow-up in the HORIZON trial. Ophthalmology 119(4):802–809. https://doi.org/10.1016/j.ophtha.2011.12.005
Scott IU, Vanveldhuisen PC, Oden NL, Ip MS, Domalpally A, Doft BH, Elman MJ, Blodi BA, Group SSI (2012) Baseline characteristics and response to treatment of participants with hemiretinal compared with branch retinal or central retinal vein occlusion in the standard care vs corticosteroid for retinal vein occlusion (SCORE) study: SCORE study report 14. Arch Ophthalmol 130(12):1517–1524. https://doi.org/10.1001/archophthalmol.2012.2728
Tan MH, McAllister IL, Gillies ME, Verma N, Banerjee G, Smithies LA, Wong WL, Wong TY (2014) Randomized controlled trial of intravitreal ranibizumab versus standard grid laser for macular edema following branch retinal vein occlusion. Am J Ophthalmol 157(1):237–247 e231. https://doi.org/10.1016/j.ajo.2013.08.013
Ota M, Tsujikawa A, Murakami T, Yamaike N, Sakamoto A, Kotera Y, Miyamoto K, Kita M, Yoshimura N (2008) Foveal photoreceptor layer in eyes with persistent cystoid macular edema associated with branch retinal vein occlusion. Am J Ophthalmol 145(2):273–280. https://doi.org/10.1016/j.ajo.2007.09.019
Murakami T, Tsujikawa A, Ohta M, Miyamoto K, Kita M, Watanabe D, Takagi H, Yoshimura N (2007) Photoreceptor status after resolved macular edema in branch retinal vein occlusion treated with tissue plasminogen activator. Am J Ophthalmol 143(1):171–173. https://doi.org/10.1016/j.ajo.2006.08.030
Ota M, Tsujikawa A, Ojima Y, Miyamoto K, Murakami T, Ogino K, Akagi-Kurashige Y, Muraoka Y, Yoshimura N (2012) Retinal sensitivity after resolution of the macular edema associated with retinal vein occlusion. Graefes Arch Clin Exp Ophthalmol 250(5):635–644. https://doi.org/10.1007/s00417-011-1860-0
Finkelstein D (1992) Ischemic macular edema. Recognition and favorable natural history in branch vein occlusion. Arch Ophthalmol 110(10):1427–1434
Nagiel A, Sadda SR, Sarraf D (2015) A promising future for optical coherence tomography angiography. JAMA Ophthalmol 133(6):629–630. https://doi.org/10.1001/jamaophthalmol.2015.0668
Spaide RF, Fujimoto JG, Waheed NK, Sadda SR, Staurenghi G (2018) Optical coherence tomography angiography. Prog Retin Eye Res 64:1–55. https://doi.org/10.1016/j.preteyeres.2017.11.003
Kadomoto S, Muraoka Y, Ooto S, Miwa Y, Iida Y, Suzuma K, Murakami T, Ghashut R, Tsujikawa A, Yoshimura N (2018) Evaluation of macular ischemia in eyes with branch retinal vein occlusion: an optical coherence tomography angiography study. Retina 38(2):272–282. https://doi.org/10.1097/IAE.0000000000001541
Wakabayashi T, Sato T, Hara-Ueno C, Fukushima Y, Sayanagi K, Shiraki N, Sawa M, Ikuno Y, Sakaguchi H, Nishida K (2017) Retinal microvasculature and visual acuity in eyes with branch retinal vein occlusion: imaging analysis by optical coherence tomography angiography. Invest Ophthalmol Vis Sci 58(4):2087–2094. https://doi.org/10.1167/iovs.16-21208
Lei J, Durbin MK, Shi Y, Uji A, Balasubramanian S, Baghdasaryan E, Al-Sheikh M, Sadda SR (2017) Repeatability and reproducibility of superficial macular retinal vessel density measurements using optical coherence tomography angiography en face images. JAMA Ophthalmol 135(10):1092–1098. https://doi.org/10.1001/jamaophthalmol.2017.3431
Uji A, Balasubramanian S, Lei J, Baghdasaryan E, Al-Sheikh M, Sadda SR (2017) Impact of multiple en face image averaging on quantitative assessment from optical coherence tomography angiography images. Ophthalmology 124(7):944–952. https://doi.org/10.1016/j.ophtha.2017.02.006
Uji A, Balasubramanian S, Lei J, Baghdasaryan E, Al-Sheikh M, Borrelli E, Sadda SR (2018) Multiple enface image averaging for enhanced optical coherence tomography angiography imaging. Acta Ophthalmol 96(7):e820–e827. https://doi.org/10.1111/aos.13740
Uji A, Balasubramanian S, Lei J, Baghdasaryan E, Al-Sheikh M, Sadda SR (2017) Choriocapillaris imaging using multiple en face optical coherence tomography angiography image averaging. JAMA Ophthalmol 135(11):1197–1204. https://doi.org/10.1001/jamaophthalmol.2017.3904
Mo S, Phillips E, Krawitz BD, Garg R, Salim S, Geyman LS, Efstathiadis E, Carroll J, Rosen RB, Chui TY (2017) Visualization of radial Peripapillary capillaries using optical coherence tomography angiography: the effect of image averaging. PLoS One 12(1):e0169385. https://doi.org/10.1371/journal.pone.0169385
Suzuki N, Hirano Y, Yoshida M, Tomiyasu T, Uemura A, Yasukawa T, Ogura Y (2016) Microvascular abnormalities on optical coherence tomography angiography in macular edema associated with branch retinal vein occlusion. Am J Ophthalmol 161:126–132 e121. https://doi.org/10.1016/j.ajo.2015.09.038
Coscas F, Glacet-Bernard A, Miere A, Caillaux V, Uzzan J, Lupidi M, Coscas G, Souied EH (2016) Optical coherence tomography angiography in retinal vein occlusion: evaluation of superficial and deep capillary plexa. Am J Ophthalmol 161(160–171):e161–e162. https://doi.org/10.1016/j.ajo.2015.10.008
Kim AY, Chu Z, Shahidzadeh A, Wang RK, Puliafito CA, Kashani AH (2016) Quantifying microvascular density and morphology in diabetic retinopathy using spectral-domain optical coherence tomography angiography. Invest Ophthalmol Vis Sci 57(9):OCT362–OCT370. https://doi.org/10.1167/iovs.15-18904
Arichika S, Uji A, Ooto S, Miyamoto K, Yoshimura N (2014) Adaptive optics-assisted identification of preferential erythrocyte aggregate pathways in the human retinal microvasculature. PLoS One 9(2):e89679. https://doi.org/10.1371/journal.pone.0089679
Martin JA, Roorda A (2009) Pulsatility of parafoveal capillary leukocytes. Exp Eye Res 88(3):356–360. https://doi.org/10.1016/j.exer.2008.07.008
Ploner SB, Moult EM, Choi W, Waheed NK, Lee B, Novais EA, Cole ED, Potsaid B, Husvogt L, Schottenhamml J, Maier A, Rosenfeld PJ, Duker JS, Hornegger J, Fujimoto JG (2016) Toward quantitative optical coherence tomography angiography: visualizing blood flow speeds in ocular pathology using variable interscan time analysis. Retina 36(Suppl 1):S118–S126. https://doi.org/10.1097/IAE.0000000000001328
Moore J, Bagley S, Ireland G, McLeod D, Boulton ME (1999) Three dimensional analysis of microaneurysms in the human diabetic retina. J Anat 194(Pt 1):89–100
Iida Y, Muraoka Y, Uji A, Ooto S, Murakami T, Suzuma K, Tsujikawa A, Arichika S, Takahashi A, Miwa Y, Yoshimura N (2017) Associations between macular edema and circulatory status in eyes with retinal vein occlusion: an adaptive optics scanning laser ophthalmoscopy study. Retina 37(10):1896–1904. https://doi.org/10.1097/IAE.0000000000001433
Arichika S, Uji A, Murakami T, Unoki N, Yoshitake S, Dodo Y, Ooto S, Miyamoto K, Yoshimura N (2014) Retinal hemorheologic characterization of early-stage diabetic retinopathy using adaptive optics scanning laser ophthalmoscopy. Invest Ophthalmol Vis Sci 55(12):8513–8522. https://doi.org/10.1167/iovs.14-15121
Spaide RF, Fujimoto JG, Waheed NK (2015) Image artifacts in optical coherence tomography angiography. Retina 35(11):2163–2180. https://doi.org/10.1097/IAE.0000000000000765
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Akihito Uji, Alcon (Recipient), Senju (Recipient), Canon (Recipient); SriniVas R. Sadda, Carl Zeiss Meditec (Financial Support), Optos (Financial Support, Consultant), Allergan (Financial Support, Consultant), Genentech (Financial Support, Consultant), Alcon (Consultant); Novartis (Consultant); Roche (Consultant), Regeneron (Consultant), Bayer (Consultant), Thrombogenics (Consultant), Stemm Cells, Inc. (Consultant), Avalanche (Consultant); Yuki Muraoka, Bayer (Recipient), Novartis Pharma K.K. (Recipient), Senju (Recipient), Nidek (Recipient); Shin Kadomoto, Nidek (Recipient), Canon (Recipient); Sotaro Ooto, Novartis Pharma K.K. (Recipient), Bayer (Recipient), Santen (Recipient), Senju (Recipient); Tomoaki Murakami, Bayer (Recipient), Novartis Pharma K.K. (Recipient), Santen (Recipient), Senju (Recipient); Tadamichi Akagi, Alcon (Recipient), Kowa (Recipient), Pfizer (Recipient), Santen (Recipient), Senju (Recipient), Canon (Recipient); Akitaka Tsujikawa, Pfizer (Financial Support, Recipient), Novartis Pharma K.K. (Financial Support, Recipient), Bayer (Financial Support, Recipient), Alcon (Financial Support, Recipient), Santen (Financial Support, Recipient), Senju (Financial Support, Recipient), Nidek (Recipient), Kowa (Financial Support, Recipient), Hoya (Financial Support, Recipient), AMO Japan (Financial Support, Recipient).
Ethical approval
All study procedures involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual study participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental Figure 1.
Difference in the influence of image averaging on quantitative measurements between binarized and skeletonized images. In binarized images, decreased pixels corresponding to background noise (black circles) might be equal to the increased pixels in annealed vessels after averaging. On the other hand, in skeletonized images, the number of decreased pixels corresponding to background noise is larger than the increased pixels in annealed vessels after averaging, because both thick and thin vessels are skeletonized into a single pixel. (PNG 1173 kb)
Rights and permissions
About this article
Cite this article
Uji, A., Sadda, S.R., Muraoka, Y. et al. Effect of image averaging on optical coherence tomography angiography data in eyes with branch retinal vein occlusion. Graefes Arch Clin Exp Ophthalmol 258, 1639–1648 (2020). https://doi.org/10.1007/s00417-020-04713-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04713-9