Abstract
Purpose
To assess the real-world 5-year treatment outcomes of ranibizumab therapy in Japanese patients with neovascular age-related macular degeneration (AMD).
Methods
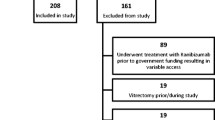
This was a retrospective, observational, and open-label effectiveness study that included 295 eyes. The participants were patients with treatment-naïve neovascular AMD who received intravitreal ranibizumab (IVR) monthly injection at least three times as the loading phase, followed by further injections as needed (pro re nata (PRN)) and follow-up assessments for 5 years. Outcomes were determined at least 5 years after the first ranibizumab injection.
Results
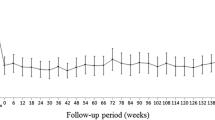
Mean logMAR best-corrected visual acuity (BCVA) at baseline was 0.52. The mean BCVA significantly improved after three loading injections; however, it declined gradually. The BCVA at 1 year was significantly better than the baseline BCVA, whereas the 3-year, 4-year, and 5-year BCVA values were significantly lower than the baseline values. The average central foveal thickness improved significantly from 366 ± 125 μm to 268 ± 134 μm (p < 0.0001). Macular atrophy was significantly more likely to occur in cases with classic choroidal neovascularization (CNV) than in cases with other AMD (p = 0.01).
Conclusions
IVR is well tolerated in eyes with AMD. However, a PRN regimen for AMD may have limited real-world effectiveness for long-term maintenance of improved visual acuity. Macular atrophy may occur more frequently in classic CNV. To maintain good vision, IVR treatment should be started earlier and performed continuously.



Similar content being viewed by others
References
Gheorghe A, Mahdi L, Musat O (2015) Age-related macular degeneration. Rom J Ophthalmol 59:74–77
Francis PJ, Klein ML (2011) Update on the role of genetics in the onset of age-related macular degeneration. Clin Ophthalmol 5:1127–1133. https://doi.org/10.2147/OPTH.S11627
Ambati J, Fowler BJ (2012) Mechanisms of age-related macular degeneration. Neuron 75:26–39. https://doi.org/10.1016/j.neuron.2012.06.018
Macular Photocoagulation Study Group (1991) Laser photocoagulation of subfoveal neovascular lesions in age-related macular degeneration. Results of a randomized clinical trial. Arch Ophthalmol 109:1220–1231
Treatment of age-related macular degeneration with photodynamic therapy (TAP) Study Group (1999) Photodynamic therapy of subfoveal choroidal neovascularization in age-related macular degeneration with verteporfin one-year results of 2 randomized clinical trials—TAP report. Arch Ophthalmol 117:1329–1345
Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, Kim RY, Group MS (2006) Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 355:1419–1431. https://doi.org/10.1056/NEJMoa054481
Brown DM, Kaiser PK, Michels M, Soubrane G, Heier JS, Kim RY, Sy JP, Schneider S, Group AS (2006) Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 355:1432–1444. https://doi.org/10.1056/NEJMoa062655
Brown DM, Heier JS, Ciulla T, Benz M, Abraham P, Yancopoulos G, Stahl N, Ingerman A, Vitti R, Berliner AJ, Yang K, Nguyen QD, Investigators C-I (2011) Primary endpoint results of a phase II study of vascular endothelial growth factor trap-eye in wet age-related macular degeneration. Ophthalmology 118:1089–1097. https://doi.org/10.1016/j.ophtha.2011.02.039
Singer MA, Awh CC, Sadda S, Freeman WR, Antoszyk AN, Wong P, Tuomi L (2012) HORIZON: an open-label extension trial of ranibizumab for choroidal neovascularization secondary to age-related macular degeneration. Ophthalmology 119:1175–1183. https://doi.org/10.1016/j.ophtha.2011.12.016
Rofagha S, Bhisitkul RB, Boyer DS, Sadda SR, Zhang K, Group S-US (2013) Seven-year outcomes in ranibizumab-treated patients in ANCHOR, MARINA, and HORIZON: a multicenter cohort study (SEVEN-UP). Ophthalmology 120:2292–2299. https://doi.org/10.1016/j.ophtha.2013.03.046
Comparison of Age-related Macular Degeneration Treatments Trials Research G, Martin DF, Maguire MG, Fine SL, Ying GS, Jaffe GJ, Grunwald JE, Toth C, Redford M, Ferris FL 3rd (2012) Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 119:1388–1398. https://doi.org/10.1016/j.ophtha.2012.03.053
Comparison of Age-related Macular Degeneration Treatments Trials Research G, Maguire MG, Martin DF, Ying GS, Jaffe GJ, Daniel E, Grunwald JE, Toth CA, Ferris FL 3rd, Fine SL (2016) Five-year outcomes with anti-vascular endothelial growth factor treatment of neovascular age-related macular degeneration: the comparison of age-related macular degeneration treatments trials. Ophthalmology 123:1751–1761. https://doi.org/10.1016/j.ophtha.2016.03.045
Boulanger-Scemama E, Sayag D, Ha Chau Tran T, Quaranta-El Maftouhi M, Rumen F, Creuzot-Garcher C, Blanco Garavito R, Jung C, Souied E (2016) Ranibizumab and exudative age-related macular degeneration: 5-year multicentric functional and anatomical results in real-life practice. J Fr Ophtalmol 39:668–674. https://doi.org/10.1016/j.jfo.2016.06.001
Heimes B, Lommatzsch A, Zeimer M, Gutfleisch M, Spital G, Dietzel M, Pauleikhoff D (2011) Long-term visual course after anti-VEGF therapy for exudative AMD in clinical practice evaluation of the German reinjection scheme. Graefes Arch Clin Exp Ophthalmol 249:639–644. https://doi.org/10.1007/s00417-010-1524-5
Maguire P, Vine AK (1986) Geographic atrophy of the retinal pigment epithelium. Am J Ophthalmol 102:621–625
Holz FG, Wolfensberger TJ, Piguet B, Gross-Jendroska M, Wells JA, Minassian DC, Chisholm IH, Bird AC (1994) Bilateral macular drusen in age-related macular degeneration: prognosis and risk factors. Ophthalmology 101:1522–1528
Sunness JS, Gonzalez-Baron J, Applegate CA, Bressler NM, Tian Y, Hawkins B, Barron Y, Bergman A (1999) Enlargement of atrophy and visual acuity loss in the geographic atrophy form of age-related macular degeneration. Ophthalmology 106:1768–1779. https://doi.org/10.1016/S0161-6420(99)90340-8
Grunwald JE, Pistilli M, Ying GS, Maguire MG, Daniel E, Martin DF, Comparison of Age-related Macular Degeneration Treatments Trials Research G (2015) Growth of geographic atrophy in the comparison of age-related macular degeneration treatments trials. Ophthalmology 122:809–816. https://doi.org/10.1016/j.ophtha.2014.11.007
Grunwald JE, Pistilli M, Daniel E, Ying GS, Pan W, Jaffe GJ, Toth CA, Hagstrom SA, Maguire MG, Martin DF, Comparison of Age-Related Macular Degeneration Treatments Trials Research G (2017) Incidence and growth of geographic atrophy during 5 years of comparison of age-related macular degeneration treatments trials. Ophthalmology 124:97–104. https://doi.org/10.1016/j.ophtha.2016.09.012
Holz FG, Strauss EC, Schmitz-Valckenberg S, van Lookeren Campagne M (2014) Geographic atrophy: clinical features and potential therapeutic approaches. Ophthalmology 121:1079–1091. https://doi.org/10.1016/j.ophtha.2013.11.023
Takahashi A, Ooto S, Yamashiro K, Oishi A, Tamura H, Nakanishi H, Ueda-Arakawa N, Tsujikawa A, Yoshimura N (2016) Photoreceptor damage and reduction of retinal sensitivity surrounding geographic atrophy in age-related macular degeneration. Am J Ophthalmol 168:260–268. https://doi.org/10.1016/j.ajo.2016.06.006
Wecker T, Ehlken C, Buhler A, Lange C, Agostini H, Bohringer D, Stahl A (2017) Five-year visual acuity outcomes and injection patterns in patients with pro-re-nata treatments for AMD, DME, RVO and myopic CNV. Br J Ophthalmol 101:353–359. https://doi.org/10.1136/bjophthalmol-2016-308668
Takahashi Y, Koizumi H, Hasegawa T, Izumi T, Maruko I, Sonoda S, Sakamoto T, Iida T (2018) Comparison of subfoveal choroidal structures in typical neovascular age-related macular degeneration and polypoidal choroidal vasculopathy. Jpn J Ophthalmol 62:576–583. https://doi.org/10.1007/s10384-018-0615-4
Ogasawara M, Koizumi H, Yamamoto A, Itagaki K, Saito M, Maruko I, Okada AA, Iida T, Sekiryu T (2018) Prognostic factors after aflibercept therapy for typical age-related macular degeneration and polypoidal choroidal vasculopathy. Jpn J Ophthalmol 62:584–591. https://doi.org/10.1007/s10384-018-0605-6
Bhisitkul RB, Mendes TS, Rofagha S, Enanoria W, Boyer DS, Sadda SR, Zhang K (2015) Macular atrophy progression and 7-year vision outcomes in subjects from the ANCHOR, MARINA, and HORIZON studies: the SEVEN-UP study. Am J Ophthalmol 159:915–924 e912. https://doi.org/10.1016/j.ajo.2015.01.032
Munk MR, Ceklic L, Ebneter A, Huf W, Wolf S, Zinkernagel MS (2016) Macular atrophy in patients with long-term anti-VEGF treatment for neovascular age-related macular degeneration. Acta Ophthalmol 94:e757–e764. https://doi.org/10.1111/aos.13157
Koh A, Lai TYY, Takahashi K, Wong TY, Chen LJ, Ruamviboonsuk P, Tan CS, Feller C, Margaron P, Lim TH, Lee WK, group EIs (2017) Efficacy and safety of ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: a randomized clinical trial. JAMA Ophthalmol 135:1206–1213. https://doi.org/10.1001/jamaophthalmol.2017.4030
Oshima Y, Kimoto K, Yoshida N, Fujisawa K, Sonoda S, Kubota T, Murata T, Sakamoto T, Yoshida S, Sonoda KH, Ishibashi T (2017) One-year outcomes following intravitreal aflibercept for polypoidal choroidal vasculopathy in Japanese patients: the APOLLO study. Ophthalmologica 238:163–171. https://doi.org/10.1159/000477448
Funding
This study was funded by the JSPS KAKENHI Grant Number (Kiban C 17K11454 (to Y.O.)).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Author T.I. has received research grants from Bayer Yakuhin, Ltd., and Santen Pharmaceutical Co., Ltd. Author K.S. has received research grants from Santen Pharmaceutical Co., Ltd.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all the participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wada, I., Oshima, Y., Shiose, S. et al. Five-year treatment outcomes following intravitreal ranibizumab injections for neovascular age-related macular degeneration in Japanese patients. Graefes Arch Clin Exp Ophthalmol 257, 1411–1418 (2019). https://doi.org/10.1007/s00417-019-04361-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-019-04361-8