Abstract
Purpose
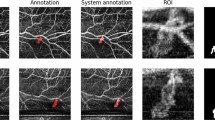
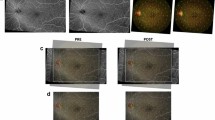
To quantify the preoperative neovascular change pattern on the fibrovascular membrane (FVM) within 7 days after intravitreal injection of conbercept (IVC) using optical coherence tomography angiography (OCTA) in proliferative diabetic retinopathy (PDR).
Methods
Prospective, observational study of PDR patients with visible FVM receiving or not receiving IVC. Neovascular changes were assessed by OCTA pre-IVC and 1, 3, 5, and 7 days post-IVC. Vessel skeleton density (SD) and vessel density (VD) were quantified by an intensity-based optical microangiography algorithm. The interclass correlation coefficient (ICC) was calculated to assess the agreement between measurements. The SD and VD were compared between follow-ups using repeated-measures analysis in the IVC group.
Results
The ICC was 0.992 (95% confidence interval [CI]: 0.982–0.996) for SD and 0.926 (95% CI: 0.838–0.912) for VD of neovascularization. The neovascularization on FVM significantly regressed in the IVC group (n = 16) compared with no IVC (n = 8) (p = 0.001 for SD and p < 0.001 for VD). The comparisons between consecutive follow-ups showed a statistically significant reduction in SD and VD at 1 and 3 days post-IVC. However, from day 3 onward, the SD and VD remained unchanged. There was no development or progression of tractional retinal detachment within the 7-day period after IVC.
Conclusion
OCTA–based quantification of the neovascularization on FVM in PDR is feasible, with high inter-reader agreement. The regression of neovascularization reaches a plateau 3 days after IVC.
Clinical Trial Registration
This trial is registered with the Chinese Clinical Trial Registry (http://www.chictr.org.cn, registration number ChiCTR-IPR-17014160).





Similar content being viewed by others
References
Cheung N, Mitchell P, Wong TY (2010) Diabetic retinopathy. Lancet 376:124–136. https://doi.org/10.1016/S0140-6736(09)62124-3
Adamis AP, Miller JW, Bernal M-T, D'Amico DJ, Folkman J, Yeo T-K, Yeo K-T (1994) Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol 118:445–450. https://doi.org/10.1016/S0002-9394(14)75794-0
Liu L, Tham YC, Cheng CY (2017) Intravitreal aflibercept for proliferative diabetic retinopathy. Lancet 390:2140–2141. https://doi.org/10.1016/s0140-6736(17)32426-1
Avery RL, Pearlman J, Pieramici DJ, Rabena MD, Castellarin AA, Nasir MA, Giust MJ, Wendel R, Patel A (2006) Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology 113:1695–1705.e1696. https://doi.org/10.1016/j.ophtha.2006.05.064
Su L, Ren X, Wei H, Zhao L, Zhang X, Liu J, Su C, Tan L, Li X (2016) Intravitreal conbercept (KH902) for surgical treatment of severe proliferative diabetic retinopathy. Retina 36:938–943. https://doi.org/10.1097/iae.0000000000000900
Smith JM, Steel DH (2015) Anti-vascular endothelial growth factor for prevention of postoperative vitreous cavity haemorrhage after vitrectomy for proliferative diabetic retinopathy. Cochrane Database Syst Rev Cd008214. https://doi.org/10.1002/14651858.CD008214.pub3
Zhang ZH, Liu HY, Hernandez-Da Mota SE, Romano MR, Falavarjani KG, Ahmadieh H, Xu X, Liu K (2013) Vitrectomy with or without preoperative intravitreal bevacizumab for proliferative diabetic retinopathy: a meta-analysis of randomized controlled trials. Am J Ophthalmol 156:106–115.e102. https://doi.org/10.1016/j.ajo.2013.02.008
Zhao XY, Xia S, Chen YX (2017) Antivascular endothelial growth factor agents pretreatment before vitrectomy for complicated proliferative diabetic retinopathy: a meta-analysis of randomised controlled trials. Br J Ophthalmol. https://doi.org/10.1136/bjophthalmol-2017-311344
Li X, Xu G, Wang Y, Xu X, Liu X, Tang S, Zhang F, Zhang J, Tang L, Wu Q, Luo D, Ke X (2014) Safety and efficacy of conbercept in neovascular age-related macular degeneration: results from a 12-month randomized phase 2 study: AURORA study. Ophthalmology 121:1740–1747. https://doi.org/10.1016/j.ophtha.2014.03.026
Li F, Sun M, Guo J, Ma A, Zhao B (2017) Comparison of conbercept with ranibizumab for the treatment of macular edema secondary to branch retinal vein occlusion. Curr Eye Res 42:1174–1178. https://doi.org/10.1080/02713683.2017.1285943
Li F, Zhang L, Wang Y, Xu W, Jiao W, Ma A, Zhao B (2017) One-year outcome of Conbercept therapy for diabetic macular edema. Curr Eye Res 1–6. https://doi.org/10.1080/02713683.2017.1379542
Yang X, Xu J (2016) A Randomized Controlled Trial of Conbercept Pretreatment before Vitrectomy in Proliferative Diabetic Retinopathy 2016:2473234. https://doi.org/10.1155/2016/2473234
Arevalo JF, Maia M, Flynn H, Saravia M, Avery RL, Wu L, Farah ME, Pieramici DJ, Berrocal MH, Sanchez JG (2008) Tractional retinal detachment following intravitreal bevacizumab (Avastin) in patients with severe proliferative diabetic retinopathy. Br J Ophthalmol 92:213–216
Hu Z, Liu Q, Paulus YM (2016) New Frontiers in retinal imaging. Int Ophthalmic Res 2:148–158
Gupta A, Bansal R, Gupta V, Dogra MR (2012) Six-month visual outcome after pars plana vitrectomy in proliferative diabetic retinopathy with or without a single preoperative injection of intravitreal bevacizumab. Int Ophthalmol 32:135–144. https://doi.org/10.1007/s10792-012-9541-5
Erdol H, Turk A, Akyol N, Imamoglu HI (2010) The results of intravitreal bevacizumab injections for persistent neovascularizations in proliferative diabetic retinopathy after photocoagulation therapy. Retina 30:570–577. https://doi.org/10.1097/IAE.0b013e3181bf5ff0
Abdelhakim MA, Macky TA, Mansour KA, Mortada HA (2011) Bevacizumab (Avastin) as an adjunct to vitrectomy in the management of severe proliferative diabetic retinopathy: a prospective case series. Ophthalmic Res 45:23–30. https://doi.org/10.1159/000314721
Hu Z, Ding Y, Zheng X, Yuan S, Li J, Xie P, Liu Q (2018) Thin rigid contact Lens used in vitreous-retinal surgery for corneal protection: a randomized controlled trial. Eye Contact Lens. https://doi.org/10.1097/ICL.0000000000000491
Jia Y, Tan O, Tokayer J, Potsaid B, Wang Y, Liu JJ, Kraus MF, Subhash H, Fujimoto JG, Hornegger J (2012) Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt Express 20:4710–4725. https://doi.org/10.1364/OE.20.004710
Amoroso F, Miere A, Semoun O, Jung C, Capuano V, Souied EH (2017) Optical coherence tomography angiography reproducibility of lesion size measurements in neovascular age-related macular degeneration (AMD). Br J Ophthalmol bjophthalmol-2017-310569. https://doi.org/10.1136/bjophthalmol-2017-310569
Venugopal JP, Rao HL, Weinreb RN, Pradhan ZS, Dasari S, Riyazuddin M, Puttiah NK, Rao DA, Devi S, Mansouri K (2017) Repeatability of vessel density measurements of optical coherence tomography angiography in normal and glaucoma eyes. Br J Ophthalmol bjophthalmol-2017-310637. https://doi.org/10.1136/bjophthalmol-2017-310637
Gao SS, Liu L, Bailey ST, Flaxel CJ, Huang D, Li D, Jia Y (2016) Quantification of choroidal neovascularization vessel length using optical coherence tomography angiography. J Biomed Opt 21:076010–076010. https://doi.org/10.1117/1.JBO.21.7.076010
Rosen RB, Andrade Romo JS, Krawitz BD, Mo S, Fawzi AA, Linderman R, Carroll J, Pinhas A, Chui TYP (2019) Earliest evidence of preclinical diabetic retinopathy revealed using OCT angiography (OCTA) perfused capillary density. Am J Ophthalmol. https://doi.org/10.1016/j.ajo.2019.01.012
Kaizu Y, Nakao S, Sekiryu H, Wada I, Yamaguchi M, Hisatomi T, Ikeda Y, Kishimoto J, Sonoda KH (2018) Retinal flow density by optical coherence tomography angiography is useful for detection of nonperfused areas in diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 256:2275–2282. https://doi.org/10.1007/s00417-018-4122-6
Dodo Y, Suzuma K, Ishihara K, Yoshitake S, Fujimoto M, Yoshitake T, Miwa Y, Murakami T (2017) Clinical relevance of reduced decorrelation signals in the diabetic inner choroid on optical coherence tomography angiography. Sci Rep 7:5227. https://doi.org/10.1038/s41598-017-05663-9
Onishi AC, Nesper PL, Roberts PK, Moharram GA, Chai H, Liu L, Jampol LM, Fawzi AA (2018) Importance of considering the middle capillary plexus on OCT angiography in diabetic retinopathy. Invest Ophthalmol Vis Sci 59:2167–2176. https://doi.org/10.1167/iovs.17-23304
Lu Y, Simonett JM, Wang J, Zhang M, Hwang T, Hagag AM, Huang D, Li D, Jia Y (2018) Evaluation of automatically quantified foveal avascular zone metrics for diagnosis of diabetic retinopathy using optical coherence tomography angiography. Invest Ophthalmol Vis Sci 59:2212–2221. https://doi.org/10.1167/iovs.17-23498
Sawada O, Ichiyama Y, Obata S, Ito Y, Kakinoki M, Sawada T, Saishin Y, Ohji M (2018) Comparison between wide-angle OCT angiography and ultra-wide field fluorescein angiography for detecting non-perfusion areas and retinal neovascularization in eyes with diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 256:1275–1280. https://doi.org/10.1007/s00417-018-3992-y
Schaal KB, Munk MR, Wyssmueller I, Berger LE, Zinkernagel MS, Wolf S (2019) Vascular abnormalities in diabetic retinopathy assessed with swept-source optical coherence tomography angiography widefield imaging. Retina 39:79–87. https://doi.org/10.1097/iae.0000000000001938
Singh A, Agarwal A, Mahajan S, Karkhur S, Singh R, Bansal R, Dogra MR, Gupta V (2017) Morphological differences between optic disc collaterals and neovascularization on optical coherence tomography angiography. Graefes Arch Clin Exp Ophthalmol 255:753–759. https://doi.org/10.1007/s00417-016-3565-x
Pan J, Chen D, Yang X, Zou R, Zhao K, Cheng D, Huang S, Zhou T, Yang Y, Chen F (2018) Characteristics of neovascularization in early stages of proliferative diabetic retinopathy by optical coherence tomography angiography. Am J Ophthalmol 192:146–156. https://doi.org/10.1016/j.ajo.2018.05.018
Zhang X, Wu C, Zhou LJ, Dai RP (2018) Observation of optic disc neovascularization using OCT angiography in proliferative diabetic retinopathy after intravitreal conbercept injections. Sci Rep 8:3972. https://doi.org/10.1038/s41598-018-22363-0
Lee J, Moon BG, Cho AR, Yoon YH (2016) Optical coherence tomography angiography of DME and its association with anti-VEGF treatment response. Ophthalmology 123:2368–2375. https://doi.org/10.1016/j.ophtha.2016.07.010
Ghasemi Falavarjani K, Iafe NA, Hubschman JP, Tsui I, Sadda SR, Sarraf D (2017) Optical coherence tomography angiography analysis of the foveal avascular zone and macular vessel density after anti-VEGF therapy in eyes with diabetic macular edema and retinal vein occlusion. Invest Ophthalmol Vis Sci 58:30–34. https://doi.org/10.1167/iovs.16-20579
Rizzo S, Genovesi-Ebert F, Di Bartolo E, Vento A, Miniaci S, Williams G (2008) Injection of intravitreal bevacizumab (Avastin) as a preoperative adjunct before vitrectomy surgery in the treatment of severe proliferative diabetic retinopathy (PDR). Graefes Arch Clin Exp Ophthalmol 246:837–842. https://doi.org/10.1007/s00417-008-0774-y
Kohno R-I, Hata Y, Mochizuki Y, Arita R, Kawahara S, Kita T, Miyazaki M, Hisatomi T, Ikeda Y, Aiello LP (2010) Histopathology of neovascular tissue from eyes with proliferative diabetic retinopathy after intravitreal bevacizumab injection. Am J Ophthalmol 150:223–229. https://doi.org/10.1016/j.ajo.2010.03.016
Manabe A, Shimada H, Hattori T, Nakashizuka H, Yuzawa M (2015) Randomized controlled study of intravitreal bevacizumab 0.16 mg injected one day before surgery for proliferative diabetic retinopathy. Retina (Philadelphia, Pa) 35:1800–1807. https://doi.org/10.1097/iae.0000000000000577
Di Lauro R, De Ruggiero P, Di Lauro R, Di Lauro MT, Romano MR (2010) Intravitreal bevacizumab for surgical treatment of severe proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 248:785–791. https://doi.org/10.1007/s00417-010-1303-3
El-Sabagh HA, Abdelghaffar W, Labib AM, Mateo C, Hashem TM, Al-Tamimi DM, Selim AA (2011) Preoperative intravitreal bevacizumab use as an adjuvant to diabetic vitrectomy: histopathologic findings and clinical implications. Ophthalmology 118:636–641. https://doi.org/10.1016/j.ophtha.2010.08.038
Da R Lucena D, Ribeiro JA, Costa RA, Barbosa JC, Scott IU, de Figueiredo-Pontes LL, Jorge R (2009) Intraoperative bleeding during vitrectomy for diabetic tractional retinal detachment with versus without preoperative intravitreal bevacizumab (IBeTra study). Br J Ophthalmol 93:688–691. https://doi.org/10.1136/bjo.2008.151233
Castillo J, Aleman I, Rush SW, Rush RB (2017) Preoperative bevacizumab administration in proliferative diabetic retinopathy patients undergoing vitrectomy: a randomized and controlled trial comparing interval variation. Am J Ophthalmol 183:1–10. https://doi.org/10.1016/j.ajo.2017.08.013
Farahvash M-S, Majidi AR, Roohipoor R, Ghassemi F (2011) Preoperative injection of intravitreal bevacizumab in dense diabetic vitreous hemorrhage. Retina (Philadelphia, Pa) 31:1254–1260. https://doi.org/10.1097/IAE.0b013e31820a68e5
Spaide RF, Fujimoto JG, Waheed NK (2015) Image artifacts in optical coherence tomography angiography. Retina (Philadelphia, Pa) 35:2163–2180. https://doi.org/10.1097/IAE.0000000000000765
Hirano T, Kakihara S, Toriyama Y, Nittala MG, Murata T, Sadda S (2017) Wide-field en face swept-source optical coherence tomography angiography using extended field imaging in diabetic retinopathy. Br J Ophthalmol. https://doi.org/10.1136/bjophthalmol-2017-311358
Acknowledgements
Mr. Song Zhang from Colinico Meditec, Inc. (Shanghai, China) provided guidance on the use of optical coherence tomography to obtain fibrovascular membrane data.
Funding
This study was supported by the National Key Project of Research and Development Plan (no. 2017YFA0104101) and General Project of the National Natural Science Fund (no. 81770973). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Clinical Trial Registration: http://www.chictr.org.cn Unique identifier: ChiCTR-IPR-17014160.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. S1
(PNG 113 kb)
Rights and permissions
About this article
Cite this article
Hu, Z., Su, Y., Xie, P. et al. OCT angiography-based monitoring of neovascular regression on fibrovascular membrane after preoperative intravitreal conbercept injection. Graefes Arch Clin Exp Ophthalmol 257, 1611–1619 (2019). https://doi.org/10.1007/s00417-019-04315-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-019-04315-0