Abstract
Objective
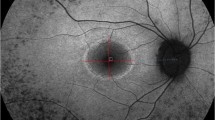
To evaluate and compare the B-scan OCT loss of ellipsoid zone, OCT en face thickness map constriction, and hyperautofluorescent ring constriction in RP patients.
Methods
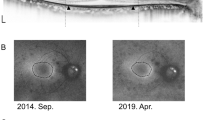
Retrospective case series study. Forty-eight eyes of 24 RP patients with a parafoveal hyperautofluorescent ring were studied. The diagnosis of RP was established by the presence of rod response impairment and a prevalent decrease of scotopic over photopic responses on electroretinography. The FAF and spectral-domain optical coherence tomography (SD-OCT) images were obtained from 24 patients with RP. The measurements of the EZ line width on B-scan OCT, hyperautofluorescent ring area on FAF, and hyperautofluorescent ring area on en face thickness map were performed by two independent graders. The measurements of these three parameters were correlated.
Results
The mean age of study patients was 46 years old (sd = 19). The external and internal FAF rings involving the fovea were identified in all study eyes. The area of the thickness ring decreased at an average rate of 0.5 (sd 0.4) mm2 per year (P < 0.001). The average rate of EZ-line constriction was estimated to be 123 (sd 63) μm per year (P < 0.001). The hyperautofluorescent ring area decreased at an average rate of 0.9 (sd 0.98) mm2 per year (P < 0.001). The strongest correlation was observed between hyperautofluorescent ring area and EZ-line width (r = 0.78).
Conclusions
We observed that the hyperautofluorescent ring area exhibits a faster progression rate than the area of the thickness ring. In addition, we found that the EZ-line width had a high positive correlation with the hyperautofluorescent ring area and a moderate positive correlation with area of the thickness ring.


Similar content being viewed by others
References
Hamel C (2006) Retinitis pigmentosa. Orphanet J Rare Dis 1(40). https://doi.org/10.1186/1750-1172-1-40
Hartong DT, Berson EL, Dryja TP (2006) Retinitis pigmentosa. Lancet 368:1795–1809. https://doi.org/10.1016/S0140-6736(06)69740-7
Robson AG, Lenassi E, Saihan Z et al (2012) Comparison of fundus autofluorescence with Photopic and scotopic fine matrix mapping in patients with retinitis Pigmentosa: 4- to 8-year follow-up. Investig Opthalmology Vis Sci 53:6187. https://doi.org/10.1167/iovs.12-10195
Wakabayashi T, Sawa M, Gomi F, Tsujikawa M (2010) Correlation of fundus autofluorescence with photoreceptor morphology and functional changes in eyes with retinitis pigmentosa. Acta Ophthalmol 88:e177–e183. https://doi.org/10.1111/j.1755-3768.2010.01926.x
Shen S, Sujirakul T, Tsang SH (2014) Next-generation sequencing revealed a novel mutation in the gene encoding the Beta subunit of rod phosphodiesterase. Ophthalmic Genet 35:142–150. https://doi.org/10.3109/13816810.2014.915328
Hood DC, Raza AS (2011) Method for comparing visual field defects to local RNFL and RGC damage seen on frequency domain OCT in patients with glaucoma. Biomed Opt Express. https://doi.org/10.1364/BOE.2.001097
Rangaswamy NV, Patel HM, Locke KG et al (2010) A comparison of visual field sensitivity to photoreceptor thickness in retinitis pigmentosa. Investig Ophthalmol Vis Sci. https://doi.org/10.1167/iovs.09-4945
Ho AC, Humayun MS, Dorn JD et al (2015) Long-term results from an Epiretinal prosthesis to restore sight to the blind. Ophthalmology. https://doi.org/10.1016/j.ophtha.2015.04.032
Milam AH, Li ZY, Fariss RN (1998) Histopathology of the human retina in retinitis pigmentosa. Prog Retin Eye Res 17:175–205
Humayun MS, De Juan E, Greenberg RJ, Marsh MJ (1997) Preservation of the inner retina in retinitis pigmentosa: a morphometric analysis. Investig Ophthalmol Vis Sci 38. doi https://doi.org/10.1001/archopht.1997.01100150513011
Li Q, Timmers AM, Hunter K et al (2001) Noninvasive imaging by optical coherence tomography to monitor retinal degeneration in the mouse. Invest Ophthalmol Vis Sci 42:2981–2989
Milam AH, Li ZY, Cideciyan AV, Jacobson SG (1996) Clinicopathologic effects of the Q64ter rhodopsin mutation in retinitis pigmentosa. Investig Ophthalmol Vis Sci 37:753–765
Berson EL, Adamian M (1992) Ultrastructural findings in an autopsy eye from a patient with Usher’s syndrome type II. Am J Ophthalmol 114:748–757. https://doi.org/10.1016/S0002-9394(14)74055-3
Hood DC, Lazow MA, Locke KG et al (2011) The transition zone between healthy and diseased retina in patients with retinitis pigmentosa. Invest Ophthalmol Vis Sci 52:101–108. https://doi.org/10.1167/iovs.10-5799\riovs.10-5799
Eriksson U, Alm A (2009) Macular thickness decreases with age in normal eyes: a study on the macular thickness map protocol in the stratus OCT. Br J Ophthalmol 93:1448–1452. https://doi.org/10.1136/bjo.2007.131094
van Velthoven MEJ, Faber DJ, Verbraak FD et al (2007) Recent developments in optical coherence tomography for imaging the retina. Prog Retin Eye Res 26:57–77. https://doi.org/10.1016/j.preteyeres.2006.10.002
Drexler W, Fujimoto JG (2008) State-of-the-art retinal optical coherence tomography. Prog Retin Eye Res 27:45–88
Sandberg MA, Brockhurst RJ, Gaudio AR, Berson EL (2005) The association between visual acuity and central retinal thickness in retinitis pigmentosa. Investig Ophthalmol Vis Sci 46:3349–3354. https://doi.org/10.1167/iovs.04-1383
Apushkin MA, Fishman GA, Alexander KR, Shahidi M (2007) Retinal thickness and visual thresholds measured in patients with retinitis pigmentosa. Retina 27:349–357. https://doi.org/10.1097/01.iae.0000224944.33863.18
Matsuo T, Morimoto N (2007) Visual acuity and perimacular retinal layers detected by optical coherence tomography in patients with retinitis pigmentosa. Br J Ophthalmol 91:888–890. https://doi.org/10.1136/bjo.2007.114538
Sugita T, Kondo M, Piao CH et al (2008) Correlation between macular volume and focal macular electroretinogram in patients with retinitis pigmentosa. Investig Ophthalmol Vis Sci 49:3551–3558. https://doi.org/10.1167/iovs.08-1954
Aleman TS, Duncan JL, Bieber ML et al (2001) Macular pigment and lutein supplementation in retinitis pigmentosa and usher syndrome. Investig Ophthalmol Vis Sci 42:1873–1881
Jacobson SG, Buraczynska M, Milam AH et al (1997) Disease expression in X-linked retinitis pigmentosa caused by a putative null mutation in the RPGR gene. Investig Ophthalmol Vis Sci 38:1983–1997
Marmor MF, Fulton AB, Holder GE, Miyake Y, Brigell M, Bach M (2009) ISCEV standard for full-field clinical electroretinography (2008 update). Doc Ophthalmol 118(1):69–77
Sujirakul T, Lin MK, Duong J et al (2015) Multimodal imaging of central retinal disease progression in a 2-year mean follow-up of retinitis Pigmentosa. Am J Ophthalmol 160:786–98 e4. https://doi.org/10.1016/j.ajo.2015.06.032
Cabral T, Sengillo JD, Duong JK et al (2017) Retrospective analysis of structural disease progression in retinitis Pigmentosa utilizing multimodal imaging. Sci Rep 7:10347. https://doi.org/10.1038/s41598-017-10473-0
Murakami T, Akimoto M, Ooto S et al (2008) Association between abnormal autofluorescence and photoreceptor disorganization in retinitis Pigmentosa. Am J Ophthalmol 145:687–694. https://doi.org/10.1016/j.ajo.2007.11.018
Aizawa S, Mitamura Y, Hagiwara A et al (2010) Changes of fundus autofluorescence, photoreceptor inner and outer segment junction line, and visual function in patients with retinitis pigmentosa. Clin Exp Ophthalmol 38:597–604. https://doi.org/10.1111/j.1442-9071.2010.02321.x
Funding
Jonas Children’s Vision Care is supported by the National Institute of Health [R24 EY027285, 5P30EY019007, R01EY018213, R01EY024698, R01EY026682, R21AG050437], National Cancer Institute Core [5P30CA013696], the Schneeweiss Stem Cell Fund, New York State [SDHDOH01-C32590GG-3450000], the Foundation Fighting Blindness New York Regional Research Center Grant [C-NY05-0705-0312].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent was obtained from all study patients as outlined by the protocol #AAAR0284 approved by the Institutional Review Board at Columbia University Medical Center. This study adhered to the tenets of the Declaration of Helsinki, and none of the study data, including imaging and genetic testing results, are identifiable to individual patients.
Competing interest
The authors declare that they have no competing interest.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 722 kb)
Rights and permissions
About this article
Cite this article
Takahashi, V.K.L., Takiuti, J.T., Jauregui, R. et al. Correlation between B-scan optical coherence tomography, en face thickness map ring and hyperautofluorescent ring in retinitis pigmentosa patients. Graefes Arch Clin Exp Ophthalmol 257, 1601–1609 (2019). https://doi.org/10.1007/s00417-019-04265-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-019-04265-7