Abstract
Purpose
To investigate the effects of hyaluronic acid (HA) on the inflammation of corneal fibroblasts induced by lipopolysaccharide (LPS).
Methods
Primary rabbit corneal keratocytes were isolated with collagenase. The keratocytes were cultured in a serum-containing medium to induce corneal fibroblasts, which represented the wound repair phenotype of corneal keratocytes. Corneal fibroblasts were treated with LPS with or without 4-methylumbelliferone (4-MU) / high molecular weight hyaluronic acid (HMWHA). The gene expression was evaluated via real-time PCR, immunofluorescence, and western blot. The release of inflammatory cytokines and HA was determined by ELISA.
Results
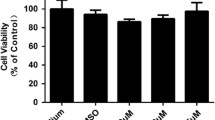
Three types of hyaluronan synthase (HAS) were detected in corneal fibroblasts. LPS stimulation caused the up-regulation of HAS1 and HAS2 expression in corneal fibroblasts. LPS-induced HAS2 expression was significantly inhibited by 4-MU, and accompanied by decreased HA release by the corneal fibroblasts. In the corneal fibroblasts, 4-MU reduced the LPS-stimulated up-regulation of inflammatory cytokines including IL-1, IL-6, IL-8, TNF-α, and also attenuated the LPS-induced up-regulation of inflammatory related receptors including TLR2, TLR4, CD44, and CXCR1. HMWHA treatment resulted in a significant decline in the expression of IL-6, IL-8, TLR4, and CXCR1 responded to LPS stimulation. Consistent with mRNA expression of level, the up-regulation of the release of IL-6 and IL-8 induced by LPS in corneal fibroblasts was significantly attenuated by 4-MU and HMWHA. The LPS-induced expression of IL-8 and its receptor CXCR1 at both the mRNA and protein level were significantly attenuated by 4-MU and HMWHA.
Conclusion
The inhibitor of HA synthesis 4-MU, and HMWHA successfully reduced LPS-induced inflammation in corneal fibroblasts. The mechanism might be via the inhibition of LPS-induced TLR4 up-regulation.





Similar content being viewed by others
References
Schaefer FDR, Bruttin O, Zografos LO, Guex-Crosier Y (2001) Bacterial keratitis: a prospective clinical and microbiological study. Br J Ophthalmol 10(5):842–847
Guo N et al (2007) A rapid transient increase in hyaluronan synthase-2 mRNA initiates secretion of hyaluronan by corneal keratocytes in response to transforming growth factor beta. J Biol Chem 282(17):12475–12483
Kumagai N et al (2005) Lipopolysaccharide-induced expression of intercellular adhesion molecule-1 and chemokines in cultured human corneal fibroblasts. Invest Ophthalmol Vis Sci 46(1):114–120
Kumagai N, Fukuda K, Fujitsu Y, Yamamoto K, Nishida T (2006) Role of structural cells of the cornea and conjunctiva in the pathogenesis of vernal keratoconjunctivitis. Prog Retin Eye Res 25(2):165–187
Lu Y et al (2006) Inhibition by triptolide of chemokine, proinflammatory cytokine, and adhesion molecule expression induced by lipopolysaccharide in corneal fibroblasts. Invest Ophthalmol Vis Sci 47(9):3796–3800
Akira S (2003) Toll-like receptor signaling. J Biol Chem 278(40):38105–38108
Mitsui Y et al (2008) Hyaluronic acid inhibits mRNA expression of proinflammatory cytokines and cyclooxygenase-2/prostaglandin E(2) production via CD44 in interleukin-1-stimulated subacromial synovial fibroblasts from patients with rotator cuff disease. J Orthop Res 26(7):1032–1037
Campo GM et al (2011) Hyaluronan reduces inflammation in experimental arthritis by modulating TLR-2 and TLR-4 cartilage expression. Biochim Biophys Acta 1812(9):1170–1181
Kogan G, Soltés L, Stern R, Gemeiner P (2007) Hyaluronic acid: a natural biopolymer with a broad range of biomedical and industrial applications. Biotechnol Lett 29(1):17–25
Vigetti D et al (2014) Hyaluronan: biosynthesis and signaling. Biochim Biophys Acta 1840(8):2452–2459
Schulz A, Vestweber AM, Dressler D (2009) Anti-inflammatory action of a hyaluronic acid-chondroitin sulfate preparation in an in vitro bladder model. Aktuelle Urol 40(2):109–112
Hirabara S, Kojima T, Takahashi N, Hanabayashi M, Ishiguro N (2013) Hyaluronan inhibits TLR-4 dependent cathepsin K and matrix metalloproteinase 1 expression in human fibroblasts. Biochem Biophys Res Commun 430(2):519–522
Pauloin T, Dutot M, Joly F, Warnet JM, Rat P (2009) High molecular weight hyaluronan decreases UVB-induced apoptosis and inflammation in human epithelial corneal cells. Mol Vis 15:577–583
Pauloin T et al (2009) Corneal protection with high-molecular-weight hyaluronan against in vitro and in vivo sodium lauryl sulfate-induced toxic effects. Cornea 28(9):1032–1041
Muto J, Yamasaki K, Taylor KR, Gallo RL (2009) Engagement of CD44 by hyaluronan suppresses TLR4 signaling and the septic response to LPS. Mol Immunol 47(2–3):449–456
Campo GM et al (2010) Molecular size hyaluronan differently modulates toll-like receptor-4 in LPS-induced inflammation in mouse chondrocytes. Biochimie 92(2):204–215
Baeva LF, Lyle DB, Rios M, Langone JJ, Lightfoote MM (2014) Different molecular weight hyaluronic acid effects on human macrophage interleukin 1beta production. J Biomed Mater Res A 102(2):305–314
Lokeshwar VB et al (2010) Antitumor activity of hyaluronic acid synthesis inhibitor 4-methylumbelliferone in prostate cancer cells. Cancer Res 70(7):2613–2623
Kakizaki I et al (2004) A novel mechanism for the inhibition of hyaluronan biosynthesis by 4-methylumbelliferone. J Biol Chem 279(32):33281–33289
Guo N et al (2010) Hyaluronan synthesis mediates the fibrotic response of keratocytes to transforming growth factor beta. J Biol Chem 285(42):32012–32019
El-Asrar AM et al (2011) CXC chemokine expression profiles in aqueous humor of patients with different clinical entities of endogenous uveitis. Immunobiology 216(9):1004–1009
Lapp T et al (2015) Identification of therapeutic targets of inflammatory monocyte recruitment to modulate the allogeneic injury to donor cornea. Invest Ophthalmol Vis Sci 56(12):7250–7259
Takeuchi O et al (1999) Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 11(4):443–451
Jiang D et al (2005) Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med 11(11):1173–1179
Kim MY, Muto J, Gallo RL (2013) Hyaluronic acid oligosaccharides suppress TLR3-dependent cytokine expression in a TLR4-dependent manner. PLoS One 8(8):e72421
McKallip RJ, Hagele HF, Uchakina ON (2013) Treatment with the hyaluronic acid synthesis inhibitor 4-methylumbelliferone suppresses SEB-induced lung inflammation. Toxins 5(10):1814–1826
McKallip RJ, Ban H, Uchakina ON (2015) Treatment with the hyaluronic acid synthesis inhibitor 4-methylumbelliferone suppresses LPS-induced lung inflammation. Inflammation 38(3):1250–1259
Stern R, Asari AA, Sugahara KN (2006) Hyaluronan fragments: an information-rich system. Eur J Cell Biol 85(8):699–715
Campo GM (2009) Glycosaminoglycans reduced inflammatory response by modulating toll-like receptor-4 in LPS-stimulated chondrocytes. Arch Biochem Biophys 491:7–15
Liang J et al (2007) CD44 is a negative regulator of acute pulmonary inflammation and lipopolysaccharide-TLR signaling in mouse macrophages. J Immunol 178(4):2469–2475
Kawana H et al (2008) CD44 suppresses TLR-mediated inflammation. J Immunol 180(6):4235–4245
Boone DL et al (2004) The ubiquitin-modifying enzyme A20 is required for termination of Toll-like receptor responses. Nat Immunol 5(10):1052–1060
Zhang G, Ghosh S (2002) Negative regulation of toll-like receptor-mediated signaling by Tollip. J Biol Chem 277(9):7059–7065
Oliviero F et al (2015) IL-1ß and IL-8 are scavenged by the hexadecylamide derivative of hyaluronic acid: a new mechanism. J Biomed Mater Res A 103(9):2823–2829. doi:10.1002/jbm.a.35422
Mongkhon JM et al (2014) Sorbitol-modified hyaluronic acid reduces oxidative stress, apoptosis and mediators of inflammation and catabolism in human osteoarthritic chondrocytes. Inflamm Res 63(8):691–701
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (81170828), Tianjin Science & Technology Foundation (15JCZDJC35300), Tianjin Health and Family Planning Communication Foundation (14KG133).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
National Natural Science Foundation of China provided financial support in the form of NSFC 81170828. Tianjin Science & Technology Foundation provided financial support in the form of 15JCZDJC35300. Tianjin Health and Family Planning Communication Foundation provided financial support in the form of 14KG133.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
This article does not contain any studies with human participants or animals carried out by any of the authors.
Additional information
Fang Li and Peng Hao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, F., Hao, P., Liu, G. et al. Effects of 4-methylumbelliferone and high molecular weight hyaluronic acid on the inflammation of corneal stromal cells induced by LPS. Graefes Arch Clin Exp Ophthalmol 255, 559–566 (2017). https://doi.org/10.1007/s00417-016-3561-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3561-1