Abstract
Purpose
To study the genotype-phenotype characteristics of Leber congenital amaurosis (LCA) in the Chinese eastern coast Han population.
Methods
Children with strictly defined LCA with novel mutations of known LCA genes identified by targeted next-generation sequencing (NGS) and a prediction of pathogenicity (in silico) were included in this study (2013–2015). Mutations were confirmed using Sanger sequencing and segregation analysis. The clinical findings were recorded, including visual function, refractive error, fundus changes, and electroretinograms (ERGs). Spectral-domain optical coherence tomography (SD-OCT) examination, fundus fluorescein angiography (FFA), and ultra-wide field scanning laser ophthalmoscopy (UWF SLO) were performed on children when available.
Results
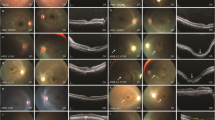
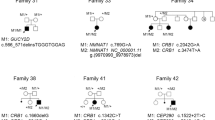
A total of 65 patients underwent NGS for mutation screening and 45 patients were identified as carrying known LCA genes. Of these, 36(80 %) children harbored novel mutations, and they were all from the eastern coast of China. A total of 50 novel variants were identified, which covered 15 known LCA genes. GUCY2D (17 %), CEP290 (14 %), NMNAT1 (14 %), AIPL1 (11 %) and RPGRIP1 (11 %) were the five most frequently mutated genes with novel mutations. A total of four (11 %) patients with AIPL1 mutations harbored the same novel mutated allele (c.C241T p.Q81X), which was homozygous in patients 1 and 2. Unusual manifestations were detected in patient 16 who had novel mutations in CRB1 with a dense proliferative membrane adhering to the posterior retina of the right eye with numerous fine glistening crystals spreading over the retina of both eyes. Ten (40 %) of the 25 available patients who underwent SD-OCT showed a normal macular appearance using fundus photography but an abnormal macular structure using OCT imaging, most of whom presented with a thickened fovea with maldevelopment of the inner and outer retinal laminae.
Conclusions
There may be a high frequency of AIPL1 novel mutations and a founder mutation of p.Q81X in the Chinese eastern coast Han population. Our findings of specific features in this population broaden the spectrum of novel mutations and the phenotype of LCA with ethnic and regional variations. Fundus multimodality imaging may help guide comprehensive assessments for patients with LCA.






Similar content being viewed by others
References
Chacon-Camacho OF (2015) Review and update on the molecular basis of Leber congenital amaurosis. World J Clin Cases 3(2):112
Khan AO, Al-Mesfer S, Al-Turkmani S, Bergmann C, Bolz HJ (2014) Genetic analysis of strictly defined Leber congenital amaurosis with (and without) neurodevelopmental delay. Br J Ophthalmol 98(12):1724–1728
Wang X, Wang H, Sun V et al (2013) Comprehensive molecular diagnosis of 179 Leber congenital amaurosis and juvenile retinitis pigmentosa patients by targeted next-generation sequencing. J Med Genet 50(10):674–688
den Hollander AI, Roepman R, Koenekoop RK, Cremers FPM (2008) Leber congenital amaurosis: Genes, proteins and disease mechanisms. Prog Retin Eye Res 27(4):391–419
Wang H, Wang X, Zou X et al (2015) Comprehensive molecular diagnosis of a large Chinese Leber congenital amaurosis cohort. Invest Ophthalmol Vis Sci 56(6):3642
Chen Y, Zhang Q, Shen T et al (2013) Comprehensive Mutation Analysis by Whole-Exome Sequencing in 41 Chinese Families With Leber Congenital AmaurosisMutation Analysis in Leber Congenital Amaurosis. Invest Ophthalmol Vis Sci 54(6):4351–4357
Li L, Xiao X, Li S et al (2011) Detection of Variants in 15 Genes in 87 Unrelated Chinese Patients with Leber Congenital Amaurosis. PLoS ONE 6(5), e19458
Chung DC, Traboulsi EI (2009) Leber congenital amaurosis: clinical correlations with genotypes, gene therapy trials update, and future directions. J AAPOS 13(6):587–592
Marmor MF, Fulton AB, Holder GE et al (2009) ISCEV Standard for full-field clinical electroretinography (2008 update). Doc Ophthalmol 118(1):69–77
Ge Z, Bowles K, Goetz K et al (2015) NGS-based molecular diagnosis of 105 eyeGENE® probands with retinitis pigmentosa. Sci Rep 5:18287
Perrault I, Delphin N, Hanein S et al (2007) Spectrum of NPHP6/CEP290 mutations in Leber congenital amaurosis and delineation of the associated phenotype. Hum Mutat 28(4):416
Li L, Xiao X, Li S et al (2011) Detection of Variants in 15 Genes in 87 Unrelated Chinese Patients with Leber Congenital Amaurosis. Toland AE, ed. PLoS ONE 6(5):e19458
Valente EM, Silhavy JL, Brancati F et al (2006) Mutations in CEP290, which encodes a centrosomal protein, cause pleiotropic forms of Joubert syndrome. Nat Genet 38(6):623–625
Galvin JA, Fishman GA, Stone EM, Koenekoop RK (2005) Clinical phenotypes in carriers of Leber congenital amaurosis mutations. Ophthalmology 112(2):349–356
Peshenko IV, Olshevskaya EV, Yao S, Ezzeldin HH, Pittler SJ, Dizhoor AM (2010) Activation of retinal guanylyl cyclase RetGC1 by GCAP1: stoichiometry of binding and effect of new LCA-related mutations. Biochemistry (Mosc) 49(4):709–717
Stone EM (2007) Leber Congenital Amaurosis–A Model for Efficient Genetic Testing of Heterogeneous Disorders: LXIV Edward Jackson Memorial Lecture. Am J Ophthalmol 144(6):791–811.e6
Perrault I, Hanein S, Zanlonghi X et al (2012) Mutations in NMNAT1 cause Leber congenital amaurosis with early-onset severe macular and optic atrophy. Nat Genet 44(9):975–977
Wang H, den Hollander AI, Moayedi Y et al (2009) Mutations in SPATA7 Cause Leber Congenital Amaurosis and Juvenile Retinitis Pigmentosa. Am J Hum Genet 84(3):380–387
Neveling K, Feenstra I, Gilissen C et al (2013) A post-hoc comparison of the utility of Sanger sequencing and exome sequencing for the diagnosis of heterogeneous diseases. Hum Mutat 34(12):1721–1726
Morimura H, Fishman GA, Grover SA, Fulton AB, Berson EL, Dryja TP (1998) Mutations in the RPE65 gene in patients with autosomal recessive retinitis pigmentosa or Leber congenital amaurosis. Proc Natl Acad Sci 95(6):3088–3093
Fu Q, Wang F, Wang H et al (2013) Next-generation sequencing-based molecular diagnosis of a Chinese patient cohort with autosomal recessive retinitis pigmentosa. Invest Ophthalmol Vis Sci 54(6):4158–4166
Zou X, Yao F, Liang X et al (2015) De novo mutations in the cone-rod homeobox gene associated with Leber congenital amaurosis in Chinese patients. Ophthalmic Genet 36(1):21–6
Xu F, Dong Q, Liu L et al (2012) Novel RPE65 mutations associated with Leber congenital amaurosis in Chinese patients. Mol Vis 18:744–50
Pennesi ME, Stover NB, Stone EM, Chiang P-W, Weleber RG (2011) Residual Electroretinograms in Young Leber Congenital Amaurosis Patients with Mutations of AIPL1. Invest Ophthalmol Vis Sci 52(11):8166
Jacobson SG, Cideciyan AV, Aleman TS et al (2011) Human retinal disease from AIPL1 gene mutations: foveal cone loss with minimal macular photoreceptors and rod function remaining. Invest Ophthalmol Vis Sci 52(1):70–79
Tan MH, Mackay DS, Cowing J et al (2012) Leber Congenital Amaurosis Associated with AIPL1: Challenges in Ascribing Disease Causation, Clinical Findings, and Implications for Gene Therapy. PLoS ONE 7(3), e32330
Yzer S1, Hollander AI, Lopez I et al (2012) Ocular and extra-ocular features of patients with Leber congenital amaurosis and mutations in CEP290. Mol Vis 18:412–25
Heegaard S, Rosenberg T, Preising M, Prause JU, Bek T (2003) An unusual retinal vascular morphology in connection with a novel AIPL1 mutation in Leber’s congenital amaurosis. Br J Ophthalmol 87(8):980–983
den Hollander AI, Heckenlively JR, van den Born LI et al (2001) Leber congenital amaurosis and retinitis pigmentosa with Coats-like exudative vasculopathy are associated with mutations in the crumbs homologue 1 (CRB1) gene. Am J Hum Genet 69(1):198–203
Bujakowska K, Audo I, Mohand-Saïd S et al (2012) CRB1 mutations in inherited retinal dystrophies. Hum Mutat 33(2):306–315
Tsang SH, Burke T, Oll M et al (2014) Whole Exome Sequencing Identifies CRB1 Defect in an Unusual Maculopathy Phenotype. Ophthalmology 121(9):1773–1782
Jacobson SG, Cideciyan AV, Huang WC et al (2016) Leber Congenital Amaurosis: Genotypes and Retinal Structure Phenotypes. Adv Exp Med Biol 854:169–175
Acknowledgments
We sincerely thank all the patients and their families for their participation. We thank Hui Wang from Baylor College of Medicine for her help on the molecular study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Shanghai Health Bureau provided financial support in the form of “Molecular Genetics Study of Leber Congenital Amaurosis by Whole-exome Sequencing in Chinese People” Funding(XBR20111060). The sponsor had no role in the design or conduct of this research.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of Xinhua Hospital and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
All authors declare that they have no conflict of interests to state. All authors have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(XLSX 224 kb)
Rights and permissions
About this article
Cite this article
Wang, S., Zhang, Q., Zhang, X. et al. Clinical and genetic characteristics of Leber congenital amaurosis with novel mutations in known genes based on a Chinese eastern coast Han population. Graefes Arch Clin Exp Ophthalmol 254, 2227–2238 (2016). https://doi.org/10.1007/s00417-016-3428-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3428-5