Abstract
Purpose
Blue–yellow short wavelength testing (BY-VEPs) has proven diagnostic relevance in detecting early ganglion cell damage, e.g., in glaucoma. To date testing has generally been conducted using individual protocols without consideration of the lens status. In this study, we compared changes in BY-VEPs and standard pattern VEPs in phakic and pseudophakic glaucoma patients and controls.
Methods
The eyes of 57 healthy controls (18 pseudophakic and 39 phakic) and 67 glaucoma patients (29 pseudophakic and 38 phakic) were included in a prospective study. Phakic eyes were arranged in three groups according to the Lens Opacities Classification System III. Transient on/off isoluminant blue–yellow 2° checks were used for BY-VEPs, transient large 1° (M1) and small 0.25° (M2) black–white checks for standard pattern reversal VEPs, according to the ISCEV standards.
Results
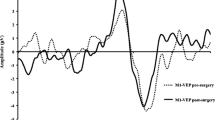
Latencies and amplitudes of M1 and M2 did not differ significantly between groups or lens status. ANOVA analysis revealed significantly longer BY-VEP latencies in glaucoma compared to controls (p = 0.002), independently of the lens status. The amplitudes showed no such pattern (p = 0.93).
Mean defect (MD) was significantly negatively correlated to BY-VEP latency (r = −0.54, p = 0.003) only in pseudophakic glaucoma patients. Different stages of cataract did not show a significant effect on the BY-VEP latencies.
Conclusions
Glaucoma led to a significant increase of BY-VEPs latencies, while standard pattern VEPs were not influenced. The correlation of MD and BY-VEP latency only in pseudophakic glaucoma patients indicates a substantial confounding effect of lens opacifications on the diagnostic value of BY-VEPs in glaucoma.



Similar content being viewed by others
References
Odom JV, Bach M, Brigell M, Holder GE, McCulloch DL, Tormene AP, Vaegan (2010) ISCEV standard for clinical visual evoked potentials (2009 update). Doc Ophthalmol 120(1):111–119. doi:10.1007/s10633-009-9195-4
Regan D (1989) Human brain electrophysiology: evoked potentials and evoked magnetic fields in science and medicine, vol xxiv. Elsevier, New York, 672 p
Lennie P, D’Zmura M (1988) Mechanisms of color vision. Crit Rev Neurobiol 3(4):333–400
Yucel YH, Zhang Q, Weinreb RN, Kaufman PL, Gupta N (2003) Effects of retinal ganglion cell loss on magno-, parvo-, koniocellular pathways in the lateral geniculate nucleus and visual cortex in glaucoma. Prog Retin Eye Res 22(4):465–481
Crognale MA, Switkes E, Rabin J, Schneck ME, Haegerstrom-Portnoy G, Adams AJ (1993) Application of the spatiochromatic visual evoked potential to detection of congenital and acquired color-vision deficiencies. J Opt Soc Am A Opt Image Sci Vis 10(8):1818–1825
Schneck M, Lee E, Fortune B, Crognale M, Adams A, Switkes E (1997) Spatio-chromatic VEPS in recovered optic neuritis and multiple sclerosis. In: Cavonius CR (ed) Colour vision deficiencies XIII, vol 59. Documenta Ophthalmologica Proceedings Series. Springer, Netherlands, pp 179–185. doi:10.1007/978-94-011-5408-6_18
Crognale M, Rabin J, Switkes E, Adams AJ (1993) Selective loss of S-pathway sensitivity in central serous choroidopathy revealed by spatio-chromatic visual evoked cortical potentials (VECP). In: Drum B (ed) Colour vision deficiencies XI, vol 56. Documenta Ophthalmologica Proceedings Series. Springer, Netherlands, pp 229–239. doi:10.1007/978-94-011-1856-9_23
Johnson CA, Adams AJ, Casson EJ, Brandt JD (1993) Blue-on-yellow perimetry can predict the development of glaucomatous visual field loss. Arch Ophthalmol 111(5):645–650
Horn FK, Jonas JB, Budde WM, Junemann AM, Mardin CY, Korth M (2002) Monitoring glaucoma progression with visual evoked potentials of the blue-sensitive pathway. Invest Ophthalmol Vis Sci 43(6):1828–1834
Copenhaver RM, Perry NW Jr (1964) Factors affecting visually evoked cortical potentials such as impaired vision of varying etiology. Invest Ophthalmol 3:665–675
Crognale MA (2002) Development, maturation, and aging of chromatic visual pathways: VEP results. J Vis 2(6):438–450. doi:10.1167/2.6.2
Fuest M, Plange N, Jamali S, Schwarzer H, Roessler G, Walter P, Mazinani B (2014) The effect of cataract surgery on blue–yellow and standard-pattern visual-evoked potentials. Graefe’s Arch Clin Exp Ophthalmol 252(11):1831–1837. doi:10.1007/s00417-014-2728-x
European Glaucoma Society [EGS] (2008) Terminology and guidelines for glaucoma, 3rd edn. European Glaucoma Society, Savona Italy
Chylack LT Jr, Wolfe JK, Singer DM, Leske MC, Bullimore MA, Bailey IL, Friend J, McCarthy D, Wu SY (1993) The lens opacities classification system III. The longitudinal study of cataract study group. Arch Ophthalmol 111(6):831–836
Epstein CM, Bej MD, Foldvary-Schaefer N, Lagerlund TD, Helmers SL, Husain AM (2006) Guideline 5: guidelines for standard electrode position nomenclature (American Clinical Neurophysiology Society). J Clin Neurophysiol 23(2):107–110
Porciatti V, Di Bartolo E, Nardi N, Fiorentini A (1997) Responses to chromatic and luminance contrast in glaucoma: a psychophysical and electrophysiological study. Vis Res 37(14):1975–1987
Nguyen XN, Korth M, Rix R, Sembritzki O (1993) [Blue-sensitive mechanisms in pattern ERG and VEP]. Ophthalmologe 90(2):143–147 (article in German)
Musch DC, Gillespie BW, Niziol LM, Janz NK, Wren PA, Rockwood EJ, Lichter PR (2006) Cataract extraction in the collaborative initial glaucoma treatment study: incidence, risk factors, and the effect of cataract progression and extraction on clinical and quality-of-life outcomes. Arch Ophthalmol 124(12):1694–1700. doi:10.1001/archopht.124.12.1694
van Lith GHM, Hekkert-Wiebenga W (1983) Cataract, pattern stimulation and visually evoked potentials. Doc Ophthalmol 55(1–2):107–112. doi:10.1007/bf00140467
Chelva E, van Lith GHM (1982) Influence of defocusing, absorption and scatter on evoked potentials to contrast and contour related stimuli. Doc Ophthalmol 31:337–344
Ho WC, Chu PH, Ng YF, Tong PP, Woo VC, Chan HH (2012) Temporal interactive response is resistant to cloudy ocular media in the slow double-stimulation multifocal electroretinogram. Br J Ophthalmol 96(7):1012–1017. doi:10.1136/bjophthalmol-2011-301099
Kubova Z, Kuba M, Spekreijse H, Blakemore C (1995) Contrast dependence of motion-onset and pattern-reversal evoked potentials. Vision Res 35(2):197–205
Sawaya RA, Haddad RS (2002) Effect of cataract on pattern reversal visual evoked potential. Ann Ophthalmol 34(4):190–193. doi:10.1007/s12009-002-0074-9
Ermers HJM, De Heer LJ, Van Lith GHM (1974) VECPS in patients with glaucoma. In: Dodt E, Pearlman J (eds) XIth I.S.C.E.R.G. Symposium, vol 4. Documenta Ophthalmologica Proceedings Series. Springer, Netherlands, pp 387–393. doi:10.1007/978-94-010-2340-5_45
Wildberger HG, Van Lith GH, Wijngaarde R, Mak GT (1976) Visually evoked cortical potentials in the evaluation of homonymous and bitemporal visual field defects. Br J Ophthalmol 60(4):273–278
Bartl G (1978) The electroretinogram and the visual evoked potential in normal and glaucomatous eyes (author’s transl). Graefes Arch Clin Exp Ophthalmol 207(4):243–269
Korth M, Nguyen NX, Junemann A, Martus P, Jonas JB (1994) VEP test of the blue-sensitive pathway in glaucoma. Invest Ophthalmol Vis Sci 35(5):2599–2610
Adams AJ, Rodic R, Husted R, Stamper R (1982) Spectral sensitivity and color discrimination changes in glaucoma and glaucoma-suspect patients. Invest Ophthalmol Vis Sci 23(4):516–524
Horn FK, Michelson G, Schnitzler E, Mardin CY, Korth M, Junemann AG (2006) Visual evoked potentials of the blue-sensitive pathway under cold provocation in normals and glaucomas. J Glaucoma 15(1):17–22
Rabin J, Switkes E, Crognale M, Schneck ME, Adams AJ (1994) Visual evoked potentials in three-dimensional color space: correlates of spatio-chromatic processing. Vis Res 34(20):2657–2671
Bron AJ, Vrensen GF, Koretz J, Maraini G, Harding JJ (2000) The ageing lens. Ophthalmologica 214(1):86–104
Anstis SM, Cavanagh P (1983) A minimum motion technique for judging equiluminance. In: Sharpe JDMLT (ed) Colour vision: psychophysics and physiology. Academic, London, pp 66–77
Berninger TA, Arden GB, Hogg CR, Frumkes T (1989) Separable evoked retinal and cortical potentials from each major visual pathway: preliminary results. Br J Ophthalmol 73(7):502–511
Porciatti V, Sartucci F (1999) Normative data for onset VEPs to red-green and blue-yellow chromatic contrast. Clin Neurophysiol 110(4):772–781
Stifter E, Sacu S, Thaler A, Weghaupt H (2006) Contrast acuity in cataracts of different morphology and association to self-reported visual function. Invest Ophthalmol Vis Sci 47(12):5412–5422. doi:10.1167/iovs.05-1564
Hess R, Woo G (1978) Vision through cataracts. Invest Ophthalmol Vis Sci 17(5):428–435
Acknowledgments
Ethics approval
Institutional review board, University Hospital RWTH Aachen
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Competing interests
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fuest, M., Kieckhoefel, J., Mazinani, B. et al. Blue–yellow and standard pattern visual evoked potentials in phakic and pseudophakic glaucoma patients and controls. Graefes Arch Clin Exp Ophthalmol 253, 2255–2261 (2015). https://doi.org/10.1007/s00417-015-3152-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3152-6