Abstract
Background
To report functional and high-resolution retinal imaging abnormalities, including adaptive optics (AO) throughout the course of acute macular neuroretinopathy (AMNR).
Methods
Two female patients (four eyes) with a diagnosis of AMNR were observed at the Clinical Investigation Center, CHNO des Quinze-Vingts, Paris, France. The patients underwent detailed ophthalmic examination including best-corrected visual acuity, slit-lamp examination, kinetic and static perimetry, full-field and multifocal electroretinogram, infrared reflectance, autofluorescence imaging and spectral-domain optical coherence tomography (SD-OCT) and AO fundus imaging at presentation and during follow-up.
Results
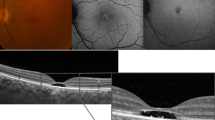
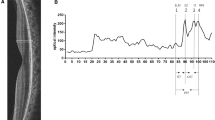
Both cases showed concomitant loss of integrity of the outer retinal structures on SD-OCT, and marked abnormalities on AO imaging with disruption of the visibility of the cone mosaic. In the first case, photoreceptor damage was seen to progress during several weeks before healing. In both cases, there were persistent morphological abnormalities of photoreceptors 1 year after onset.
Conclusion
This study further highlights the value of AO fundus imaging to facilitate detection, mapping, and monitoring of damage to the cone outer segments during AMNR. In particular, residual damage to the cone mosaic can be precisely documented.







Similar content being viewed by others
References
Bos PJ, Deutman AF (1975) Acute macular neuroretinopathy. Am J Ophthalmol 80(4):573–584
Turbeville SD, Cowan LD, Gass JD (2003) Acute macular neuroretinopathy: a review of the literature. Surv Ophthalmol 48(1):1–11
Amin P, Cox TA (1998) Acute macular neuroretinopathy. Arch Ophthalmol 116(1):112–113
Gandorfer A, Ulbig MW (2002) Scanning laser ophthalmoscope findings in acute macular neuroretinopathy. Am J Ophthalmol 133(3):413–415
Hughes EH, Siow YC, Hunyor AP (2009) Acute macular neuroretinopathy: anatomic localisation of the lesion with high-resolution OCT. Eye 23(11):2132–2134
Gomez-Torreiro M, Gomez-Ulla F, Bolivar Montesa P, Rodriguez-Cid MJ (2002) Scanning laser opthalmoscope findings in acute macular neuroretinopathy. Retina 22(1):108–109
Neuhann IM, Inhoffen W, Koerner S, Bartz-Schmidt KU, Gelisken F (2010) Visualization and follow-up of acute macular neuroretinopathy with the spectralis HRA+OCT device. Graefes Arch Clin Exp Ophthalmol 248(7):1041–1044
Kuznik-Borkowska A, Cohen SY, Broido-Hooreman O, Gaudric A (2006) Acute macular neuroretinopathy. J Fr Ophtalmol 29(3):319–322
Mirshahi A, Scharioth GB, Klais CM, Baatz H (2006) Enhanced visualization of acute macular neuroretinopathy by Heidelberg retina tomography. Clin Exp Ophthalmol 34(6):596–599
Chan WM, Liu DT, Tong JP, Law RW, Lam DS (2005) Longitudinal findings of acute macular neuroretinopathy with multifocal electroretinogram and optical coherence tomography. Clin Exp Ophthalmol 33(4):439–442
Monson BK, Greenberg PB, Greenberg E, Fujimoto JG, Srinivasan VJ, Duker JS (2007) High-speed, ultra-high-resolution optical coherence tomography of acute macular neuroretinopathy. Br J Ophthalmol 91(1):119–120
Maschi C, Schneider-Lise B, Paoli V, Gastaud P (2011) Acute macular neuroretinopathy: contribution of spectral-domain optical coherence tomography and multifocal ERG. Graefes Arch Clin Exp Ophthalmol 249(6):827–831
Vance SK, Spaide RF, Freund KB, Wiznia R, Cooney MJ (2011) Outer retinal abnormalities in acute macular neuroretinopathy. Retina 31(3):441–445
Yeh S, Hwang TS, Weleber RG, Watzke RC, Francis PJ (2011) Acute macular outer retinopathy (AMOR): a reappraisal of acute macular neuroretinopathy using multimodality diagnostic testing. Arch Ophthalmol 129(3):365–368
Baumuller S, Holz FG (2012) Early spectral-domain optical coherence tomography findings in acute macular neuroretinopathy. Retina 32(2):409–410
Azar G, Wolff B, Cornut PL, Sahel JA, Mauget-Faysse M (2012) Spectral domain optical coherence tomography evolutive features in acute macular neuroretinopathy. Eur J Ophthalmol 22(5):850–852
Sieving PA, Fishman GA, Salzano T, Rabb MF (1984) Acute macular neuroretinopathy: early receptor potential change suggests photoreceptor pathology. Br J Ophthalmol 68(4):229–234
Browning AC, Gupta R, Barber C, Lim CS, Amoaku WM (2003) The multifocal electroretinogram in acute macular neuroretinopathy. Arch Ophthalmol 121(10):1506–1507
Corver HD, Ruys J, Kestelyn-Stevens AM, De Laey JJ, Leroy BP (2007) Two cases of acute macular neuroretinopathy. Eye 21(9):1226–1229
Maturi RK, Yu M, Sprunger DT (2003) Multifocal electroretinographic evaluation of acute macular neuroretinopathy. Arch Ophthalmol 121(7):1068–1069
Munch IC, Traustason S, Olgaard K, Larsen M (2012) Acute macular neuroretinopathy in relation to anti-thymocyte globulin infusion. Acta Ophthalmol 90(4):e321–e322
Miller MH, Spalton DJ, Fitzke FW, Bird AC (1989) Acute macular neuroretinopathy. Ophthalmology 96(2):265–269
O'Brien DM, Farmer SG, Kalina RE, Leon JA (1989) Acute macular neuroretinopathy following intravenous sympathomimetics. Retina 9(4):281–286
Desai UR, Sudhamathi K, Natarajan S (1993) Intravenous epinephrine and acute macular neuroretinopathy. Arch Ophthalmol 111(8):1026–1027
Leys M, Van Slycken S, Koller J, Van de Sompel W (1991) Acute macular neuroretinopathy after shock. Bull Soc Belge Ophtalmol 241:95–104
Stilma JS, de Lange JJ, Crezee FC (1987) Bilateral central scotoma with preservation of central vision in 2 patients following caesarean section under spinal anesthesia. Doc Ophthalmol Adv Ophthalmol 67(1–2):59–68
Guzak SV, Kalina RE, Chenoweth RG (1983) Acute macular neuroretinopathy following adverse reaction to intravenous contrast media. Retina 3(4):312–317
Sarraf D, Rahimy E, Fawzi AA, Sohn E, Barbazetto I, Zacks DN, Mittra RA, Klancnik JM Jr, Mrejen S, Goldberg NR, Beardsley R, Sorenson JA, Freund KB (2013) Paracentral acute middle maculopathy: a new variant of acute macular neuroretinopathy associated with retinal capillary ischemia. JAMA Ophthalmol 131(10):1275–1287
Hirooka K, Saito W, Noda K, Ishida S (2014) Enhanced-depth imaging optical coherence tomography and laser speckle flowgraphy in a patient with acute macular neuroretinopathy. Ocul Immunol Inflamm 22(6):485–489
Liang J, Williams DR, Miller DT (1997) Supernormal vision and high-resolution retinal imaging through adaptive optics. J Opt Soc Am A Opt Image Sci Vis 14(11):2884–2892
Pallikaris A, Williams DR, Hofer H (2003) The reflectance of single cones in the living human eye. Invest Ophthalmol Vis Sci 44(10):4580–4592
Roorda A (2000) Adaptive optics ophthalmoscopy. J Refract Surg 16(5):S602–S607
Chui TY, Song H, Burns SA (2008) Adaptive-optics imaging of human cone photoreceptor distribution. J Opt Soc Am A Opt Image Sci Vis 25(12):3021–3029
Hood DC, Bach M, Brigell M, Keating D, Kondo M, Lyons JS, Palmowski-Wolfe AM (2008) ISCEV guidelines for clinical multifocal electroretinography (2007 edition). Doc Ophthalmol Adv Ophthalmol 116(1):1–11
Loquin KBI, Nakashima K, Rossant F, Boelle P-Y, Paques M (2011) Photoreceptor detection in in-vivo adaptive optics images of the retina: towards a simple interactive tool for the physicians. IEEE International Symposium on Biomedical Imaging (ISBI), Chicago, pp 191–194
Hansen SO, Cooper RF, Dubra A, Carroll J, Weinberg DV (2013) Selective cone photoreceptor injury in acute macular neuroretinopathy. Retina 33(8):1650–1658
Mrejen S, Pang CE, Sarraf D, Goldberg NR, Gallego-Pinazo R, Klancnik JM, Sorenson JA, Yannuzzi LA, Freund KB (2014) Adaptive optics imaging of cone mosaic abnormalities in acute macular neuroretinopathy. Ophthalmic Surg Lasers Imaging Retina 45(6):562–569
Fawzi AA, Pappuru RR, Sarraf D, Le PP, McCannel CA, Sobrin L, Goldstein DA, Honowitz S, Walsh AC, Sadda SR, Jampol LM, Eliott D (2012) Acute macular neuroretinopathy: long-term insights revealed by multimodal imaging. Retina 32(8):1500–1513
Feigl B, Haas A (2000) Optical coherence tomography (OCT) in acute macular neuroretinopathy. Acta Ophthalmol Scand 78(6):714–716
Gass JD, Hamed LM (1989) Acute macular neuroretinopathy and multiple evanescent white dot syndrome occurring in the same patients. Arch Ophthalmol 107(2):189–193
Acknowledgments
The authors are grateful to patients reported in this study, to the staff from Imagine Eyes for their technical support, and to the orthoptists, nurses, and study coordinators from the CIC503, as well as to the librarian of Quinze-Vingts hospital for her assistance with reference articles.
None of the co-authors have a commercial relationship in relation associated with the data presented in this study.
The project was financially supported by the ANR TecSan 09–09 iPhot and Foundation Fighting Blindness (FFB FFB center grant C-GE-0912-0601-INSERM02), and by IA FFB grant N°: CD-CL-0808-0466-CHNO).
Conflict of interest statement
All authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Isabelle Audo and Kiyoko Gocho contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary figure
Outer nuclear layer mapping for case 1 at presentation and 9 months and case 2 at 9 and 18 months after onset revealed ONL thinning. (PDF 1592 kb)
Supplementary material
video showing close-up follow-up adaptive optics images of Fig. 4 from the left eye for Case 1, taken from the 2° nasal 2° superior from the foveal center, shows similar pattern of abnormalities as for the right eye, i.e., sharp hyporeflective lesion at presentation, increased in lesion size then decrease with persistent abnormalities at M12 and patchy hyper reflective dots. Scale bar shows 100 μm. (GIF 2924 kb)
Rights and permissions
About this article
Cite this article
Audo, I., Gocho, K., Rossant, F. et al. Functional and high-resolution retinal imaging monitoring photoreceptor damage in acute macular neuroretinopathy. Graefes Arch Clin Exp Ophthalmol 254, 855–864 (2016). https://doi.org/10.1007/s00417-015-3136-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3136-6