Abstract
Background
Corneal graft rejection is the major cause of corneal failures. Previous studies have shown that the CD25 monoclonal antibody can inhibit corneal graft rejection during the acute phase of rejection in rat models. In the current study, we evaluated the safety and efficacy of the topical Entranster™ vector in rat corneal applications and further investigated the effects of CD25siRNA gene transfer on high-risk rat corneal graft rejection.
Methods
Fluorescence detection, clinical assessment, hematoxylin and eosin (HE), terminal deoxynucleotidyl transferase biotin-dUTP nick end labelling (TUNEL) and CD11b assays were used to evaluate the safety and efficacy of CD25siRNA gene transfer in normal SD rat corneas. Orthotopic corneal transplants were performed in alkali burned SD rats. Corneal recipients were divided into four groups that received different treatments. Clinical assessment, western blotting, quantitative real-time polymerase chain reaction (qRT-PCR), immunohistochemistry, HE and transmission electron microscopy (TEM) were performed on all grafts.
Results
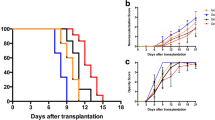
Low toxicity, no immunogenicity and high transfection efficiency were observed in rat corneas treated with the Entranster™ vector. Reduced endothelial cell apoptosis, inflammatory cell infiltration and graft neovascularisation were observed in the CD25siRNA treatment groups. The graft survival curves showed that CD25siRNA treatment significantly prolonged graft survival time, with better graft transparency and less graft oedema. Lower CD25 and higher IL-10 expression were detected in the CD25siRNA treatment groups during the study period, and a higher FOXP3 level was found in the CD25siRNA group than in the CD25siRNA-twice group on days 14 and 21 post-operation.
Conclusions
The Entranster™ vector is an effective vector for corneal gene therapy. CD25 siRNA gene transfer inhibits corneal graft rejection via upregulation of anti-inflammatory molecule expression.







Similar content being viewed by others
References
Yuan XB, Yuan YB, Jiang W, Liu J, Tian EJ, Shun HM, Huang DH, Yuan XY, Li H, Sheng J (2008) Preparation of rapamycin-loaded chitosan/PLA nanoparticles for immunosuppression in corneal transplantation. Int J Pharm 349:241–248
Coster DJ, Williams KA (2005) The impact of corneal allograft rejection on the long-term outcome of corneal transplantation. Am J Opthalmol 140:1112–1122
Vail A, Gore SM, Bradley BA, Easty DL, Rogers CA, Armitage WJ (1997) Conclusions of the corneal transplant follow up study. Collaborating surgeons. Br J Ophthalmol 81:631–636
Williams KA, Esterman AJ, Bartlett C, Holland H, Hornsby NB, Coster DJ (2006) How effective is penetrating corneal transplantation? Factors influencing long-term outcome in multivariate analysis. Transplantation 81:896–901
Xiao H, Zhang H, Yu ZY, Zhang L, Yu M, Huang YF, Li S, Shen BF, Li Y (2007) J2 prolongs the corneal allograft survival through inhibition of the CD4 + T cell-mediated response in vivo. Transpl Immunol 18:130–137
Jia Z, Jiao C, Zhao S, Li X, Ren X, Zhang L, Han ZC, Zhang X (2012) Immunomodulatory effects of mesenchymal stem cells in a rat corneal allograft rejection model. Exp Eye Res 102:44–49
Holland EJ, Chan CC, Wetzig RP, Palestine AG, Nussenblatt RB (1991) Clinical and immunohistologic studies of corneal rejection in the rat penetrating keratoplasty model. Cornea 10:374–380
Létourneau S, Krieg C, Pantaleo G, Boyman O (2009) IL-2- and CD25-dependent immunoregulatory mechanisms in the homeostasis of T-cell subsets. J Allergy Clin Immunol 123:758–762
Nashan B, Schwinzer R, Schlitt HJ, Wonigeit K, Pichlmayr R (1995) Immunological effects of the anti-lL-2 receptor monoclonal antibody BT 563 in liver allografted patients. Transpl Immunol 3:203–211
Cheikhelard A, Go S, Canioni D, Leborgne M, Brousse N, Révillon Y, Cerf-Bensussan N, Sarnacki S (2005) Enhanced in situ expression of NF-kappaBp65 is an early marker of intestinal graft rejection in rats. J Pediatr Surg 40:1420–1427
Jirasiritham S, Sumethkul V, Mavichak V, Lertsithichai P, Jirasiritham S (2004) The role of anti-IL-2 receptor in high-risk kidney transplant patients. Transplantation 36:2110–2112
Saengkrit N, Sanitrum P, Woramongkolchai N, Saesoo S, Pimpha N, Chaleawlert-Umpon S, Tencomnao T, Puttipipatkhachorn S (2012) The PEI-introduced CS shell/PMMA core nanoparticle for silencing the expression of E6/E7 oncogenes in human cervical cells. Carbohydr Polym 90:1323–1329
Huh MS, Lee SY, Park S, Lee S, Chung H, Lee S, Choi Y, Oh YK, Park JH, Jeong SY, Choi K, Kim K, Kwon IC (2010) Tumor-homing glycol chitosan/polyethylenimine nanoparticles for the systemic delivery of siRNA in tumor-bearing mice. J Control Release 144:134–143
Fattal E, Bochot A (2006) Ocular delivery of nucleic acids: antisense oligonucleotides, aptamers and siRNA. Adv Drug Deliv Rev 58:1203–1223
Davis ME, Zuckerman JE, Choi CH, Seligson D, Tolcher A, Alabi CA, Yen Y, Heidel JD, Ribas A (2010) Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles. Nature 464:1067–1070
Sharma A, Tandon A, Tovey JC, Gupta R, Robertson JD, Fortune JA, Klibanov AM, Cowden JW, Rieger FG, Mohan RR (2011) Polyethylenimine-conjugated gold nanoparticles: gene transfer potential and low toxicity in the cornea. Nanomedicine 7:505–513
Jiang Z, Chen CH, Chen YY, Han JY, Riley J, Zhou CM (2014) Autophagic effect of programmed cell death 5 (PDCD5) after focal cerebral ischemic reperfusion injury in rats. Neurosci Lett 566:298–303
He F, Yao H, Xiao Z, Han J, Zou J, Liu Z (2014) Inhibition of IL-2 inducible T-cell kinase alleviates T-cell activation and murine myocardial inflammation associated with CVB3 infection. Mol Immunol 59:30–38
Zhang Y, Zhang L, Wu J, Di C, Xia Z (2013) Heme oxygenase-1 exerts a protective role in ovalbumin-induced neutrophilic airway inflammation by inhibiting Th17 cell-mediated immune response. J Biol Chem 288:34612–34626
Liu J, Li G, Peng H, Tu G, Kong F, Liu S, Gao Y, Xu H, Qiu S, Fan B, Zhu Q, Yu S, Zheng C, Wu B, Peng L, Song M, Wu Q, Peng L, Song M, Wu Q, Li G, Liang S (2013) Sensory-sympathetic coupling in superior cervical ganglia after myocardial ischemic injury facilitates sympathoexcitatory action via P2X7 receptor. Purinergic Signal 9:463–479
Jie Y, Pan Z, Chen Y, Wei Y, Zhang W, Xu L, Wu Y, Peng H (2004) SEB combined with IL-1ra could prolong the survival of the rat allografts in high-risk corneal transplantation. Transplant Proc 36:3267–3271
Monaghan M, Pandit A (2011) RNA interference therapy via functionalized scaffolds. Adv Drug Deliv Rev 63:197–208
Bartlett DW, Davis ME (2006) Insights into the kinetics of siRNA-mediated gene silencing from live-cell and live-animal bioluminescent imaging. Nucleic Acids Res 34:322–333
Armitage WJ, Easty DL (1997) Factors influencing the suitability of organ-cultured corneas for transplantation. Invest Ophthalmol Vis Sci 38:16–24
Hu J, Kovtun A, Tomaszewski A, Singer BB, Seitz B, Epple M, Steuhl KP, Ergun S, Fuchsluger TA (2012) A new tool for the transfection of corneal endothelial cells: calcium phosphate nanoparticles. Acta Biomater 8:1156–1163
Niederkorn JY (1999) The immune privilege of corneal allografts. Transplantation 67:1503–1508
Hsu CC, Chang HM, Lin TC, Hung KH, Chien KH, Chen SY, Chen SN, Chen YT (2015) Corneal neovascularization and contemporary antiangiogenic therapeutics. J Chin Med Assoc. doi:10.1016/j.jcma.2014.10.002
Cheung KW, Sze DM, Chan WK, Deng RX, Tu W, Chan GC (2011) Brazilian green propolis and its constituent, Artepillin C inhibits allogeneic activated human CD4 T cells expansion and activation. J Ethnopharmacol 138:463–471
Hire K, Ngo DK, Stewart-Maynard KM, Hering B, Bansal-Pakala P (2012) FoxP3+, and not CD25+, T cells increase post-transplant in islet allotransplant recipients following anti-CD25+ rATG immunotherapy. Cell Immunol 274:83–88
Acknowledgments
This study was supported by funding from the National Natural Science Foundation of China (81170822).
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, Q., Shi, Y., Zhao, Q. et al. Effects of CD25siRNA gene transfer on high-risk rat corneal graft rejection. Graefes Arch Clin Exp Ophthalmol 253, 1765–1776 (2015). https://doi.org/10.1007/s00417-015-3067-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3067-2