Abstract
Purpose
To characterise the changes of the retinal layers in patients with acute anterior ischaemic optic neuropathy (AION), aiming to identify imaging markers for predicting the residual visual function.
Methods
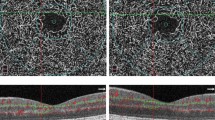
This was a retrospective review of consecutive patients with unilateral AION from January 2010 to December 2013. We analysed affected eyes at baseline and 1 month later, compared to fellow healthy eyes. Utilising novel image analysis software, we conducted algorithmic segmentation in layers and division in early treatment of diabetic retinopathy study (ETDRS) quadrants of optical coherence tomography images of the macula. Pearson product moment regression analysis of retinal layer thickness and best corrected visual acuity (BCVA) in logMAR units and mean deviation of the SITA 24–2 visual field (VF) were carried out at the 1-month time point.
Results
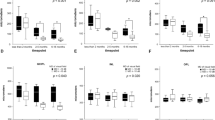
Twenty eyes from 20 patients were included and compared to 20 healthy fellow eyes. At baseline, we found a significantly increased mean thickness of the retinal nerve fibre layer (RNFL) of 42.2 μm (±6.7SD) in AION eyes compared to 37.9 μm (±4.2 SD) in healthy eyes (p = 0.002). The outer nuclear layer (ONL) was also significantly thickened at 96.6 μm (±7.2 SD) compared to 90.8 μm (±5.7 SD) in the fellow eye (p < 0.001). After 1 month, the RNFL and the ganglion cell layer (GCL) were thinned 17.7 % [to 31.2 μm (±6.4 SD), p < 0.001] and 19.3 % [to 66.5 μm (±7.0 SD), p < 0.001] compared to the contralateral eye. Additionally, the ONL remained thickened at 96.7 μm (±7.0 SD, p < 0.001). At baseline, we found a significant correlation between the ONL thickness and the VF (r = −0.482, p = 0.005) and the BCVA at discharge (r = 0.552, p < 0.001), indicating that a thicker ONL correlates with poorer visual function. The GCL thickness also correlates with the BCVA at discharge (r = 0.411, p = 0.02), where a thinner GCL predicts worse BCVA. At the 1-month time point, the GCL thinning was correlated with both the VF (r = 0.471, p = 0.005) and the BCVA (r = −0.456, p = 0.007), indicating worse visual function.
Conclusions
Changes in the thickness of different layers of the retina occur early in the course of AION and evolve over time, resulting in the atrophy of the GCL and RNFL. ONL thickening at baseline is associated with visual dysfunction. Thinning of the GCL after 1 month correlates with poorer VF and BCVA at 1 month after acute AION.


Similar content being viewed by others
References
Miller NR (2011) Current Concepts in the Diagnosis, Pathogenesis, and Management of Nonarteritic Anterior Ischemic Optic Neuropathy. J Neuro-Ophthalmol 31:e1–e3
Hayreh SS (2013) Ischemic optic neuropathies - where are we now? Graefes Arch Clin Exp Ophthalmol 251:1873–1884
Lee MS, Grossman D, Arnold AC, Sloan FA (2011) Incidence of nonarteritic anteriorischemic optic neuropathy: increased risk among diabetic patients. Ophthalmology 118:959–963
Lee AG, Biousse V (2010) Should steroids be offered to patients with nonarteritic anteriorischemic optic neuropathy (NAION)? J Neuroophthalmol 30:193–198
Kardon R (2011) The Role of the Macula OCT Scan in Neuro-ophthalmology. J Neuroophthalmol 31:353–361
Contreras I, Noval S, Rebolleda G, Muñoz-Negrete FJ (2007) Follow-up of nonarteriticanterior ischemic optic neuropathy with optical coherence tomography. Ophthalmology 114:2338–2344
Hood DC, Anderson S, Rouleau J, Wenick AS, Grover LK, Behrens MM, Odel JG, Lee AG, Kardon RH (2008) Retinal nerve fiber structure versus visual field function in patients with ischemic optic neuropathy. A test of a linear model. Ophthalmology 115:904–910
Bellusci C, Savini G, Carbonelli M, Carelli V, Sadun AA, Barboni P (2008) Retinal nerve fiber layer thickness in nonarteritic anterior ischemic optic neuropathy:OCT characterization of the acute and resolving phases. Graefes Arch Clin Exp Ophthalmol 246:641–647
Kupersmith MJ, Anderson S, Durbin M, Kardon R (2013) Scanning laser polarimetry, but not optical coherence tomography predicts permanent visual field loss in acute nonarteritic anterior ischemic optic neuropathy. Invest Ophthalmol Vis Sci 54:5514–5519
Kernstock C, Beisse F, Wiethoff S, Mast A, Krapp E, Grund R, Dietzsch J, Lagrèze W, Fischer D, Schiefer U (2014) Assessment of functional and morphometric endpoints in patients with non-arteritic anterior ischemic optic neuropathy (NAION). Graefes Arch Clin Exp Ophthalmol 252:515–521
Papchenko T, Grainger BT, Savino PJ, Gamble GD, Danesh-Meyer HV (2012) Macular thickness predictive of visual field sensitivity in ischaemic optic neuropathy. Acta Ophthalmol 290:e463–e469
Gonul S, Koktekir BE, Bakbak B, Gedik S (2013) Comparison of the ganglion cell complex and retinal nerve fibre layer measurements using Fourier domain optical coherence tomography to detect ganglion cell loss in non-arteritic anterior ischaemic optic neuropathy. Br J Ophthalmol 97:1045–1050
Aggarwal D, Tan O, Huang D, Sadun AA (2012) Patterns of ganglion cell complex and nerve fiber layer loss in nonarteritic ischemic optic neuropathy by Fourier-domain optical coherence tomography. Invest Ophthalmol Vis Sci 53:4539–4545
The IONDT Research Group (1998) The ischemic optic neuropathy decompression trial (IONDT): design and methods. Control Clin Trials 19:276–296
Schulze-Bonsel K, Feltgen N, Burau H, Hansen L, Bach M (2006) Visual Acuities “Hand Motion” and “Counting Fingers”Can Be Quantified with the Freiburg Visual Acuity Test. Invest Ophthalmol Vis Sci 47:1236–1240
Oakley JD, Gabilondo I, Songster C, Russakoff D, Green A, Villoslada P. (2014) Assessing manual versus automated segmentation of the macula using Optical Coherence Tomography. ARVO Meeting abstract number 4790 - B0166
Gabilondo I, Sepúlveda M, Ortiz-Perez S, Fraga-Pumar E, Martínez-Lapiscina EH, Llufriu S, Solá N, Saiz A, Sanchez-Dalmau B, Villoslada P (2013) Retrograde retinal damage after acute optic tract lesion in MS. J Neurol Neurosurg Psychiatry 84:824–826
Ho JK, Stanford MP, Shariati MA, Dalal R, Liao YJ (2013) Optical coherence tomography study of experimental anterior ischemic optic neuropathy and histologic confirmation. Invest Ophthalmol Vis Sci 54:5981–5988
Keller J, Sánchez-Dalmau BF, Villoslada P (2014) Lesions in the posterior visual pathway promote trans-synaptic degeneration of retinal ganglion cells. PLoS One 9:e97444
Hayreh SS, Zimmerman MB (2008) Non-arteritic anterior ischemic optic neuropathy: role of systemic corticosteroid therapy. Graefes Arch Clin Exp Ophthalmol 246:1029–1046
Rebolleda G, Pérez-López M, Casas-LLera P, Contreras I, Muñoz-Negrete FJ (2013) Visual and anatomical outcomes of non-arteritic anterior ischemic optic neuropathy with high-dose systemic corticosteroids. Graefes Arch Clin Exp Ophthalmol 251:255–260
Acknowledgments
This work was supported by grants from the Instituto de Salud Carlos III, Spain (FIS programme PS09/00259 and RETICS programme RD07/0060/01) to PV. Funding agencies had no role in design and conduct of the study; collection, management, analysis, and interpretation of the data; or preparation, review, or approval of the manuscript.
Conflict of Interest
Jonathan Oakley and Daniel Russakoff are co-founders of Voxeleron LLC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keller, J., Oakley, J.D., Russakoff, D.B. et al. Changes in macular layers in the early course of non-arteritic ischaemic optic neuropathy. Graefes Arch Clin Exp Ophthalmol 254, 561–567 (2016). https://doi.org/10.1007/s00417-015-3066-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3066-3