Abstract
Purpose
Our aim was to analyze retinal structure in young patients with Best disease with reference to future gene therapy.
Methods
This was a retrospective observational spectral domain optical coherence tomography study of four patients aged 10 years or less with Best disease.
Results
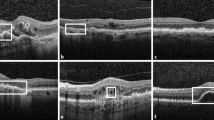
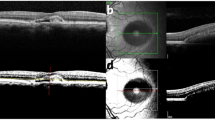
Findings ranged from subtle thickening at the level of the retinal pigment epithelium-photoreceptor interdigitation line, to subretinal fluid and precipitate-like changes at the level of the photoreceptor outer segments, and further to choroidal neovascularization. The photoreceptor inner segment ellipsoid layer could be visualized seemingly undisturbed above the vitelliform lesions, except in the case of choroidal neovascularization.
Conclusions
Clinical variability is evident even among young patients aged 10 years or less with Best disease. The earliest structural alterations seem to occur at the level of the retinal pigment epithelium-photoreceptor interdigitation line. The photoreceptor inner segment seems to be unaffected unless choroidal neovascularization develops, which seems promising regarding future gene therapy.




Similar content being viewed by others
References
MacDonald IM, Lee T. Best vitelliform macular dystrophy. In: GeneReviews [Internet]. Available at: http://www.ncbi.nlm.nih.gov/books/NBK1167/. Accessed: December 4, 2013.
Yardley J, Leroy BP, Hart-Holden N et al (2004) Mutations of VMD2 splicing regulators cause nanophthalmos and autosomal dominant vitreoretinochoroidopathy (ADVIRC). Invest Ophthalmol Vis Sci 45:3683–3689
Bitner H, Mizrahi-Meissonnier L, Griefner G, Erdinest I, Sharon D, Banin E (2011) A homozygous frameshift mutation in BEST1 causes the classical form of Best disease in an autosomal recessive mode. Invest Ophthalmol Vis Sci 52:5332–5338
Burgess R, Millar ID, Leroy BP et al (2008) Biallelic mutation of BEST1 causes a distinct retinopathy in humans. Am J Hum Genet 82:19–31
Burgess R, MacLaren RE, Davidson AE et al (2009) ADVIRC is caused by distinct mutations in BEST1 that alter pre-mRNA splicing. J Med Genet 46:620–625
Bitner H, Schatz P, Mizrahi-Meissonnier L, Sharon D, Rosenberg T (2012) Frequency, genotype, and clinical spectrum of best vitelliform macular dystrophy: data from a national center in Denmark. Am J Ophthalmol 154:403–412
Petrukhin K, Koisti MJ, Bakall B et al (1998) Identification of the gene responsible for Best macular dystrophy. Nat Genet 19:241–247
Gomez NM, Tamm ER, Straubeta O (2013) Role of bestrophin-1 in store-operated calcium entry in retinal pigment epithelium. Pflugers Arch 465:481–495
Guziewicz KE, Slavik J, Lindauer SJ, Aguirre GD, Zangerl B (2011) Molecular consequences of BEST1 gene mutations in canine multifocal retinopathy predict functional implications for human bestrophinopathies. Invest Ophthalmol Vis Sci 52:4497–4505
Schatz P, Bitner H, Sander B et al (2010) Evaluation of macular structure and function by OCT and electrophysiology in patients with vitelliform macular dystrophy due to mutations in BEST1. Invest Ophthalmol Vis Sci 51:4754–4765
Piñeiro-Gallego T, Álvarez M, Pereiro I et al (2011) Clinical evaluation of two consanguineous families with homozygous mutations in BEST1. Mol Vis 17:1607–1617
Andersen MK, Christoffersen NL, Sander B et al (2010) Oligocone trichromacy: clinical and molecular genetic investigations. Invest Ophthalmol Vis Sci 51:89–95
Wittstrom E, Ekvall S, Schatz P, Bondeson ML, Ponjavic V, Andreasson S (2011) Morphological and functional changes in multifocal vitelliform retinopathy and biallelic mutations in BEST1. Ophthalmic Genet 32:83–96
Wang M, Sander B, la Cour M, Larsen M (2005) Clinical characteristics of subretinal deposits in central serous chorioretinopathy. Acta Ophthalmol Scand 83:691–696
Singh R, Shen W, Kuai D et al (2013) iPS cell of Best disease: insights into the pathophysiology of an inherited macular degeneration. Hum Mol Genet 22:593–607
Chung MM, Oh KT, Streb LM, Kimura AE, Stone EM (2001) Visual outcome following subretinal hemorrhage in Best disease. Retina 21:575–580
Frennesson CI, Wadelius C, Nilsson SE (2014) Best vitelliform macular dystrophy in a Swedish family: genetic analysis and a seven-year follow-up of photodynamic treatment of a young boy with choroidal neovascularization. Acta Ophthalmol 92(3):238–242
Iannaccone A, Kerr NC, Kinnick TR, Calzada JI, Stone EM (2011) Autosomal recessive best vitelliform macular dystrophy: report of a family and management of early-onset neovascular complications. Arch Ophthalmol 129:211–217
Querques G, Zerbib J, Santacroce R et al (2011) The spectrum of subclinical Best vitelliform macular dystrophy in subjects with mutations in BEST1 gene. Invest Ophthalmol Vis Sci 52:4678–4684
Querques G, Zerbib J, Georges A et al (2014) Multimodal analysis of the progression of Best vitelliform macular dystrophy. Mol Vis 27:575–592
Ferrara DC, Costa RA, Tsang S, Calucci D, Jorge R, Freund KB (2010) Multimodal fundus imaging in Best vitelliform macular dystrophy. Graefes Arch Clin Exp Ophthalmol 248:1377–1386
Manes G, Meunier I, Avila-Fernández A et al (2013) Mutations in IMPG1 cause macular dystrophies. Am J Hum Genet 93:571–578
Meunier I, Manes G, Bocquet B et al (2014) Frequency and clinical pattern of vitelliform macular dystrophy caused by mutations of interphotoreceptor matrix IMPG1 and IMPG2 genes. Ophthalmology 121:2406–2414
Acknowledgments
We appreciate the technical assistance with imaging modalities provided by Ms. Hajer Ahmad Al-Abaiji at the Department of Ophthalmology, Glostrup Hospital, Glostrup, Denmark and Mr Johnny Ring at the Department of Ophthalmology, Clinical Sciences, Lund University, Sweden.
Funding/support
Grants from the Cronqvist Stiftelse of the Swedish Society of Medicine, Eye Foundation, Dag Lenard Foundation, and Foundation for the Visually Impaired in Skane County.
Contributions to authors
Design of the study (PS, DS, SAH, SA, ML).
Conduct of the study (PS, DS, SAH, SA, ML).
Collection, management, analysis, and interpretation of the data (PS, DS, SAH, SA, ML).
Preparation, review and approval of the manuscript (PS, DS, SAH, SA, ML).
Statement
Patrik Schatz had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis
Conflict of interest
All authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Grant support is listed in the Acknowledgements section. None of the authors have any propriety interest in the materials described in the manuscript.
Rights and permissions
About this article
Cite this article
Schatz, P., Sharon, D., Al-Hamdani, S. et al. Retinal structure in young patients aged 10 years or less with Best vitelliform macular dystrophy. Graefes Arch Clin Exp Ophthalmol 254, 215–221 (2016). https://doi.org/10.1007/s00417-015-3025-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3025-z